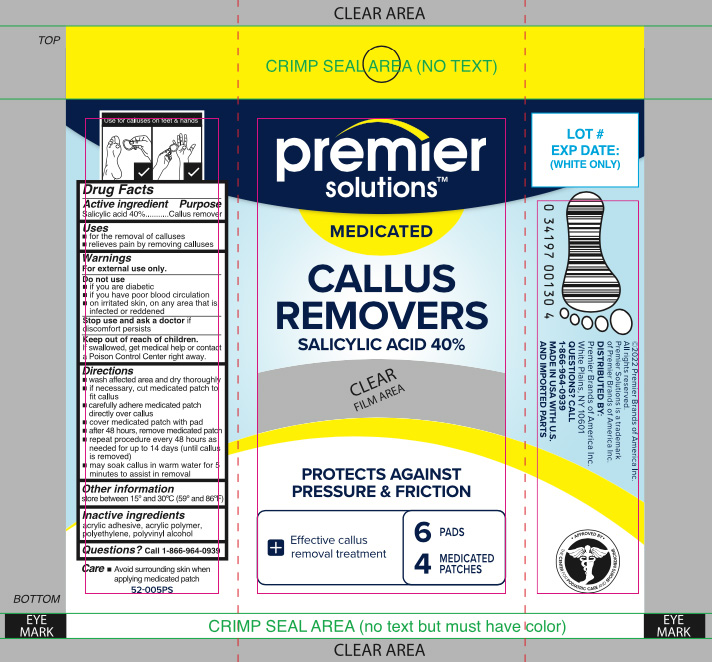
Label: SALICYLIC ACID- callus removers patch
- NDC Code(s): 56104-244-01
- Packager: Premier Brands of America Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated February 1, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Use
- Warnings
-
Directions
- wash affected area and dry thoroughly
- if necessary, cut medicated patch to fit callus
- carefully adhere medicated patch directly over callus
- cover medicated patch with pad
- after 48 hours, remove medicated patch
- repeat procedure every 48 hours as needed for up to 14 days (until callus is removed)
- may soak callus in warm water for 5 minutes to assist in removal
- Other information
- Inactive ingredients
- Questions?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SALICYLIC ACID
callus removers patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:56104-244 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 40 mg in 4 Inactive Ingredients Ingredient Name Strength POLYVINYL ALCOHOL (UNII: 532B59J990) VINYL ACETATE (UNII: L9MK238N77) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:56104-244-01 4 in 1 PACKAGE; Type 0: Not a Combination Product 06/16/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M030 06/16/2022 Labeler - Premier Brands of America Inc. (117557458)