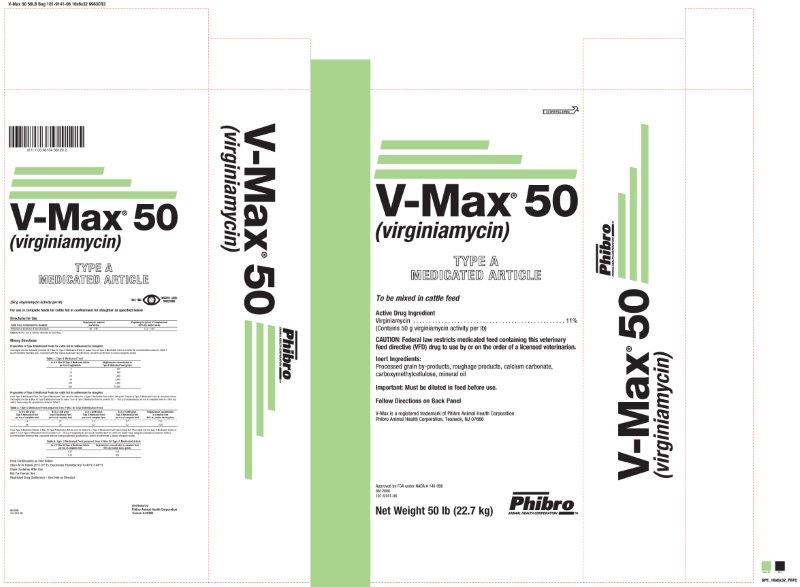
Label: V-MAX 50- virginiamycin powder
- NDC Code(s): 66104-9601-1
- Packager: Phibro Animal Health
- Category: VFD TYPE A MEDICATED ARTICLE ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated November 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION
- Active Drug Ingredient
-
INDICATIONS & USAGE
Important: Must be diluted in feed before use
Follow Directions on Back Panel
V-Max is a registered trademark of Phibro Animal Health Corporation
Phibro Animal Health Corporation, Teaneck, NJ 07666
Approved by FDA under NADA # 140-998
8812000
101-9141-06
NET CONTENTS: 50 lb (22.7 kg)
Distributed by:
PHIBRO ANIMAL HEALTH, INC.
Teaneck, NJ 07666, USA
- (50 g virginiamycin activity per lb)For use in complete feeds for cattle fed in confinement for slaughter as specified belowDirections for Use
-
Mixing Directions
Preparation ofType B Medicated Feeds for cattle fed in confinement for slaughter—Thoroughly mix the following amounts of V-Max 50 Type A Medicated Article to make 1 ton of Type B Medicated Feed to provide the concentrations shown in Table 1. An intermediate blending step, consistent with the mixing equipment specifications, should be performed to ensure adequate mixing.
Table 1. Type B Medicated Feed
lb of V-Max 50 Type A Medicated Article per ton of supplement
Virginiamycin concentration in Type B Medicated Feed (g/ton)
5
10
20
40
100
200
250
500
1,000
2,000
5,000
10,000
Preparation of Type C Medicated Feed for cattle fed in confinement for slaughter:
From Type B Medicated Feed: The Type B Medicated Feed must be diluted to a Type C Medicated Feed before being fed. Prepare a Type B Medicated Feed as described above. Thoroughly mix the V-Max 50 Type B Medicated Feed to make 1 ton of Type C Medicated Feed to provide 13.5 16.0 g of virginiamycin per ton of complete feed on a 90% dry matter basis using the examples provided in Table 2.
Table 2. Type C Medicated Feed prepared from V-Max 50 Type B Medicated Feed
lb of a 500g/ton Type B Medicated Feed per ton of complete feed
lb of a 1,000 g/ton Type B Medicated Feed per ton of complete feed
lb of a 5,000 g/ton Type B Medicated Feed per ton of complete feed
lb of a 10,000 g/ton Type B Medicated Feed per ton of complete feed
Virginiamycin concentration in complete feed 90% dry matter basis (g/ton)
54
27
5.4
2.7
13.5
64
32
6.4
3.2
16.0
From Type A Medicated Article: V-Max 50 Type A Medicated Article must be diluted to a Type C Medicated Feed before being fed. Thoroughly mix the Type A Medicated Article to make 1 ton of Type C Medicated Feed to provide 13.5 – 16.0 g of virginiamycin per ton of complete feed on a 90% dry matter basis using the examples provided in Table 3.
An intermediate blending step, consistent with the blending equipment specifications, should be performed to ensure adequate mixing.
Table 3. Type C Medicated Feed prepared from V-Max 50 Type A Medicated Article
lb of V-Max 50 Type A Medicated Article per ton of complete feed
Virginiamycin concentration in complete feed 90% dry matter basis (g/ton)
0.27
0.32
13.5
16.0
Feed continuously as sole ration
- Store at or Below 25°C/77F°, Excursions Permitted Up To 40oC (104oF)
- INDICATIONS & USAGE
- V-Max 50
-
INGREDIENTS AND APPEARANCE
V-MAX 50
virginiamycin powderProduct Information Product Type VFD TYPE A MEDICATED ARTICLE ANIMAL DRUG Item Code (Source) NDC:66104-9601 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VIRGINIAMYCIN (UNII: C49WS9N75L) (VIRGINIAMYCIN - UNII:C49WS9N75L) VIRGINIAMYCIN 227 g in 0.45 kg Inactive Ingredients Ingredient Name Strength CALCIUM CARBONATE (UNII: H0G9379FGK) MINERAL OIL (UNII: T5L8T28FGP) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66104-9601-1 22.7 kg in 1 BAG Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA140998 04/01/2010 Labeler - Phibro Animal Health (006989008) Registrant - Phibro Animal Health (006989008)