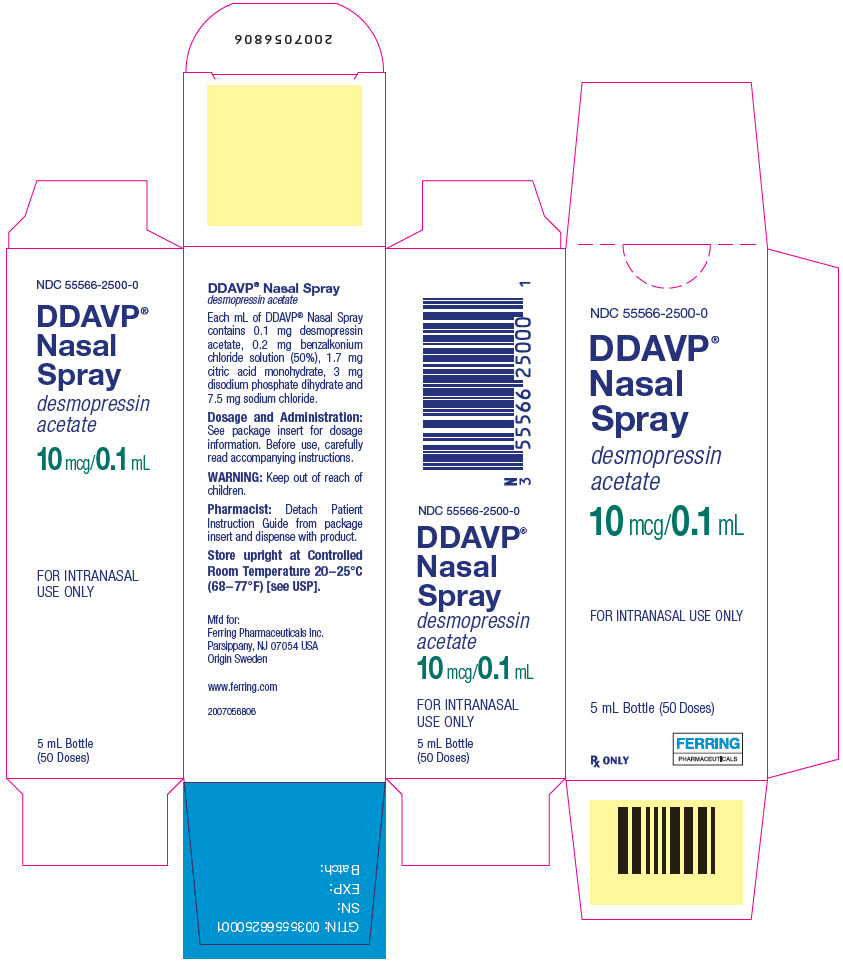
DDAVP- desmopressin acetate spray
Ferring Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use DDAVP NASAL SPRAY safely and effectively. See full prescribing information for DDAVP NASAL SPRAY.
DDAVP (desmopressin acetate) nasal spray Initial U.S. Approval: 1978 INDICATIONS AND USAGEDDAVP Nasal Spray is a vasopressin analog indicated as antidiuretic replacement therapy in the management of central diabetes insipidus for adults and pediatric patients 4 years of age and older (1)
Limitations of Use:
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSNasal Spray: 10 mcg per 0.1 mL spray, available in a 5 mL bottle with spray pump delivering 50 sprays (3) CONTRAINDICATIONSWARNINGS AND PRECAUTIONSADVERSE REACTIONSAdverse reactions that have been identified in patients administered DDAVP Nasal Spray are headache, nasal congestion, rhinitis, nosebleed, sore throat, cough, upper respiratory infections, nausea, flushing, and mild abdominal cramps (6) To report SUSPECTED ADVERSE REACTIONS, contact Ferring Pharmaceuticals Inc. at 1-888-FERRING (1-888-337-7464) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSUSE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 10/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
DDAVP Nasal Spray is indicated as antidiuretic replacement therapy in the management of central diabetes insipidus in adults and pediatric patients 4 years of age and older.
Limitations of Use:
DDAVP Nasal Spray is not indicated for:
- Treatment of nephrogenic diabetes insipidus,
- Treatment of primary nocturnal enuresis [see Warnings and Precautions (5.1)],
- Use in patients with conditions that compromise the intranasal route of administration (e.g., severe nasal congestion and blockage, nasal mucosa atrophy, severe atrophic rhinitis, recent nasal surgery such as transsphenoidal hypophysectomy) [see Warnings and Precautions (5.2)].
- Use in patients with an impaired level of consciousness,
- Use in patients requiring doses less than 10 mcg or doses that are not multiples of 10 mcg [see Dosage Forms and Strengths (3)].
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
Administer DDAVP Nasal Spray by intranasal use only. Instruct patients about appropriate fluid restriction during DDAVP Nasal Spray treatment [see Warnings and Precautions (5.1)].
Must prime the spray pump prior to the first use. Instruct patients to:
- Prime pump by pressing down on pump four times (if the spray pump is not used for one week, re-prime the pump by pressing down on the pump once).
- Discard DDAVP Nasal Spray after 50 sprays since the amount delivered thereafter may be substantially less than the recommended dosage.
2.2 Recommended Dosage
The use of DDAVP Nasal Spray is not indicated for patients who require less than 10 mcg doses or doses that are not multiples of 10 mcg because the spray pump can only deliver doses of 10 mcg [see Indications and Usage (1)]. If other doses are required, use another desmopressin acetate product.
Individualize the dosage of DDAVP Nasal Spray for each patient with particular attention in pediatric and elderly patients and adjust according to the diurnal pattern of response to limit nocturia and to ensure fluid intake with respect to urine output is not excessive [see Warnings and Precautions (5.1)]. Monitor continued response to DDAVP Nasal Spray by urine volume and osmolality to ensure adequate diuresis to limit the risk of hyponatremia, and include measurements of serum sodium and plasma osmolality as needed.
Adults
The recommended dosage in adults is 10 mcg once daily into one nostril up to 40 mcg once daily (or 40 mcg divided into two or three daily doses). If administered more than once a day, adjust for an adequate diurnal rhythm of urine output.
Pediatric Patients
- For pediatric patients requiring doses less than 10 mcg, DDAVP Nasal Spray is not indicated.
- For pediatric patients 4 years of age and older, the recommended starting dosage of DDAVP Nasal Spray is 10 mcg once daily into one nostril. The dose can be titrated up to 30 mcg once daily (or 30 mcg divided into two daily doses, typically with 20 mcg given in the morning and 10 mcg given at nighttime). If administered more than once a day, adjust for an adequate diurnal rhythm of urine output.
Because administration of desmopressin acetate can been associated with decreased responsiveness with prolonged use, consider increasing the dosage of DDAVP Nasal Spray if patients demonstrate decreased response over a long period of time.
2.3 Switching Between Desmopressin Acetate Formulations
When switching from the desmopressin acetate injection to DDAVP Nasal Spray, administer 10 times the amount of desmopressin acetate, rounding down to the nearest 10 mcg.
When switching from the desmopressin acetate tablets to DDAVP Nasal Spray individual dose titration is required because intranasal desmopressin is approximately 10 to 40 fold more potent than oral (tablet) desmopressin.
3 DOSAGE FORMS AND STRENGTHS
DDAVP Nasal Spray pump delivers 10 mcg (0.1 mL) of desmopressin acetate per spray.
DDAVP Nasal Spray is available as a 5 mL bottle with spray pump delivering 50 sprays.
4 CONTRAINDICATIONS
DDAVP Nasal Spray is contraindicated in patients with:
- Known hypersensitivity to desmopressin acetate or to any of the components of DDAVP Nasal Spray. Severe allergic reactions and anaphylaxis have been reported [see Adverse Reactions (6)].
- Renal impairment defined as estimated creatinine clearance (CLcr) by Cockcroft-Gault equation less than 50 mL/min [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
- Hyponatremia or a history of hyponatremia [see Warnings and Precautions (5.1)].
5 WARNINGS AND PRECAUTIONS
5.1 Hyponatremia
Excessive fluid intake when urine output is limited by the antidiuretic effect of desmopressin may lead to water intoxication with hyponatremia. Cases of hyponatremia have been reported from postmarketing experience in patients treated with desmopressin acetate. Unless properly diagnosed and treated, hyponatremia can be fatal.
All patients receiving DDAVP Nasal Spray should be observed for the following signs or symptoms associated with hyponatremia: headache, nausea/vomiting, decreased serum sodium, weight gain, restlessness, fatigue, lethargy, disorientation, depressed reflexes, loss appetite, irritability, muscle weakness, muscle spasms or cramps and abnormal mental status such as hallucinations, decreased consciousness, and confusion. Severe symptoms due to an extreme decrease in serum sodium and plasma osmolality may include one or a combination of the following: seizure, coma, and/or respiratory arrest.
In order to decrease the risk of water intoxication with hyponatremia, fluid restriction is recommended. Careful fluid intake restriction is particularly important in pediatric and geriatric patients because these patients are at greater risk of developing hyponatremia [see Use in Specific Populations (8.4, 8.5)]. More frequent monitoring of serum sodium levels is recommended in the following patients: those with conditions associated with fluid and electrolyte imbalance, such as cystic fibrosis, heart failure, renal disorders, habitual or psychogenic polydipsia or those taking concomitant drugs that may cause hyponatremia [see Drug Interactions (7.1)].
DDAVP Nasal Spray is not an indicated formulation for the treatment of primary nocturnal enuresis due to a higher risk of hyponatremia and hyponatremic convulsions with the use of the nasal spray formulation compared to desmopressin tablets seen in postmarketing reports [see Indications and Usage (1)].
5.2 Altered Absorption in Patients with Nasal Mucosa Abnormalities
Chronic administration of DDAVP Nasal Spray may result in changes to nasal mucosa. Nasal mucosa abnormalities (such as scarring and edema) due to chronic administration, or due to other causes (nasal blockage, nasal mucosal atrophy, severe atrophic rhinitis, recent nasal surgery such as transsphenoidal hypophysectomy) may cause erratic, unreliable absorption. Avoid use of DDAVP Nasal Spray in such patients [see Indications and Usage (1)] and consider use of other formulations of desmopressin acetate given by other routes of administration.
6 ADVERSE REACTIONS
The following serious reactions are described below and elsewhere in the labeling:
- Hyponatremia [see Warnings and Precautions (5.1)].
- Altered Absorption in Patients with Changes in Nasal Mucosa [see Warnings and Precautions (5.2)].
The following adverse reactions have been identified during post-approval use of desmopressin acetate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Increase in blood pressure, headache, nasal congestion, rhinitis, nosebleed, sore throat, cough, upper respiratory infections, nausea, flushing, and abdominal cramps.
Water intoxication with hyponatremia
Hyponatremic convulsions associated with concomitant use of the following medications: oxybutinin and imipramine [see Drug Interactions (7.1)].
Severe allergic reactions and anaphylaxis [see Contraindications (4)]
7 DRUG INTERACTIONS
7.1 Other Drugs that may Increase Risk of Hyponatremia
The concomitant administration of DDAVP Nasal Spray with other drugs that may increase the risk of water intoxication with hyponatremia, (e.g., tricyclic antidepressants, selective serotonin re-uptake inhibitors, chlorpromazine, opiate analgesics, NSAIDs, lamotrigine, oxybutynin and carbamazepine) requires more frequent serum sodium monitoring [see Warnings and Precautions (5.1) and Adverse Reactions (6)].
7.2 Other Vasoconstrictors
Desmopressin acetate can elevate blood pressure. Use of large doses of DDAVP Nasal Spray with other vasocontrictors may require a reduction of the DDAVP dosage [see Adverse Reactions (6)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk summary
Prolonged experience with desmopressin in pregnant women over several decades, based on the available published data and case reports, did not identify a drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. In addition, in vitro studies with human placenta demonstrate poor placental transfer of desmopressin. No adverse developmental outcomes were observed in animal reproduction studies with administration of desmopressin during organogenesis to pregnant rats and rabbits at doses approximately <1 and 38 times, respectively, the maximum recommended human dose based on body surface area (mg/m2) (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population are unknown. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Desmopressin acetate at up to 50 ng/kg/day was given by subcutaneous injection to pregnant rats, from gestation day 1 to 20 during the period of early embryonic development and organogenesis without teratogenic effects. Desmopressin acetate at up to 10 mcg/kg/day was given to pregnant rabbits by subcutaneous injection from gestation day 6 to 18 during fetal organogenesis without teratogenic effects. These doses of desmopressin acetate represent approximately <1 times (rat) and 38 times (rabbit) the maximum recommended human dose based on body surface area (mg/m2).
8.2 Lactation
Risk Summary
Breastfeeding is not expected to result in clinically relevant exposure of the infant to desmopressin following maternal intranasal administration. Desmopressin is poorly transferred into human breastmilk at negligible amounts (see Data). There is no information on the effects of desmopressin on the breastfed infant or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for DDAVP Nasal Spray and any potential adverse effects on the breastfed infant from DDAVP Nasal Spray or from the underlying maternal condition.
Data
A trial was conducted in six healthy lactating women, at greater than 4 months postpartum, to evaluate intranasal administration of 300 mcg single dose of another desmopressin product (7.5 times the recommended adult dose of DDAVP Nasal Spray). Samples of maternal plasma and breastmilk were obtained at 0, 30, 60, 120, 240, 360 and 480 min after the drug administration. At 8 hours after dose intake, the levels in the milk ranged between 4.16 and 101 pg/ml, and the plasma levels ranged between 40 and 242 pg/ml. The total amount of desmopressin present in the milk over the 8 hours ranged between 491 pg and 16 ng, which corresponds to 0.0001 - 0.005% of the administered dose to the breastfeeding mother.
8.4 Pediatric Use
DDAVP Nasal Spray is indicated as antidiuretic replacement therapy in the management of central diabetes insipidus in pediatric patients 4 years of age and older. DDAVP Nasal Spray is not indicated in pediatric patients less than 4 years of age.
Use of DDAVP Nasal Spray in pediatric patients 4 years of age and older is supported by evidence from adults and pediatric patients with central diabetes insipidus. Use in pediatric patients requires careful fluid intake restriction to prevent possible water intoxication with hyponatremia [see Warnings and Precautions (5.1)].
8.5 Geriatric Use
Clinical studies of DDAVP Nasal Spray did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at a low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
Because elderly patients are more likely to have renal impairment, care should be taken in dose selection, and monitoring renal function is recommended [see Contraindications (4), Use in Specific Populations (8.6)].
Use of DDAVP Nasal Spray in geriatric patients requires careful fluid intake restriction to prevent possible water intoxication with hyponatremia [see Warnings and Precautions (5.1)].
8.6 Renal Impairment
Desmopressin acetate is substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with renal impairment than patients with normal renal function. DDAVP Nasal Spray is contraindicated in patients with estimated CLcr by Cockcroft-Gault equation less than 50 mL/min [see Clinical Pharmacology (12.1, 12.3), Contraindications (4)].
10 OVERDOSAGE
Signs of desmopressin acetate overdosage may include confusion, drowsiness, continuing headache, problems with passing urine, and rapid weight gain due to fluid retention [see Warnings and Precautions (5.1)]. In case of overdosage, reduce the dosage, decrease the frequency of administration, or discontinue DDAVP Nasal Spray. There is no known specific antidote for desmopressin acetate.
11 DESCRIPTION
DDAVP Nasal Spray is a vasopressin analogue of the natural pituitary hormone 8-arginine vasopressin (ADH), an antidiuretic hormone affecting renal water conservation. It is chemically defined as follows:
Molecular weight: 1183.34
Empirical formula: C46H64N14O12S2∙C2H4O2∙3H2O

1-(3-mercaptopropionic acid)-8-D-arginine vasopressin monoacetate (salt) trihydrate.
DDAVP Nasal Spray is an aqueous solution for intranasal use. Each mL contains:
- Desmopressin acetate 0.1 mg
- Sodium chloride 7.5 mg
- Citric acid monohydrate 1.7 mg
- Disodium phosphate dihydrate 3 mg
- Benzalkonium chloride solution (50%) 0.2 mg
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The antidiuretic effects of desmopressin are mediated by stimulation of vasopressin 2 (V2) receptors, thereby increasing water re-absorption in the kidney, and hence reducing urine production. Desmopressin is a replacement hormone for antidiuretic hormone in the treatment of central diabetes insipidus. The change in structure of arginine vasopressin to desmopressin acetate resulted in a decreased vasopressor action and decreased actions on visceral smooth muscle relative to the enhanced antidiuretic activity, so that clinically effective antidiuretic doses were usually below threshold levels for effects on vascular or visceral smooth muscle.
12.2 Pharmacodynamics
The use of DDAVP Nasal Spray in patients with central diabetes insipidus reduces urinary output, increases urine osmolality, and decreases plasma osmolality.
12.3 Pharmacokinetics
Elimination: Desmopressin acetate exhibits a biphasic elimination profile, with half-lives of 7.8 and 75.5 minutes for the initial and terminal phases, respectively.
Specific Populations
Renal Impairment: Desmopressin acetate is mainly excreted in the urine. A pharmacokinetic study was conducted in subjects with normal renal function and patients with mild, moderate, and severe renal impairment (n=24, 6 subjects each group) with a single 2 mcg dose of desmopressin acetate injection (this results in approximately 20 times the exposure of 10 mcg of DDAVP Nasal Spray). The terminal half-life was 2.8 hours in subjects with normal renal function, 4.0 hours in mild renal impairment, 6.6 hours in moderate renal impairment and 8.7 hours in severe renal impairment. In patients with mild, moderate and severe renal impairment, mean desmopressin exposure was 1.5 fold, 2.4 fold and 3.6 fold higher, respectively compared to that of subjects with normal renal function [see Contraindications (4), Use in Specific Populations (8.6)].
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use)
Administration
- Inform caregivers for pediatric patients that administration should be supervised to ensure the patient receives the prescribed dose.
- Inform patients that the pump must be primed prior to first use and again if not used for greater than one week. Inform patients that the DDAVP Nasal Spray bottle delivers 50 sprays of 10 mcg each following the initial 4 priming pumps.
- Inform patients to discard any solution remaining after 50 sprays since the amount delivered thereafter may be substantially less than 10 mcg of drug.
U.S. Pat. Nos. 5,500,413, 5,596,078, and 5,674,850
Manufactured for:
Ferring Pharmaceuticals Inc.
Parsippany, NJ 07054 USA
Origin Sweden
XXXXXXXXXX
| This Patient Information has been approved by the Food and Drug Administration | 09/18 | ||
| Patient Information DDAVP (phonetic spelling) Nasal Spray, 10 mcg per 0.1 mL for Intranasal Use Only |
|||
| What is DDAVP? | |||
| DDAVP is a prescription medicine called a vasopressin analog used as an antidiuretic replacement to manage central diabetes insipidus. | |||
|
|||
| Who should not use DDAVP? | |||
| Do not use DDAVP if you: | |||
|
|||
| What should I tell my healthcare provider before using DDAVP? Before using DDAVP, tell your healthcare provider about all your medical conditions including if you: |
|||
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| How should I use DDAVP? | |||
|
|||
| What are the possible side effects of DDAVP? | |||
| DDAVP may cause serious side effects including: | |||
|
|||
|
|
|
|
|
|||
| The most common side effects of DDAVP include: headache, stuffy nose, runny nose, nosebleed, sore throat, cough, upper respiratory infections, nausea, flushing, and stomach cramps. | |||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
| General information about the safe and effective use of DDAVP. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DDAVP for a condition for which it was not prescribed. Do not give DDAVP to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about DDAVP that is written for health professionals. For more information call 1-888-FERRING (1-888-337-7464). | |||
| What are the ingredients in DDAVP? | |||
| Active ingredient: desmopressin acetate | |||
| Inactive ingredients: benzalkonium chloride solution (50%), sodium chloride, citric acid monohydrate, disodium phosphate dihydrate, purified water | |||
Instructions for Use
DDAVP
Nasal Spray, 10 mcg per 0.1 mL
For Intranasal Use Only
Read these instructions before using DDAVP Nasal Spray, and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
Children should be helped by an adult when using DDAVP Nasal Spray, to make sure the right amount of medicine is used.
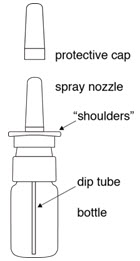
The parts of your DDAVP Nasal Spray pump (see Figure A):
Priming your DDAVP Nasal Spray:
Your DDAVP Nasal Spray pump must be primed before you use it for the first time.
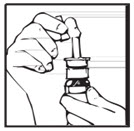
- Remove the protective cap (see Figure B).
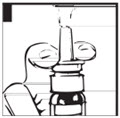
- Press down on the shoulders at the top of your DDAVP Nasal Spray pump 4 times. Hold the spray tip away from your face and eyes (see Figure C).
- After your DDAVP Nasal Spray pump is primed, it will spray 10 micrograms (1 dose) of medicine each time it is pressed.
Using your DDAVP Nasal Spray:
Step 1. Remove the protective cap.
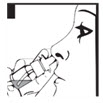
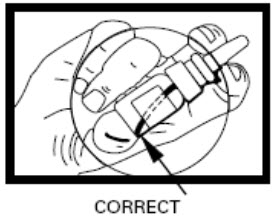
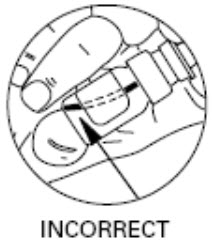
Step 2. To make sure you get the right dose of medicine tilt your DDAVP Nasal Spray pump so the dip tube inside the bottle draws the medicine up from the deepest part of the medicine inside the bottle (see Figures D and E).
 |  |
|
Figure D |
Figure E |
Step 3. Put the spray nozzle tip of your DDAVP Nasal Spray into your nostril and press the spray pump 1 time for 1 dose. (see Figure F). If 2 doses are prescribed, spray each nostril 1 time.
Step 4. Put the protective cap back on the spray nozzle tip when you finish using your DDAVP Nasal Spray.
Keeping track of your Nasal Sprays:
- Use the check-off chart to help you keep track of your DDAVP Nasal Sprays used (see Figure G).
| DDAVP Nasal Spray |
|---|
| 50-Spray Check-off Chart |
 |
| Figure G |
- Keep this chart with your DDAVP Nasal Spray or put it someplace where you can easily get it.
- Check off number 1 on the chart with your first dose of DDAVP Nasal Spray. Check off the numbers after each use of your DDAVP Nasal Spray. If your healthcare provider prescribed a 2-spray dose, then 2 numbers should be checked off.
- Your DDAVP Nasal Spray holds 50 sprays with the right amount of medicine in each spray.
- If any medicine is left in your DDAVP Nasal Spray after 50 sprays, do not use it. You may not get the right amount of medicine.
- Throw away your DDAVP Nasal Spray after 50 sprays.
- Do not count the priming sprays. Your DDAVP Nasal Spray has been filled with extra medicine for your priming sprays.
- Do not try to remove any medicine from your DDAVP Nasal Spray pump and place it in another bottle.
How should I store DDAVP Nasal Spray?
- Store DDAVP Nasal Spray at room temperature between 68°F to 77°F (20°C to 25°C).
- Store DDAVP Nasal Spray standing upright.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Ferring Pharmaceuticals Inc.
Parsippany, NJ 07054 USA
Origin Sweden
Rev. 09/18
XXXXXXXXXX
| DDAVP
desmopressin acetate spray |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Ferring Pharmaceuticals Inc. (103722955) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ferring GmbH | 328609615 | MANUFACTURE(55566-2500) , ANALYSIS(55566-2500) , PACK(55566-2500) , LABEL(55566-2500) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Rechon Life Science AB | 775207769 | MANUFACTURE(55566-2500) , ANALYSIS(55566-2500) , PACK(55566-2500) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ferring International Center SA | 481210362 | PACK(55566-2500) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| PolyPeptide Laboratories (Sweden) AB | 356580779 | API MANUFACTURE(55566-2500) , ANALYSIS(55566-2500) | |