HYDROCODONE BITARTRATE AND PSEUDOEPHEDRINE HYDROCHLORIDE SOLUTION- hydrocodone bitartrate and pseudoephedrine hydrochloride solution
Tris Pharma Inc
----------
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Hydrocodone Bitartrate and Pseudoephedrine Hydrochloride Oral Solution safely and effectively. See full prescribing information for Hydrocodone Bitartrate and Pseudoephedrine Hydrochloride Oral Solution.
Hydrocodone Bitartrate and Pseudoephedrine Hydrochloride Oral Solution, for oral use Initial U.S. Approval: 2011 INDICATIONS AND USAGEHydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is a combination product containing an antitussive and nasal decongestant indicated for: (1) DOSAGE AND ADMINISTRATIONFor oral use only.
DOSAGE FORMS AND STRENGTHSEach 5 mL of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution contains: hydrocodone bitartrate, USP, 5 mg; and pseudoephedrine hydrochloride, USP, 60 mg. (3) CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution include: Sedation, drowsiness, mental clouding, lethargy, impairment of mental and physical performance, anxiety, fear, dysphoria, dizziness, psychic dependence, mood changes, nervousness, sleeplessness, tremor, or arrhythmia. (6) To report SUSPECTED ADVERSE REACTIONS, contact Tris Pharma, Inc. at 1-732-940-0358 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION. Revised: 10/2014 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Common Cold
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is indicated for:
Relief of cough and nasal congestion associated with common cold.
Important Limitations of Use:
Not indicated for pediatric patients under 18 years of age [see Pediatric Use (8.4)].
2 DOSAGE AND ADMINISTRATION
Administer hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution by the oral route only. Measure hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution with an accurate milliliter measuring device. Do not use a household teaspoon to measure the dose [see Warnings and Precautions (5.9)]
3 DOSAGE FORMS AND STRENGTHS
Hydrocodone Bitartrate and Pseudoephedrine Hydrochloride is a clear, colorless to light yellow, grape-flavored liquid.
Each 5 mL of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution contains: hydrocodone bitartrate, USP, 5 mg; and pseudoephedrine hydrochloride, USP, 60 mg [see Description (11)].
4 CONTRAINDICATIONS
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is contraindicated in:
-
Patients with known hypersensitivity to hydrocodone bitartrate, pseudoephedrine hydrochloride, or any of the inactive ingredients of Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution.
-
Patients receiving MAOI therapy or within 14 days of stopping such therapy [see Drug Interactions (7.2)].
-
Patients with narrow angle glaucoma, urinary retention, severe hypertension or severe coronary artery disease.
5 WARNINGS AND PRECAUTIONS
5.1 Respiratory Depression
Hydrocodone bitartrate, one of the active ingredients in hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution, produces dose-related respiratory depression by directly acting on brain stem respiratory centers. Overdose of hydrocodone bitartrate in adults has been associated with fatal respiratory depression, and the use of hydrocodone bitartrate in children less than 6 years of age has been associated with fatal respiratory depression. Exercise caution when administering hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution because of the potential for respiratory depression. If respiratory depression occurs, it may be antagonized by the use of naloxone hydrochloride and other supportive measures when indicated [see Overdosage (10)].
5.2 Drug Dependence
Hydrocodone can produce drug dependence of the morphine type and therefore, has the potential for being abused. Psychic dependence, physical dependence, and tolerance may develop upon repeated administration of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution. Prescribe and administer Hydrocodone Bitartrate and Pseudoephedrine Hydrochloride with the same degree of caution appropriate to the use of other opioid drugs [see Drug Abuse and Dependence (9.2 ,9.3)].
5.3 Head Injury and Increased Intracranial Pressure
The respiratory depression effects of opioids and their capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions, or a pre-existing increase in intracranial pressure. Furthermore, opioids produce adverse reactions which may obscure the clinical course of patients with head injuries. The use of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be avoided in these patients.
5.4 Activities Requiring Mental Alertness
Hydrocodone bitartrate, one of the active ingredients in hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution, may produce marked drowsiness and impair the mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery. Advise patients to avoid engaging in hazardous tasks requiring mental alertness and motor coordination after ingestion of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution. Concurrent use of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution with alcohol or other central nervous system depressants should be avoided because additional impairment of central nervous system performance may occur.
5.5 Acute Abdominal Conditions
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be used with caution in patients with acute abdominal conditions since the administration of hydrocodone may obscure the diagnosis or clinical course of patients with acute abdominal conditions. The concurrent use of other anticholinergics with hydrocodone may produce paralytic ileus [see Drug Interactions (7.3)].
5.6 Co-administration with Anticholinergics
The concurrent use of anticholinergics with hydrocodone may produce paralytic ileus. Exercise caution when using hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution in patients taking anticholinergic medications [see Drug Interactions (7.3)].
5.7 Co-administration with MAOIs or Tricyclic Antidepressants
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should not be used in patients receiving MAOI therapy or within 14 days of stopping such therapy as an increase in blood pressure or hypertensive crisis, may occur. In addition, the use of MAOIs or tricyclic antidepressants with hydrocodone bitartrate, one of the active ingredients in hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution, may increase the effect of either the antidepressant or hydrocodone [see Contraindications (4) and Drug Interactions (7.2)].
5.8 Cardiovascular and Central Nervous System Effects
The pseudoephedrine hydrochloride contained in hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution can produce cardiovascular and central nervous system effects in some patients such as insomnia, dizziness, weakness, tremor, or arrhythmias. In addition, central nervous system stimulation with convulsions or cardiovascular collapse with accompanying hypotension has been reported. Therefore, hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be used with caution in patients with cardiovascular disorders, and should not be used in patients with severe hypertension or coronary artery disease.
5.9 Dosing
Patients should be advised to measure hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution with an accurate milliliter measuring device. Patients should be informed that a household teaspoon is not an accurate measuring device and could lead to overdosage, which can result in serious adverse reactions [see Overdosage (10)]. Patients should be advised to ask their pharmacist to recommend an appropriate measuring device and for instructions for measuring the correct dose.
5.10 Coexisting Conditions
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be used with caution in patients with diabetes, thyroid disease, Addison's disease, prostatic hypertrophy or urethral stricture, and asthma.
5.11 Renal Impairment
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be used with caution in patients with severe renal impairment [see Use in Specific Populations (8.6); Pharmacokinetics (12.3)].
5.12 Hepatic Impairment
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be used with caution in patients with severe hepatic impairment [see Use in Specific Populations (8.7)].
6 ADVERSE REACTIONS
Use of hydrocodone bitartrate, a semisynthetic opioid, may result in the following:
-
Respiratory depression [see Warnings and Precautions (5.1) and Overdosage (10)]
-
Drug dependence [see Warnings and Precautions (5.2)]
-
Increased intracranial pressure [see Warnings and Precautions (5.3) and Overdosage (10)]
-
Decreased mental alertness with impaired mental and/or physical abilities [see Warnings and Precautions(5.4)]
-
Paralytic ileus [see Warnings and Precautions (5.5)]
Use of pseudoephedrine, a sympathomimetic amine, may result in the following:
-
Central nervous system effects such as insomnia, dizziness, weakness, tremor, or convulsions [see Warnings and Precautions (5.8)]
-
Cardiovascular system effects such as arrhythmias, or increased blood pressure, cardiovascular collapse with accompanying hypotension [see Warnings and Precautions (5.8)]
The most common adverse reactions are central nervous system and cardiovascular reactions and include the following: Sedation, drowsiness, mental clouding, lethargy, impairment of mental and physical performance, anxiety, fear, dysphoria, dizziness, psychic dependence, mood changes, nervousness, sleeplessness, tremor or arrhythmia.
Other adverse reactions include:
Gastrointestinal System: Nausea and vomiting (more frequent in ambulatory than in recumbent patients), constipation.
Genitourinary System: Ureteral spasm, spasm of vesicle sphincters, urinary retention.
Cardiovascular System: Fast, slow heartbeat, hypertension, hypotension, orthostatic hypotension, palpitation, shock-like state, syncope.
Dermatological System: Skin rash, pruritus.
7 DRUG INTERACTIONS
No specific interaction studies have been conducted with hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution.
7.1 Opioids, Antihistamines, Antipsychotics, Anti-anxiety Agents, or Other CNS Depressants (Including Alcohol)
The use of opioids, antihistamines, antipsychotics, anti-anxiety agents, or other CNS depressants concomitantly with hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution may cause an additive CNS depressant effect and should be avoided.
7.2 Monoamine Oxidase Inhibitors and Tricyclic Antidepressants
Do not prescribe hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution if the patient is taking a prescription MAOI (i.e., certain drugs used for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping a MAOI drug. The use of MAOIs or tricyclic antidepressants with hydrocodone preparations may increase the effect of either the antidepressant or hydrocodone. An increase in blood pressure or hypertensive crisis may also occur when pseudoephedrine containing preparations are used with MAOIs [see Warnings and Precautions (5.7)].
7.3 Anticholinergic Drugs
Hydrocodone should be administered cautiously to persons receiving anticholinergic drugs in order to avoid paralytic ileus and excessive anticholinergic effects [see Warnings and Precautions (5.6)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C
There are no adequate and well controlled studies of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution in pregnant women. Reproductive toxicity studies have not been conducted with hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution; however, studies are available with an individual active ingredient or related active ingredient. Hydrocodone was teratogenic in hamsters. Codeine, an opiate related to hydrocodone, increased resorptions and decreased fetal weight in rats. Because animal reproduction studies are not always predictive of human response, hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be used during pregnancy only if the benefit justifies the potential risk to the fetus.
Hydrocodone:
Hydrocodone has been shown to be teratogenic in hamsters when given in a dose approximately 35 times the maximum recommended human daily dose (MRHDD) (on a mg/m2 basis at a single subcutaneous dose of 102 mg/kg on gestation day 8). Reproductive toxicology studies were also conducted with codeine, an opiate related to hydrocodone. In a study in which pregnant rats were dosed throughout organogenesis, a dose of codeine approximately 50 times the MRHDD of hydrocodone (on a mg/m2 basis at an oral dose of 120 mg/kg/day of codeine) increased dermatological System: Skin rash, pruritus. resorptions and decreased fetal weight; however, these effects occurred in the presence of maternal toxicity. In studies in which rabbits and mice were dosed throughout organogenesis, doses of codeine up to approximately 25 and 120 times, respectively, the MRHDD of hydrocodone (on a mg/m2 basis at oral doses of 30 and 600 mg/kg/day, respectively), produced no adverse developmental effects.
Nonteratogenic Effects: Babies born to mothers who have been taking opioids regularly prior to delivery will be physically dependent. The withdrawal signs include irritability and excessive crying, tremors, hyperactive reflexes, increased respiratory rate, increased stools, sneezing, yawning, vomiting, and fever. The intensity of the syndrome does not always correlate with the duration of maternal opioid use or dose.
8.2 Labor and Delivery
As with all opioids, administration of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution to the mother shortly before delivery may result in some degree of respiratory depression in the newborn, especially if higher doses are used.
8.3 Nursing Mothers
Caution should be exercised when Hydrocodone Bitartrate and Pseudoephedrine Hydrochloride is administered to nursing mothers. Hydrocodone and pseudoephedrine are excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Safety and effectiveness of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution in pediatric patients under 18 years of age have not been established. The use of hydrocodone in children less than 6 years of age has been associated with fatal respiratory depression [see Warnings and Precautions (5.1)].
8.5 Geriatric Use
Clinical studies have not been conducted with hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution. Other reported clinical experience with the individual active ingredients of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution has not identified differences in responses between the elderly and patients younger than 65 years of age. In general, dose selection for an elderly patient should be made with caution, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. The pseudoephedrine contained in hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is known to be substantially excreted by the kidney and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
8.6 Renal Impairment
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be given with caution in patients with severe impairment of renal function. Pseudoephedrine is primarily excreted unchanged in the urine as unchanged drug with the remainder apparently being metabolized in the liver. Therefore, pseudoephedrine may accumulate in patients with renal impairment.
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is a Schedule II controlled prescription containing hydrocodone bitartrate and should be prescribed and administered with caution.
9.2 Abuse
Hydrocodone can produce drug dependence of the morphine type and therefore, has the potential for being abused. Psychic dependence, physical dependence, and tolerance may develop upon repeated administration of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution, and it should be prescribed and administered with the same degree of caution appropriate to the use of other opioid drugs.
9.3 Dependence
Psychic dependence, physical dependence, and tolerance may develop upon repeated administration of opioids; therefore, hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution should be prescribed and administered with caution.
Physical dependence, the condition in which continued administration of the drug is required to prevent the appearance of a withdrawal syndrome, assumes clinically significant proportions only after several weeks of continued oral opioid use, although some mild degree of physical dependence may develop after a few days of opioid therapy.
10 OVERDOSAGE
No human overdosage data are available for hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution.
Hydrocodone:
Overdosage with hydrocodone is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, and sometimes bradycardia and hypotension. In severe overdosage, apnea, circulatory collapse, cardiac arrest, and death may occur.
Pseudoephedrine:
Overdosage with sympathomimetics, such as pseudoephedrine, may give rise to giddiness, headache, nausea, vomiting, sweating, thirst, tachycardia, precordial pain, palpitations, difficulty in micturition, muscle weakness and tenseness, anxiety, restlessness, and insomnia. Many patients can present a toxic psychosis with delusion and hallucinations. Some may develop cardiac arrhythmias, circulatory collapse, convulsion, coma, and respiratory failure.
Treatment of overdosage consists of discontinuation of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution together with institution of appropriate therapy. Primary attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and the institution of assisted or controlled ventilation. The opioid antagonist naloxone hydrochloride is a specific antidote for respiratory depression which may result from overdosage or unusual sensitivity to opioids including hydrocodone. Therefore, an appropriate dose of naloxone hydrochloride should be administered, preferably by the intravenous route, simultaneously with efforts at respiratory resuscitation. For further information, see full prescribing information for naloxone hydrochloride. An antagonist should not be administered in the absence of clinically significant respiratory depression. Oxygen, intravenous fluids, vasopressors, and other supportive measures should be employed as indicated. Gastric emptying may be useful in removing unabsorbed drug.
11 DESCRIPTION
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution contains hydrocodone bitartrate (a semisynthetic centrally-acting opioid antitussive) and pseudoephedrine hydrochloride (a sympathomimetic amine).
Each 5 mL dose of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution contains: hydrocodone bitartrate, USP, 5 mg; and pseudoephedrine hydrochloride, USP, 60 mg.
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution also contains: anhydrous citric acid, glycerin, grape flavor, methylparaben, propylene glycol, propylparaben, purified water, sodium citrate, sodium saccharin, and sucrose
Hydrocodone bitartrate is morphinan-6-one, 4,5-epoxy-3-methoxy-17-methyl-, (5α)-,[R-(R*,R*)]-2,3-dihydroxybutanedioate (1:1), hydrate (2:5); also known as 4,5α-Epoxy-3-methoxy-17-methylmorphinan-6-one tartrate (1:1) hydrate (2:5); a fine white crystal or crystalline powder, which is derived from the opium alkaloid, thebaine; and may be represented by the following structural formula:
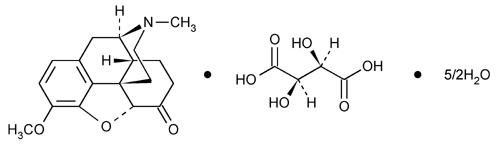
Hydrocodone Bitartrate
C18H21NO3 ● C4H6O6 ● 5/2 H2O
Molecular weight = 494.5
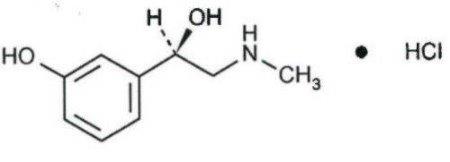
Pseudoephedrine hydrochloride is benzenemethanol, α-[1-(methylamino)ethyl]-, [S-(R*,R*)] hydrochloride and has the following chemical structure:
Pseudoephedrine Hydrochloride
C10H15NO● HCl
Molecular weight = 201.69
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Hydrocodone is a semisynthetic narcotic antitussive and analgesic with multiple actions qualitatively similar to those of codeine. The precise mechanism of action of hydrocodone and other opiates is not known; however, hydrocodone is believed to act directly on the cough center. In excessive doses, hydrocodone will depress respiration. Hydrocodone can produce miosis, euphoria, and physical and physiological dependence.
Pseudoephedrine hydrochloride is an orally active sympathomimetic amine and exerts a decongestant action on the nasal mucosa. Pseudoephedrine hydrochloride is recognized as an effective agent for the relief of nasal congestion due to the common cold. Pseudoephedrine produces peripheral effects similar to those of ephedrine and central effects similar to, but less intense than, amphetamines. It has the potential for excitatory side effects.
12.3 Pharmacokinetics
Systemic exposure (in terms of peak plasma concentrations and area under plasma concentration versus time curve) of hydrocodone bitartrate and pseudoephedrine hydrochloride after single-dose administration of 5 mg hydrocodone and 60 mg pseudoephedrine are equivalent to respective reference solutions of 5 mL hydrocodone bitartrate (5 mg/5 mL) and 5 mL pseudoephedrine hydrochloride (60 mg/5 mL).
Hydrocodone had a mean (SD) peak plasma concentration of 10.6 (2.63) ng/mL at 1.4 (0.55) hours. The mean plasma half-life of hydrocodone is approximately 4.9 hours. Pseudoephedrine had a mean (SD) peak plasma concentration of 212 (46.2) ng/mL at 1.8 (0.56) hours. The mean plasma half-life of pseudoephedrine is approximately 5.6 hours.
Specific Populations
Renal Impairment
Pseudoephedrine is primarily excreted unchanged in the urine as unchanged drug with the remainder apparently being metabolized in the liver. Therefore, pseudoephedrine may accumulate in patients with renal impairment.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis and Mutagenesis and Impairment of Fertility
Carcinogenicity, mutagenicity, and reproductive studies have not been conducted with hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution; however, published information is available for the individual active ingredients or related active ingredients.
Hydrocodone:
Carcinogenicity studies were conducted with codeine, an opiate related to hydrocodone. In 2 year studies in F344/N rats and B6C3F1 mice, codeine showed no evidence of tumorigenicity at dietary doses up to 70 and 400 mg/kg/day, respectively (approximately 30 and 80 times, respectively, the MRHDD of hydrocodone on a mg/m2 basis).
Pseudoephedrine:
Two-year feeding studies in rats and mice demonstrated no evidence of carcinogenic potential with ephedrine sulfate, a structurally related drug with pharmacological properties similar to pseudoephedrine, at dietary doses up to 10 and 27 mg/kg, respectively (approximately 0.3 and 0.5 times, respectively, the MRHDD of pseudoephedrine hydrochloride on a mg/m2 basis).
14 CLINICAL STUDIES
Efficacy studies were not conducted with hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution. Efficacy of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is based on demonstration of bioequivalence to the individual reference products [see Pharmacokinetics (12.3)].
16 HOW SUPPLIED
Hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution is supplied as a clear, colorless solution containing 5 mg hydrocodone bitartrate, USP, and 60 mg pseudoephedrine hydrochloride, USP in each 5 mL. It is available in bottles of 16 fluid ounces (473 mL), NDC 27808-088-01.
Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a tight, light-resistant container, as defined in the USP, with a child-resistant closure.
17 PATIENT COUNSELING INFORMATION
[See FDA-Approved Patient Labeling]
17.1 Overdosage
Patients should be advised not to increase the dose or dosing frequency of hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution because serious adverse events such as respiratory depression may occur with overdosage [see Warnings and Precautions (5.1) ; Overdosage (10)].
17.2 Dosing
Patients should be advised to measure hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution with an accurate milliliter measuring device. Patients should be informed that a household teaspoon is not an accurate measuring device and could lead to overdosage, especially when half a teaspoon is measured. Patients should be advised to ask their pharmacist to recommend an appropriate measuring device and for instructions for measuring the correct dose [see Dosing and Administration (2) Warnings and Precautions (5.9)].
17.3 Concomitant Use of Alcohol and Other Central Nervous System Depressants
Patients should be advised to avoid the use of alcohol and other central nervous system depressants while taking hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution because additional reduction in mental alertness may occur [see Warnings and Precautions (5.4)].
17.4 Activities Requiring Mental Alertness
Patients should be advised to avoid engaging in hazardous tasks that require mental alertness and motor coordination such as operating machinery or driving a motor vehicle as hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution may produce marked drowsiness [see Warnings and Precautions (5.4)].
17.5 Drug Dependence
Patients should be cautioned that hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution contains hydrocodone bitartrate and can produce drug dependence [see Warnings and Precautions (5.2)].
17.6 MAOIs
Patients should be informed that due to its pseudoephedrine component, they should not use hydrocodone bitartrate and pseudoephedrine hydrochloride oral solution with a MAOI or within 14 days of stopping the use of an MAOI [see Warnings and Precautions (5.7)].
Manufactured by
Tris Pharma, Inc.
Monmouth Junction, NJ 08852
www.trispharma.com
LB8263
Rev 00
October 2014
| HYDROCODONE BITARTRATE AND PSEUDOEPHEDRINE HYDROCHLORIDE
SOLUTION
hydrocodone bitartrate and pseudoephedrine hydrochloride solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Tris Pharma Inc (947472119) |
