ZOFRAN- ondansetron hydrochloride injection
GlaxoSmithKline LLC
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ZOFRAN INJECTION safely and effectively. See full prescribing information for ZOFRAN INJECTION.
ZOFRAN® (ondansetron hydrochloride) injection, for intravenous or intramuscular use Initial U.S. Approval: 1991 RECENT MAJOR CHANGES
INDICATIONS AND USAGEDOSAGE AND ADMINISTRATIONPrevention of Nausea and Vomiting Associated With Initial and Repeat Courses of Emetogenic Cancer Chemotherapy (2.1):
Prevention of Postoperative Nausea and/or Vomiting (2.2):
Patients With Severe Hepatic Impairment (2.3):
DOSAGE FORMS AND STRENGTHSInjection: 40 mg/20 mL multiple-dose vial. (3) CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSChemotherapy-Induced Nausea and Vomiting:
Postoperative Nausea and/or Vomiting:
To report SUSPECTED ADVERSE REACTIONS, contact Novartis Pharmaceuticals Corporation at 1-888-669-6682 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See 17 for PATIENT COUNSELING INFORMATION. Revised: 10/2021 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Prevention of Nausea and Vomiting Associated With Initial and Repeat Courses of Emetogenic Cancer Chemotherapy
ZOFRAN® Injection is indicated for the prevention of nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including high-dose cisplatin.
ZOFRAN is approved for patients aged 6 months and older.
1.2 Prevention of Postoperative Nausea and/or Vomiting
ZOFRAN Injection is indicated for the prevention of postoperative nausea and/or vomiting. As with other antiemetics, routine prophylaxis is not recommended for patients in whom there is little expectation that nausea and/or vomiting will occur postoperatively. In patients in whom nausea and/or vomiting must be avoided postoperatively, ZOFRAN Injection is recommended even when the incidence of postoperative nausea and/or vomiting is low. For patients who do not receive prophylactic ZOFRAN Injection and experience nausea and/or vomiting postoperatively, ZOFRAN Injection may be given to prevent further episodes.
ZOFRAN is approved for patients aged 1 month and older.
2 DOSAGE AND ADMINISTRATION
2.1 Prevention of Nausea and Vomiting Associated With Initial and Repeat Courses of Emetogenic Chemotherapy
Important Preparation Instructions
- Dilution of ZOFRAN Injection in 50 mL of 5% Dextrose Injection or 0.9% Sodium Chloride Injection is required before administration to adult and pediatric patients for the prevention of nausea and vomiting associated with emetogenic chemotherapy.
For pediatric patients between 6 months and 1 year of age and/or 10 kg or less: Depending on the fluid needs of the patient, ZOFRAN Injection may be diluted in 10 to 50 mL of 5% Dextrose Injection or 0.9% Sodium Chloride Injection. - Occasionally, ondansetron precipitates at the stopper/vial interface in vials stored upright. Potency and safety are not affected. If a precipitate is observed, resolubilize by shaking the vial vigorously.
- Do not mix ZOFRAN Injection with solutions for which physical and chemical compatibility has not been established. In particular, this applies to alkaline solutions as a precipitate may form.
- Inspect the diluted ZOFRAN Injection solution for particulate matter and discoloration before administration; discard if present.
- Storage: After dilution, do not use beyond 24 hours. Although ZOFRAN Injection is chemically and physically stable when diluted as recommended, sterile precautions should be observed because diluents generally do not contain preservative.
- Compatibility: ZOFRAN Injection is compatible and stable at room temperature under normal lighting conditions for 48 hours after dilution with the following intravenous fluids: 0.9% Sodium Chloride Injection, 5% Dextrose Injection, 5% Dextrose and 0.9% Sodium Chloride Injection, 5% Dextrose and 0.45% Sodium Chloride Injection, and 3% Sodium Chloride Injection.
Dosage and Administration
The recommended dosage for adult and pediatric patients 6 months of age and older for prevention of nausea and vomiting associated with emetogenic chemotherapy is 0.15-mg/kg per dose for 3 doses (maximum of 16 mg per dose).
Caution: Dilution of ZOFRAN Injection is required in adult and pediatric patients prior to administration.
Infuse intravenously over 15 minutes beginning 30 minutes before the start of emetogenic chemotherapy and then repeat 4 and 8 hours after the first dose.
2.2 Prevention of Postoperative Nausea and/or Vomiting
Important Preparation Instructions
- Dilution of ZOFRAN Injection is not required before administration to adult and pediatric patients.
- Inspect ZOFRAN Injection visually for particulate matter and discoloration before administration; discard if present.
Dosage and Administration
The recommended dose and administration instructions for adult and pediatric patients 1 month of age and older for prevention of postoperative nausea and vomiting are shown in Table 1.
| a Few patients above 80 kg have been studied. b Administration of a second intravenous dose of 4 mg ondansetron postoperatively in adult patients who received a 4 mg prophylactic dose does not provide additional control of nausea and vomiting [see Clinical Studies (14.3)]. c For pediatric patients (1 month to 12 years) prevention of nausea and vomiting was only studied in patients who had not received prophylactic ondansetron. |
|||
|
Population |
Recommended Single Dose |
Administration Instructions |
Timing of Administration |
|
Adults and pediatric patients older than 12 years of age |
4 mga |
May be administered intravenously or intramuscularly:
|
Administer immediately before induction of anesthesia, or postoperatively if the patient did not receive prophylactic antiemetics and experiences nausea and/or vomiting occurring within 2 hours after surgeryb,c |
|
Pediatric patients 1 month to 12 years and more than 40 kg |
4 mg |
Infuse intravenously over at least 30 seconds and preferably longer (over 2 to 5 minutes). |
|
|
Pediatric patients 1 month to 12 years and 40 kg or less |
0.1 mg/kg |
Infuse intravenously over at least 30 seconds and preferably longer (over 2 to 5 minutes). |
|
2.3 Dosage Adjustment for Patients With Hepatic Impairment
In patients with severe hepatic impairment (Child-Pugh score of 10 or greater), a single maximal daily dose of 8 mg infused over 15 minutes beginning 30 minutes before the start of the emetogenic chemotherapy is recommended. There is no experience beyond first-day administration of ondansetron in these patients [see Use in Specific Populations (8.6)].
3 DOSAGE FORMS AND STRENGTHS
ZOFRAN Injection: 40 mg/20 mL clear, colorless, nonpyrogenic, sterile solution in a multiple-dose vial.
4 CONTRAINDICATIONS
ZOFRAN Injection is contraindicated for patients known to have hypersensitivity (e.g., anaphylaxis) to this product or any of its components. Anaphylactic reactions have been reported in patients taking ondansetron [see Adverse Reactions (6.2)].
The concomitant use of apomorphine with ondansetron is contraindicated based on reports of profound hypotension and loss of consciousness when apomorphine was administered with ondansetron.
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis and bronchospasm, have been reported in patients who have exhibited hypersensitivity to other selective 5-HT3 receptor antagonists.
5.2 QT Prolongation
Ondansetron prolongs the QT interval in a dose-dependent manner [see Clinical Pharmacology (12.2)]. In addition, postmarketing cases of Torsade de Pointes have been reported in patients using ondansetron. Avoid ZOFRAN in patients with congenital long QT syndrome. Electrocardiogram (ECG) monitoring is recommended in patients with electrolyte abnormalities (e.g., hypokalemia or hypomagnesemia), congestive heart failure, bradyarrhythmias, or patients taking other medicinal products that lead to QT prolongation.
5.3 Serotonin Syndrome
The development of serotonin syndrome has been reported with 5-HT3 receptor antagonists. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors, mirtazapine, fentanyl, lithium, tramadol, and intravenous methylene blue). Some of the reported cases were fatal. Serotonin syndrome occurring with overdose of ZOFRAN alone has also been reported. The majority of reports of serotonin syndrome related to 5-HT3 receptor antagonist use occurred in a post-anesthesia care unit or an infusion center.
Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, with or without gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome, especially with concomitant use of ZOFRAN and other serotonergic drugs. If symptoms of serotonin syndrome occur, discontinue ZOFRAN and initiate supportive treatment. Patients should be informed of the increased risk of serotonin syndrome, especially if ZOFRAN is used concomitantly with other serotonergic drugs [see Drug Interactions (7.5), Overdosage (10)].
5.4 Myocardial Ischemia
Myocardial ischemia has been reported in patients treated with ondansetron. In some cases, predominantly during intravenous administration, the symptoms appeared immediately after administration but resolved with prompt treatment. Coronary artery spasm appears to be the most common underlying cause. Therefore, do not exceed the recommended infusion rate of ZOFRAN and monitor patients for signs and symptoms of myocardial ischemia during and after administration [see Dosage and Administration (2.1, 2.2) and Adverse Reactions (6.2)].
5.5 Masking of Progressive Ileus and Gastric Distension
The use of ZOFRAN in patients following abdominal surgery or in patients with chemotherapy-induced nausea and vomiting may mask a progressive ileus and gastric distension. Monitor for decreased bowel activity, particularly in patients with risk factors for gastrointestinal obstruction.
ZOFRAN is not a drug that stimulates gastric or intestinal peristalsis. It should not be used instead of nasogastric suction.
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- QT Prolongation [see Warnings and Precautions (5.2)]
- Serotonin Syndrome [see Warnings and Precautions (5.3)]
- Myocardial Ischemia [see Warnings and Precautions (5.4)]
- Masking of Progressive Ileus and Gastric Distension [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The following adverse reactions have been reported in clinical trials of adult patients treated with ondansetron, the active ingredient of intravenous ZOFRAN across a range of dosages. A causal relationship to therapy with ZOFRAN (ondansetron) was unclear in many cases.
Chemotherapy-Induced Nausea and Vomiting
|
Number of Adult Patients With Reaction |
|||
|
Adverse Reaction |
ZOFRAN Injection 0.15 mg/kg x 3 (n = 419) |
Metoclopramide (n = 156) |
Placebo (n = 34) |
|
Diarrhea |
16% |
44% |
18% |
|
Headache |
17% |
7% |
15% |
|
Fever |
8% |
5% |
3% |
Cardiovascular: Rare cases of angina (chest pain), electrocardiographic alterations, hypotension, and tachycardia have been reported.
Gastrointestinal: Constipation has been reported in 11% of chemotherapy patients receiving multiday ondansetron.
Hepatic: In comparative trials in cisplatin chemotherapy patients with normal baseline values of aspartate transaminase (AST) and alanine transaminase (ALT), these enzymes have been reported to exceed twice the upper limit of normal in approximately 5% of patients. The increases were transient and did not appear to be related to dose or duration of therapy. On repeat exposure, similar transient elevations in transaminase values occurred in some courses, but symptomatic hepatic disease did not occur.
Integumentary: Rash has occurred in approximately 1% of patients receiving ondansetron.
Neurological: There have been rare reports consistent with, but not diagnostic of, extrapyramidal reactions in patients receiving ZOFRAN Injection, and rare cases of grand mal seizure.
Other: Rare cases of hypokalemia have been reported.
Postoperative Nausea and/or Vomiting
The adverse reactions in Table 3 have been reported in ≥ 2% of adults receiving ondansetron at a dosage of 4 mg intravenous over 2 to 5 minutes in clinical trials.
|
Adverse Reactiona,b |
ZOFRAN Injection 4 mg Intravenous (n = 547) |
Placebo (n = 547) |
|
Headache |
92 (17%) |
77 (14%) |
|
Drowsiness/Sedation |
44 (8%) |
37 (7%) |
|
Injection-site reaction |
21 (4%) |
18 (3%) |
|
Fever |
10 (2%) |
6 (1%) |
|
Cold sensation |
9 (2%) |
8 (1%) |
|
Pruritus |
9 (2%) |
3 (< 1%) |
|
Paresthesia |
9 (2%) |
2 (< 1%) |
a Adverse reactions: Rates of these reactions were not significantly different in the ondansetron and placebo groups.
b Patients were receiving multiple concomitant perioperative and postoperative medications.
Pediatric Use: Rates of adverse reactions were similar in both the ondansetron and placebo groups in pediatric patients receiving ondansetron (a single 0.1-mg/kg dose for pediatric patients weighing 40 kg or less, or 4 mg for pediatric patients weighing more than 40 kg) administered intravenously over at least 30 seconds. Diarrhea was seen more frequently in patients taking ZOFRAN (2%) compared with placebo (< 1%) in the 1-month to 24-month age-group. These patients were receiving multiple concomitant perioperative and postoperative medications.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ondansetron. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to ondansetron.
Cardiovascular
Arrhythmias (including ventricular and supraventricular tachycardia, premature ventricular contractions, and atrial fibrillation), bradycardia, electrocardiographic alterations (including second-degree heart block, QT/QTc interval prolongation, and ST segment depression), palpitations, and syncope. Rarely and predominantly with intravenous ondansetron, transient ECG changes, including QT/QTc interval prolongation have been reported [see Warnings and Precautions (5.2)].
Myocardial ischemia was reported predominately with intravenous administration [see Warnings and Precautions (5.4)].
General
Flushing: Rare cases of hypersensitivity reactions, sometimes severe (e.g., anaphylactic reactions, angioedema, bronchospasm, cardiopulmonary arrest, hypotension, laryngeal edema, laryngospasm, shock, shortness of breath, stridor) have also been reported. A positive lymphocyte transformation test to ondansetron has been reported, which suggests immunologic sensitivity to ondansetron.
Hepatobiliary
Liver enzyme abnormalities have been reported. Liver failure and death have been reported in patients with cancer receiving concurrent medications, including potentially hepatotoxic cytotoxic chemotherapy and antibiotics.
Local Reactions
Pain, redness, and burning at site of injection.
Lower Respiratory
Hiccups.
Neurological
Oculogyric crisis, appearing alone, as well as with other dystonic reactions. Transient dizziness during or shortly after intravenous infusion.
Skin
Urticaria, Stevens-Johnson syndrome, and toxic epidermal necrolysis.
Eye Disorders
Cases of transient blindness, predominantly during intravenous administration, have been reported. These cases of transient blindness were reported to resolve within a few minutes up to 48 hours. Transient blurred vision, in some cases associated with abnormalities of accommodation, has also been reported.
7 DRUG INTERACTIONS
7.1 Drugs Affecting Cytochrome P-450 Enzymes
Ondansetron does not appear to induce or inhibit the cytochrome P-450 drug-metabolizing enzyme system of the liver. Because ondansetron is metabolized by hepatic cytochrome P-450 drug-metabolizing enzymes (CYP3A4, CYP2D6, CYP1A2), inducers or inhibitors of these enzymes may change the clearance and, hence, the half-life of ondansetron [see Clinical Pharmacology (12.3)]. On the basis of limited available data, no dosage adjustment is recommended for patients on these drugs.
7.2 Apomorphine
Based on reports of profound hypotension and loss of consciousness when apomorphine was administered with ondansetron, the concomitant use of apomorphine with ondansetron is contraindicated [see Contraindications (4)].
7.3 Phenytoin, Carbamazepine, and Rifampin
In patients treated with potent inducers of CYP3A4 (i.e., phenytoin, carbamazepine, and rifampin), the clearance of ondansetron was significantly increased and ondansetron blood concentrations were decreased. However, on the basis of available data, no dosage adjustment for ondansetron is recommended for patients on these drugs [see Clinical Pharmacology (12.3)].
7.4 Tramadol
Although there are no data on pharmacokinetic drug interactions between ondansetron and tramadol, data from two small trials indicate that concomitant use of ondansetron may result in reduced analgesic activity of tramadol. Patients on concomitant ondansetron self-administered tramadol more frequently in these trials, leading to an increased cumulative dose in patient-controlled administration of tramadol.
7.5 Serotonergic Drugs
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT3 receptor antagonists and other serotonergic drugs, including SSRIs and SNRIs [see Warnings and Precautions (5.3)].
7.6 Chemotherapy
In humans, carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron.
In a crossover trial in 76 pediatric patients, intravenous ondansetron did not increase blood levels of high-dose methotrexate.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Published epidemiological studies on the association between ondansetron use and major birth defects have reported inconsistent findings and have important methodological limitations that preclude conclusions about the safety of ondansetron use in pregnancy (see Data). Available postmarketing data have not identified a drug-associated risk of miscarriage or adverse maternal outcomes. Reproductive studies in rats and rabbits did not show evidence of harm to the fetus when ondansetron was administered intravenously during organogenesis at approximately 3.6 and 2.9 times the maximum recommended human intravenous dose of 0.15 mg/kg given three times a day, based on body surface area (BSA), respectively (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, miscarriages, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriages in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Available data on ondansetron use in pregnant women from several published epidemiological studies preclude an assessment of a drug-associated risk of adverse fetal outcomes due to important methodological limitations, including the uncertainty of whether women who filled a prescription actually took the medication, the concomitant use of other medications or treatments, recall bias, and other unadjusted confounders.
Ondansetron exposure in utero has not been associated with overall major congenital malformations in aggregate analyses. One large retrospective cohort study examined 1970 women who received a prescription for ondansetron during pregnancy and reported no association between ondansetron exposure and major congenital malformations, miscarriage, stillbirth, preterm delivery, infants of low birth weight, or infants small for gestational age.
Two large retrospective cohort studies and one case-control study have assessed ondansetron exposure in the first trimester and risk of cardiovascular defects with inconsistent findings. Relative risks (RR) ranged from 0.97 (95% CI 0.86 to 1.10) to 1.62 (95% CI 1.04, 2.54). A subset analysis in one of the cohort studies observed that ondansetron was specifically associated with cardiac septal defects (RR 2.05, 95% CI 1.19, 3.28); however this association was not confirmed in other studies.
Several studies have assessed ondansetron and the risk of oral clefts with inconsistent findings. A retrospective cohort study of 1.8 million pregnancies in the US Medicaid Database showed an increased risk of oral clefts among 88,467 pregnancies in which oral ondansetron was prescribed in the first trimester (RR 1.24, 95% CI 1.03, 1.48), but no such association was reported with intravenous ondansetron in 23,866 pregnancies (RR 0.95, 95% CI 0.63, 1.43). In the subgroup of women who received both forms of administration, the RR was 1.07 (95% CI 0.59, 1.93). Two case-control studies, using data from birth defects surveillance programs, reported conflicting associations between maternal use of ondansetron and isolated cleft palate (OR 1.6 [95% CI 1.1, 2.3] and 0.5 [95% CI 0.3, 1.0]). It is unknown whether ondansetron exposure in utero in the cases of cleft palate occurred during the time of palate formation (the palate is formed between the 6th and 9th weeks of pregnancy).
Animal Data
In embryo-fetal development studies in rats and rabbits, pregnant animals received intravenous doses of ondansetron up to 10 mg/kg/day and 4 mg/kg/day, respectively, during the period of organogenesis. With the exception of short periods of maternal weight loss and a slight increase in the incidence of early uterine deaths at the high dose level in rabbits, there were no significant effects of ondansetron on the maternal animals or the development of the offspring. At doses of 10 mg/kg/day in rats and 4 mg/kg/day in rabbits, the maternal exposure margin was approximately 3.6 and 2.9 times the maximum recommended human oral dose of 0.15 mg/kg given three times a day, respectively, based on BSA.
No intravenous pre- and post-natal developmental toxicity study was performed with ondansetron. In an oral pre- and post-natal development study pregnant rats received oral doses of ondansetron up to 15 mg/kg/day from Day 17 of pregnancy to litter Day 21. With the exception of a slight reduction in maternal body weight gain, there were no effects upon the pregnant rats and the pre- and postnatal development of their offspring, including reproductive performance of the mated F1 generation.
8.2 Lactation
Risk Summary
It is not known whether ondansetron is present in human milk. There are no data on the effects of ZOFRAN on the breastfed infant or the effects on milk production. However, it has been demonstrated that ondansetron is present in the milk of rats. When a drug is present in animal milk, it is likely that the drug will be present in human milk.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ZOFRAN and any potential adverse effects on the breast-fed infant from ZOFRAN or from the underlying maternal condition.
8.4 Pediatric Use
Little information is available about the use of ondansetron in pediatric surgical patients younger than 1 month [see Clinical Studies (14.2)]. Little information is available about the use of ondansetron in pediatric cancer patients younger than 6 months [see Clinical Studies (14.1), Dosage and Administration (2)].
The clearance of ondansetron in pediatric patients aged 1 month to 4 months is slower and the half-life is ~2.5-fold longer than patients who are aged > 4 to 24 months. As a precaution, it is recommended that patients younger than 4 months receiving this drug be closely monitored [see Clinical Pharmacology (12.3)].
8.5 Geriatric Use
Of the total number of subjects enrolled in cancer chemotherapy-induced and postoperative nausea and vomiting US- and foreign-controlled clinical trials, 862 were aged 65 years and older. No overall differences in safety or effectiveness were observed between subjects 65 years and older and younger subjects. A reduction in clearance and increase in elimination half-life were seen in patients older than 75 years compared with younger subjects [see Clinical Pharmacology (12.3)]. There were an insufficient number of patients older than 75 years of age and older in the clinical trials to permit safety or efficacy conclusions in this age-group. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Dosage adjustment is not needed in patients over the age of 65.
8.6 Hepatic Impairment
In patients with severe hepatic impairment (Child-Pugh score of 10 or greater), clearance is reduced and apparent volume of distribution is increased with a resultant increase in plasma half-life [see Clinical Pharmacology (12.3)]. In such patients, a total daily dose of 8 mg should not be exceeded [see Dosage and Administration (2.3)].
9 DRUG ABUSE AND DEPENDENCE
Animal studies have shown that ondansetron is not discriminated as a benzodiazepine nor does it substitute for benzodiazepines in direct addiction studies.
10 OVERDOSAGE
There is no specific antidote for ondansetron overdose. Patients should be managed with appropriate supportive therapy. Individual intravenous doses as large as 150 mg and total daily intravenous doses as large as 252 mg have been inadvertently administered without significant adverse events. These doses are more than 10 times the recommended daily dose.
In addition to the adverse reactions listed above, the following events have been described in the setting of ondansetron overdose: “Sudden blindness” (amaurosis) of 2 to 3 minutes’ duration plus severe constipation occurred in one patient that was administered 72 mg of ondansetron intravenously as a single dose. Hypotension (and faintness) occurred in another patient that took 48 mg of ondansetron hydrochloride tablets. Following infusion of 32 mg over only a 4-minute period, a vasovagal episode with transient second-degree heart block was observed. In all instances, the events resolved completely.
Pediatric cases consistent with serotonin syndrome have been reported after inadvertent oral overdoses of ondansetron (exceeding estimated ingestion of 5 mg/kg) in young children. Reported symptoms included somnolence, agitation, tachycardia, tachypnea, hypertension, flushing, mydriasis, diaphoresis, myoclonic movements, horizontal nystagmus, hyperreflexia, and seizure. Patients required supportive care, including intubation in some cases, with complete recovery without sequelae within 1 to 2 days.
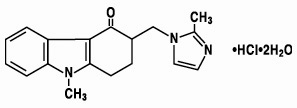
11 DESCRIPTION
The active ingredient of ZOFRAN Injection is ondansetron hydrochloride, a selective blocking agent of the serotonin 5-HT3 receptor type. Its chemical name is (±) 1, 2, 3, 9-tetrahydro-9-methyl-3-[(2-methyl-1H-imidazol-1-yl)methyl]-4H-carbazol-4-one, monohydrochloride, dihydrate. It has the following structural formula:
The empirical formula is C18H19N3O•HCl•2H2O, representing a molecular weight of 365.9 g/mol.
Ondansetron HCl is a white to off-white powder that is soluble in water and normal saline.
Each 1 mL of aqueous solution in the 20-mL multidose vial contains 2 mg of ondansetron as the hydrochloride dihydrate; 8.3 mg of sodium chloride, USP; 0.5 mg of citric acid monohydrate, USP and 0.25 mg of sodium citrate dihydrate, USP as buffers; and 1.2 mg of methylparaben, NF and 0.15 mg of propylparaben, NF as preservatives in Water for Injection, USP.
ZOFRAN Injection is a clear, colorless, nonpyrogenic, sterile solution for intravenous use. The pH of the injection solution is 3.3 to 4.0.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ondansetron is a selective 5-HT3 receptor antagonist. While ondansetron’s mechanism of action has not been fully characterized, it is not a dopamine-receptor antagonist.
12.2 Pharmacodynamics
In normal volunteers, single intravenous doses of 0.15 mg/kg of ondansetron had no effect on esophageal motility, gastric motility, lower esophageal sphincter pressure, or small intestinal transit time. In another trial in 6 normal male volunteers, a 16 mg dose infused over 5 minutes showed no effect of the drug on cardiac output, heart rate, stroke volume, blood pressure, or ECG. Multiday administration of ondansetron has been shown to slow colonic transit in normal volunteers. Ondansetron has no effect on plasma prolactin concentrations. In a gender balanced pharmacodynamic trial (n = 56), ondansetron 4 mg administered intravenously or intramuscularly was dynamically similar in the prevention of nausea and vomiting using the ipecacuanha model of emesis.
Cardiac Electrophysiology
QTc interval prolongation was studied in a double-blind, single intravenous dose, placebo- and positive-controlled, crossover trial in 58 healthy subjects. The maximum mean (95% upper confidence bound) difference in QTcF from placebo after baseline correction was 19.5 (21.8) ms and 5.6 (7.4) ms after 15-minute intravenous infusions of 32 mg and 8 mg ZOFRAN, respectively. A significant exposure-response relationship was identified between ondansetron concentration and ΔΔQTcF. Using the established exposure-response relationship, 24 mg infused intravenously over 15 minutes had a mean predicted (95% upper prediction interval) ΔΔQTcF of 14.0 (16.3) ms. In contrast, 16 mg infused intravenously over 15 minutes using the same model had a mean predicted (95% upper prediction interval) ΔΔQTcF of 9.1 (11.2) ms. In this study, the 8-mg dose infused over 15 minutes did not prolong the QT interval to any clinically relevant extent.
12.3 Pharmacokinetics
In normal adult volunteers, the following mean pharmacokinetic data have been determined following a single 0.15-mg/kg intravenous dose.
|
Age-group (years) |
n |
Peak Plasma Concentration (ng/mL) |
Mean Elimination Half-life (h) |
Plasma Clearance (L/h/kg) |
|
19-40 |
11 |
102 |
3.5 |
0.381 |
|
61-74 |
12 |
106 |
4.7 |
0.319 |
|
≥ 75 |
11 |
170 |
5.5 |
0.262 |
Absorption
A trial was performed in normal volunteers (n = 56) to evaluate the pharmacokinetics of a single 4-mg dose administered as a 5-minute infusion compared with a single intramuscular injection. Systemic exposure as measured by mean area under curve (AUC) were equivalent, with values of 156 [95% CI 136, 180] and 161 [95% CI 137, 190] ng•h/mL for intravenous and intramuscular groups, respectively. Mean peak plasma concentrations were 42.9 [95% CI 33.8, 54.4] ng/mL at 10 minutes after intravenous infusion and 31.9 [95% CI 26.3, 38.6] ng/mL at 41 minutes after intramuscular injection.
Distribution
Plasma protein binding of ondansetron as measured in vitro was 70% to 76%, over the pharmacologic concentration range of 10 to 500 ng/mL. Circulating drug also distributes into erythrocytes.
Elimination
Metabolism: Ondansetron is extensively metabolized in humans, with approximately 5% of a radiolabeled dose recovered as the parent compound from the urine. The primary metabolic pathway is hydroxylation on the indole ring followed by subsequent glucuronide or sulfate conjugation.
Although some nonconjugated metabolites have pharmacologic activity, these are not found in plasma at concentrations likely to significantly contribute to the biological activity of ondansetron. The metabolites are observed in the urine.
In vitro metabolism studies have shown that ondansetron is a substrate for multiple human hepatic cytochrome P-450 enzymes, including CYP1A2, CYP2D6, and CYP3A4. In terms of overall ondansetron turnover, CYP3A4 plays a predominant role while formation of the major in vivo metabolites is apparently mediated by CYP1A2. The role of CYP2D6 in ondansetron in vivo metabolism is relatively minor.
The pharmacokinetics of intravenous ondansetron did not differ between subjects who were poor metabolizers of CYP2D6 and those who were extensive metabolizers of CYP2D6, further supporting the limited role of CYP2D6 in ondansetron disposition in vivo.
Excretion: In adult cancer patients, the mean ondansetron elimination half-life was 4.0 hours, and there was no difference in the multidose pharmacokinetics over a 4-day period. In a dose-proportionality trial, systemic exposure to 32 mg of ondansetron was not proportional to dose as measured by comparing dose-normalized AUC values with an 8-mg dose. This is consistent with a small decrease in systemic clearance with increasing plasma concentrations.
Specific Populations
Geriatric Patients: A reduction in clearance and increase in elimination half-life are seen in patients older than 75 years of age [see Use in Specific Populations (8.5)].
Pediatric Patients: Pharmacokinetic samples were collected from 74 cancer patients aged 6 to 48 months, who received a dose of 0.15 mg/kg of intravenous ondansetron every 4 hours for 3 doses during a safety and efficacy trial. These data were combined with sequential pharmacokinetics data from 41 surgery patients aged 1 month to 24 months, who received a single dose of 0.1 mg/kg of intravenous ondansetron prior to surgery with general anesthesia, and a population pharmacokinetic analysis was performed on the combined data set. The results of this analysis are included in Table 5 and are compared with the pharmacokinetic results in cancer patients aged 4 to 18 years.
|
Subjects and Age-group |
N |
CL (L/h/kg) |
Vdss (L/kg) |
t½ (h) |
|
Geometric Mean |
Mean |
|||
|
Pediatric Cancer Patients 4 to 18 years |
N = 21 |
0.599 |
1.9 |
2.8 |
|
Population PK Patientsa 1 month to 48 months |
N = 115 |
0.582 |
3.65 |
4.9 |
a Population PK (Pharmacokinetic) Patients: 64% cancer patients and 36% surgery patients.
Based on the population pharmacokinetic analysis, cancer patients aged 6 to 48 months who receive a dose of 0.15 mg/kg of intravenous ondansetron every 4 hours for 3 doses would be expected to achieve a systemic exposure (AUC) consistent with the exposure achieved in previous pediatric trials in cancer patients (4 to 18 years) at similar doses.
In a trial of 21 pediatric patients (3 to 12 years) who were undergoing surgery requiring anesthesia for a duration of 45 minutes to 2 hours, a single intravenous dose of ondansetron, 2 mg (3 to 7 years) or 4 mg (8 to 12 years), was administered immediately prior to anesthesia induction. Mean weight-normalized clearance and volume of distribution values in these pediatric surgical patients were similar to those previously reported for young adults. Mean terminal half-life was slightly reduced in pediatric patients (range, 2.5 to 3 hours) in comparison with adults (range, 3 to 3.5 hours).
In a trial of 51 pediatric patients (aged 1 month to 24 months) who were undergoing surgery requiring general anesthesia, a single intravenous dose of ondansetron, 0.1 or 0.2 mg/kg, was administered prior to surgery. As shown in Table 6, the 41 patients with pharmacokinetic data were divided into 2 groups, patients aged 1 month to 4 months and patients aged 5 to 24 months, and are compared with pediatric patients aged 3 to 12 years.
|
Subjects and Age-group |
N |
CL (L/h/kg) |
Vdss (L/kg) |
t½ (h) |
|
Geometric Mean |
Mean |
|||
|
Pediatric Surgery Patients 3 to 12 years |
N = 21 |
0.439 |
1.65 |
2.9 |
|
Pediatric Surgery Patients 5 to 24 months |
N = 22 |
0.581 |
2.3 |
2.9 |
|
Pediatric Surgery Patients 1 month to 4 months |
N = 19 |
0.401 |
3.5 |
6.7 |
In general, surgical and cancer pediatric patients younger than 18 years tend to have a higher ondansetron clearance compared with adults leading to a shorter half-life in most pediatric patients. In patients aged 1 month to 4 months, a longer half-life was observed due to the higher volume of distribution in this age-group.
In a trial of 21 pediatric cancer patients (aged 4 to 18 years) who received three intravenous doses of 0.15 mg/kg of ondansetron at 4-hour intervals, patients older than 15 years exhibited ondansetron pharmacokinetic parameters similar to those of adults.
Patients with Renal Impairment: Due to the very small contribution (5%) of renal clearance to the overall clearance, renal impairment was not expected to significantly influence the total clearance of ondansetron. However, ondansetron mean plasma clearance was reduced by about 41% in patients with severe renal impairment (creatinine clearance < 30 mL/min). This reduction in clearance is variable and was not consistent with an increase in half-life [see Use in Specific Populations (8.7)].
Patients with Hepatic Impairment: In patients with mild-to-moderate hepatic impairment, clearance is reduced 2-fold and mean half-life is increased to 11.6 hours compared with 5.7 hours in those without hepatic impairment. In patients with severe hepatic impairment (Child-Pugh score of 10 or greater), clearance is reduced 2-fold to 3-fold and apparent volume of distribution is increased with a resultant increase in half-life to 20 hours [see Dosage and Administration (2.3), Use in Specific Populations (8.6)].
Drug Interaction Studies
CYP 3A4 Inducers: Ondansetron elimination may be affected by cytochrome P-450 inducers. In a pharmacokinetic trial of 16 epileptic patients maintained chronically on CYP3A4 inducers, carbamazepine, or phenytoin, a reduction in AUC, Cmax, and t½ of ondansetron was observed. This resulted in a significant increase in the clearance of ondansetron. In a pharmacokinetic study of 10 healthy subjects receiving a single-dose intravenous dose of ondansetron 8 mg after 600 mg rifampin once daily for five days, the AUC and the t½ of ondansetron were reduced by 48% and 46%, respectively. These changes in ondansetron exposure with CYP3A4 inducers are not thought to be clinically relevant [see Drug Interactions (7.3)].
Chemotherapeutic Agents: Carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron [see Drug Interactions (7.6)].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenic effects were not seen in 2-year studies in rats and mice with oral ondansetron doses up to 10 and 30 mg/kg per day, respectively (approximately 3.6 and 5.4 times the recommended human intravenous dose of 0.15 mg/kg given three times a day, based on BSA). Ondansetron was not mutagenic in standard tests for mutagenicity.
Oral administration of ondansetron up to 15 mg/kg per day (approximately 3.8 times the recommended human intravenous dose, based on BSA) did not affect fertility or general reproductive performance of male and female rats.
14 CLINICAL STUDIES
The clinical efficacy of ondansetron hydrochloride, the active ingredient of ZOFRAN, was assessed in clinical trials as described below.
14.1 Chemotherapy-Induced Nausea and Vomiting
Adults
In a double-blind trial of three different dosing regimens of ZOFRAN Injection, 0.015 mg/kg, 0.15 mg/kg, and 0.30 mg/kg, each given three times during the course of cancer chemotherapy, the 0.15-mg/kg dosing regimen was more effective than the 0.015-mg/kg dosing regimen. The 0.30-mg/kg dosing regimen was not shown to be more effective than the 0.15-mg/kg dosing regimen.
Cisplatin-Based Chemotherapy: In a double-blind trial in 28 patients, ZOFRAN Injection (three 0.15-mg/kg doses) was significantly more effective than placebo in preventing nausea and vomiting induced by cisplatin-based chemotherapy. Therapeutic response was as shown in Table 7.
|
ZOFRAN Injection
|
Placebo |
P-valueb |
|
|
Number of patients |
14 |
14 | |
|
Treatment response | |||
|
2 (14%) |
0 (0%) | |
|
8 (57%) |
0 (0%) | |
|
2 (14%) |
1 (7%) | |
|
2 (14%) |
13 (93%) |
0.001 |
|
Median number of emetic episodes |
1.5 |
Undefinedc | |
|
Median time to first emetic episode (h) |
11.6 |
2.8 |
0.001 |
|
Median nausea scores (0-100)d |
3 |
59 |
0.034 |
|
Global satisfaction with control of nausea and vomiting (0-100)e |
96 |
10.5 |
0.009 |
a Chemotherapy was high dose (100 and 120 mg/m2; ZOFRAN Injection n = 6, placebo n = 5) or moderate dose (50 and 80 mg/m2; ZOFRAN Injection n = 8, placebo n = 9). Other chemotherapeutic agents included fluorouracil, doxorubicin, and cyclophosphamide. There was no difference between treatments in the types of chemotherapy that would account for differences in response.
b Efficacy based on "all-patients-treated" analysis.
c Median undefined since at least 50% of the patients were rescued or had more than five emetic episodes.
d Visual analog scale assessment of nausea: 0 = no nausea, 100 = nausea as bad as it can be.
e Visual analog scale assessment of satisfaction: 0 = not at all satisfied, 100 = totally satisfied.
Ondansetron injection (0.15-mg/kg x 3 doses) was compared with metoclopramide (2 mg/kg x 6 doses) in a single-blind trial in 307 patients receiving cisplatin ≥ 100 mg/m2 with or without other chemotherapeutic agents. Patients received the first dose of ondansetron or metoclopramide 30 minutes before cisplatin. Two additional ondansetron doses were administered 4 and 8 hours later, or five additional metoclopramide doses were administered 2, 4, 7, 10, and 13 hours later. Cisplatin was administered over a period of 3 hours or less. Episodes of vomiting and retching were tabulated over the period of 24 hours after cisplatin. The results of this trial are summarized in Table 8.
|
ZOFRAN Injection |
Metoclopramide | ||
|
0.15 mg/kg x 3 |
2 mg/kg x 6 |
P-value |
|
|
Number of patients in efficacy population |
136 |
138 | |
|
Treatment response | |||
|
54 (40%) |
41 (30%) | |
|
34 (25%) |
30 (22%) | |
|
19 (14%) |
18 (13%) | |
|
29 (21%) |
49 (36%) | |
|
Comparison of treatments with respect to | |||
|
54/136 |
41/138 |
0.083 |
|
29/136 |
49/138 |
0.009 |
|
Median number of emetic episodes |
1 |
2 |
0.005 |
|
Median time to first emetic episode (h) |
20.5 |
4.3 |
< 0.001 |
|
Global satisfaction with control of nausea and vomiting (0-100)b |
85 |
63 |
0.001 |
|
Acute dystonic reactions |
0 |
8 |
0.005 |
|
Akathisia |
0 |
10 |
0.002 |
a In addition to cisplatin, 68% of patients received other chemotherapeutic agents, including cyclophosphamide, etoposide, and fluorouracil. There was no difference between treatments in the types of chemotherapy that would account for differences in response.
b Visual analog scale assessment: 0 = not at all satisfied, 100 = totally satisfied.
Cyclophosphamide-Based Chemotherapy: In a double-blind, placebo-controlled trial of ZOFRAN Injection (three 0.15-mg/kg doses) in 20 patients receiving cyclophosphamide (500 to 600 mg/m2) chemotherapy, ZOFRAN Injection was significantly more effective than placebo in preventing nausea and vomiting. The results are summarized in Table 9.
|
ZOFRAN Injection (0.15 mg/kg x 3) |
Placebo |
P-valueb |
|
|
Number of patients |
10 |
10 | |
|
Treatment response | |||
|
7 (70%) |
0 (0%) |
0.001 |
|
0 (0%) |
2 (20%) | |
|
2 (20%) |
4 (40%) | |
|
1 (10%) |
4 (40%) |
0.131 |
|
Median number of emetic episodes |
0 |
4 |
0.008 |
|
Median time to first emetic episode (h) |
Undefinedc |
8.79 | |
|
Median nausea scores (0-100)d |
0 |
60 |
0.001 |
|
Global satisfaction with control of nausea and vomiting (0-100)e |
100 |
52 |
0.008 |
a Chemotherapy consisted of cyclophosphamide in all patients, plus other agents, including fluorouracil, doxorubicin, methotrexate, and vincristine. There was no difference between treatments in the type of chemotherapy that would account for differences in response.
b Efficacy based on "all-patients-treated" analysis.
c Median undefined since at least 50% of patients did not have any emetic episodes.
d Visual analog scale assessment of nausea: 0 = no nausea, 100 = nausea as bad as it can be.
e Visual analog scale assessment of satisfaction: 0 = not at all satisfied, 100 = totally satisfied.
Re-treatment: In uncontrolled trials, 127 patients receiving cisplatin (median dose, 100 mg/m2) and ondansetron who had two or fewer emetic episodes were re-treated with ondansetron and chemotherapy, mainly cisplatin, for a total of 269 re-treatment courses (median: 2; range, 1 to 10). No emetic episodes occurred in 160 (59%), and two or fewer emetic episodes occurred in 217 (81%) re-treatment courses.
Pediatrics
Four open-label, noncomparative (one US, three foreign) trials have been performed with 209 pediatric cancer patients aged 4 to 18 years given a variety of cisplatin or noncisplatin regimens. In the three foreign trials, the initial dose of ZOFRAN Injection ranged from 0.04 to 0.87 mg/kg for a total dose of 2.16 to 12 mg. This was followed by the oral administration of ondansetron ranging from 4 to 24 mg daily for 3 days. In the US trial, ZOFRAN was administered intravenously (only) in three doses of 0.15 mg/kg each for a total daily dose of 7.2 to 39 mg. In these trials, 58% of the 196 evaluable patients had a complete response (no emetic episodes) on Day 1. Thus, prevention of vomiting in these pediatric patients was essentially the same as for patients older than 18 years.
An open-label, multicenter, noncomparative trial has been performed in 75 pediatric cancer patients aged 6 to 48 months receiving at least one moderately or highly emetogenic chemotherapeutic agent. Fifty-seven percent (57%) were females; 67% were white, 18% were American Hispanic, and 15% were black patients. ZOFRAN was administered intravenously over 15 minutes in three doses of 0.15 mg/kg. The first dose was administered 30 minutes before the start of chemotherapy; the second and third doses were administered 4 and 8 hours after the first dose, respectively. Eighteen patients (25%) received routine prophylactic dexamethasone (i.e., not given as rescue). Of the 75 evaluable patients, 56% had a complete response (no emetic episodes) on Day 1. Thus, prevention of vomiting in these pediatric patients was comparable to the prevention of vomiting in patients aged 4 years and older.
14.2 Prevention of Postoperative Nausea and/or Vomiting
Adults
Adult surgical patients who received ondansetron immediately before the induction of general balanced anesthesia (barbiturate: thiopental, methohexital, or thiamylal; opioid: alfentanil or fentanyl; nitrous oxide; neuromuscular blockade: succinylcholine/curare and/or vecuronium or atracurium; and supplemental isoflurane) were evaluated in two double-blind US trials involving 554 patients. ZOFRAN Injection (4 mg) intravenous given over 2 to 5 minutes was significantly more effective than placebo. The results of these trials are summarized in Table 10.
|
Ondansetron 4 mg Intravenous |
Placebo |
P-value |
|
|
Study 1 | |||
|
Emetic episodes: | |||
|
Number of patients |
136 |
139 | |
|
Treatment response over 24-h postoperative period | |||
|
103 (76%) |
64 (46%) |
< 0.001 |
|
13 (10%) |
17 (12%) | |
|
20 (15%) |
58 (42%) | |
|
Nausea assessments: | |||
|
Number of patients |
134 |
136 | |
|
No nausea over 24-h postoperative period |
56 (42%) |
39 (29%) | |
|
Study 2 | |||
|
Emetic episodes: | |||
|
Number of patients |
136 |
143 | |
|
Treatment response over 24-h postoperative period | |||
|
85 (63%) |
63 (44%) |
0.002 |
|
16 (12%) |
29 (20%) | |
|
35 (26%) |
51 (36%) | |
|
Nausea assessments: | |||
|
Number of patients |
125 |
133 | |
|
No nausea over 24-h postoperative period |
48 (38%) |
42 (32%) |
The populations in Table 10 consisted mainly of females undergoing laparoscopic procedures.
In a placebo-controlled trial conducted in 468 males undergoing outpatient procedures, a single 4-mg intravenous ondansetron dose prevented postoperative vomiting over a 24-hour period in 79% of males receiving drug compared with 63% of males receiving placebo (P < 0.001).
Two other placebo-controlled trials were conducted in 2,792 patients undergoing major abdominal or gynecological surgeries to evaluate a single 4-mg or 8-mg intravenous ondansetron dose for prevention of postoperative nausea and vomiting over a 24-hour period. At the 4-mg dosage, 59% of patients receiving ondansetron versus 45% receiving placebo in the first trial (P < 0.001) and 41% of patients receiving ondansetron versus 30% receiving placebo in the second trial (P = 0.001) experienced no emetic episodes. No additional benefit was observed in patients who received intravenous ondansetron 8 mg compared with patients who received intravenous ondansetron 4 mg.
Pediatrics
Three double-blind, placebo-controlled trials have been performed (one US, two foreign) in 1049 male and female patients (aged 2 to 12 years) undergoing general anesthesia with nitrous oxide. The surgical procedures included tonsillectomy with or without adenoidectomy, strabismus surgery, herniorrhaphy, and orchidopexy. Patients were randomized to either single intravenous doses of ondansetron (0.1 mg/kg for pediatric patients weighing 40 kg or less, 4 mg for pediatric patients weighing more than 40 kg) or placebo. Study drug was administered over at least 30 seconds, immediately prior to or following anesthesia induction. Ondansetron was significantly more effective than placebo in preventing nausea and vomiting. The results of these trials are summarized in Table 11.
|
Treatment Response Over 24 Hours |
Ondansetron n (%) |
Placebo n (%) |
P-value |
|
Study 1 | |||
|
Number of patients |
205 |
210 | |
|
0 Emetic episodes |
140 (68%) |
82 (39%) |
≤0.001 |
|
Failurea |
65 (32%) |
128 (61%) | |
|
Study 2 | |||
|
Number of patients |
112 |
110 | |
|
0 Emetic episodes |
68 (61%) |
38 (35%) |
≤0.001 |
|
Failurea |
44 (39%) |
72 (65%) | |
|
Study 3 | |||
|
Number of patients |
206 |
206 | |
|
0 Emetic episodes |
123 (60%) |
96 (47%) |
≤0.01 |
|
Failurea |
83 (40%) |
110 (53%) | |
|
Nausea assessmentsb | |||
|
Number of patients |
185 |
191 | |
|
None |
119 (64%) |
99 (52%) |
≤0.01 |
a Failure was one or more emetic episodes, rescued, or withdrawn.
b Nausea measured as none, mild, or severe.
A double-blind, multicenter, placebo-controlled trial was conducted in 670 pediatric patients aged 1 month to 24 months who were undergoing routine surgery under general anesthesia. Seventy-five percent (75%) were males; 64% were white, 15% were black, 13% were American Hispanic, 2% were Asian, and 6% were “other race” patients. A single 0.1-mg/kg intravenous dose of ondansetron administered within 5 minutes following induction of anesthesia was statistically significantly more effective than placebo in preventing vomiting. In the placebo group, 28% of patients experienced vomiting compared with 11% of subjects who received ondansetron (P ≤ 0.01). Overall, 32 (10%) of placebo patients and 18 (5%) of patients who received ondansetron received antiemetic rescue medication(s) or prematurely withdrew from the trial.
14.3 Prevention of Further Postoperative Nausea and/or Vomiting
Adults
Adult surgical patients receiving general balanced anesthesia (barbiturate: thiopental, methohexital, or thiamylal; opioid: alfentanil or fentanyl; nitrous oxide; neuromuscular blockade: succinylcholine/curare and/or vecuronium or atracurium; and supplemental isoflurane) who received no prophylactic antiemetics and who experienced nausea and/or vomiting within 2 hours postoperatively were evaluated in two double-blind US trials involving 441 patients. Patients who experienced an episode of postoperative nausea and/or vomiting were given ZOFRAN Injection (4 mg) intravenously over 2 to 5 minutes, and this was significantly more effective than placebo. The results of these trials are summarized in Table 12.
|
Ondansetron 4 mg Intravenous |
Placebo |
P-value |
|
|
Study 1 | |||
|
Emetic episodes: | |||
|
Number of patients |
104 |
117 | |
|
Treatment response 24 h after study drug | |||
|
49 (47%) |
19 (16%) |
<0.001 |
|
12 (12%) |
9 (8%) | |
|
43 (41%) |
89 (76%) | |
|
Median time to first emetic episode (min)a |
55.0 |
43.0 | |
|
Nausea assessments: | |||
|
Number of patients |
98 |
102 | |
|
Mean nausea score over 24-h postoperative periodb |
1.7 |
3.1 | |
|
Study 2 | |||
|
Emetic episodes: | |||
|
Number of patients |
112 |
108 | |
|
Treatment response 24 h after study drug | |||
|
49 (44%) |
28 (26%) |
0.006 |
|
14 (13%) |
3 (3%) | |
|
49 (44%) |
77 (71%) | |
|
Median time to first emetic episode (min)a |
60.5 |
34.0 | |
|
Nausea assessments: | |||
|
Number of patients |
105 |
85 | |
|
Mean nausea score over 24-h postoperative periodb |
1.9 |
2.9 |
a After administration of study drug.
b Nausea measured on a scale of 0-10 with 0 = no nausea, 10 = nausea as bad as it can be.
The populations in Table 12 consisted mainly of women undergoing laparoscopic procedures.
Repeat Dosing in Adults:
In patients who do not achieve adequate control of postoperative nausea and vomiting following a single, prophylactic, preinduction, intravenous dose of ondansetron 4 mg, administration of a second intravenous dose of ondansetron 4 mg postoperatively does not provide additional control of nausea and vomiting.
Pediatrics
One double-blind, placebo-controlled, US trial was performed in 351 male and female outpatients (aged 2 to 12 years) who received general anesthesia with nitrous oxide and no prophylactic antiemetics. Surgical procedures were unrestricted. Patients who experienced two or more emetic episodes within 2 hours following discontinuation of nitrous oxide were randomized to either single intravenous doses of ondansetron (0.1 mg/kg for pediatric patients weighing 40 kg or less, 4 mg for pediatric patients weighing more than 40 kg) or placebo administered over at least 30 seconds. Ondansetron was significantly more effective than placebo in preventing further episodes of nausea and vomiting. The results of the trial are summarized in Table 13.
|
Treatment Response Over 24 Hours |
Ondansetron n (%) |
Placebo n (%) |
P-value |
|
Number of patients |
180 |
171 | |
|
0 Emetic episodes |
96 (53%) |
29 (17%) |
≤0.001 |
|
Failurea |
84 (47%) |
142 (83%) |
a Failure was one or more emetic episodes, rescued, or withdrawn.
16 HOW SUPPLIED/STORAGE AND HANDLING
ZOFRAN Injection, 2 mg/mL, is supplied as follows:
NDC 0173-0442-00 20-mL multiple-dose vial (Singles)
Storage: Store vials between 2°C and 30°C (36°F and 86°F). Protect from light.
17 PATIENT COUNSELING INFORMATION
Hypersensitivity Reactions
Inform patients that ZOFRAN may cause hypersensitivity reactions, some as severe as anaphylaxis and bronchospasm. The patient should report any signs and symptoms of hypersensitivity reactions, including fever, chills, rash, or breathing problems [see Warnings and Precautions (5.1)].
QT Prolongation
Patients should be informed that ZOFRAN may cause serious cardiac arrhythmias, such as QT prolongation. Patients should be instructed to tell their healthcare provider right away if they perceive a change in their heart rate, if they feel lightheaded, or if they have a syncopal episode.
Patients should be informed that the chances of developing severe cardiac arrhythmias, such as QT prolongation and Torsade de Pointes are higher in the following people:
- Patients with a personal or family history of abnormal heart rhythms, such as congenital long QT syndrome;
- Patients who take medications, such as diuretics, which may cause electrolyte abnormalities;
- Patients with hypokalemia or hypomagnesemia.
ZOFRAN should be avoided in these patients, since they may be more at risk for cardiac arrhythmias, such as QT prolongation and Torsade de Pointes [see Warnings and Precautions (5.2)].
Drug Interactions
- Instruct the patient to report the use of all medications, especially apomorphine, to their healthcare provider. Concomitant use of apomorphine and ZOFRAN may cause a significant drop in blood pressure and loss of consciousness.
- Advise patients of the possibility of serotonin syndrome with concomitant use of ZOFRAN and another serotonergic agent, such as medications to treat depression and migraines. Advise patients to seek immediate medical attention if the following symptoms occur: changes in mental status, autonomic instability, neuromuscular symptoms with or without gastrointestinal symptoms [see Warnings and Precautions (5.3)].
Myocardial Ischemia
Inform patients that ZOFRAN may cause myocardial ischemia during or after the administration. Advise patients to seek immediate medical help if any symptoms suggestive of a myocardial ischemia occur, such as sudden chest pain or chest tightness [see Warnings and Precautions (5.4)].
Masking of Progressive Ileus and Gastric Distension
Inform patients following abdominal surgery or those with chemotherapy-induced nausea and vomiting that ZOFRAN may mask signs and symptoms of bowel obstruction. Instruct patients to immediately report any signs or symptoms consistent with a potential bowel obstruction to their healthcare provider [see Warnings and Precautions (5.5)].
Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
T2021-142
| ZOFRAN
ondansetron hydrochloride injection |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - GlaxoSmithKline LLC (167380711) |