DOXAZOSIN- doxazosin mesylate tablet
Preferred Pharmaceuticals, Inc
----------
DOXAZOSIN TABLETS USP
Rx only
DESCRIPTION
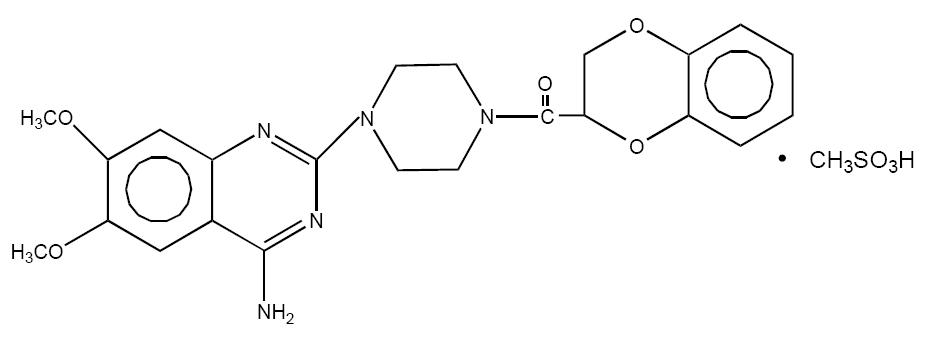
Doxazosin mesylate is a quinazoline compound that is a selective inhibitor of the alpha1 subtype of alpha-adrenergic receptors. The chemical name of doxazosin mesylate is 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(1,4-benzodioxan-2-ylcarbonyl) piperazine methanesulfonate. It has the following structure:
C23H25N5O5•CH4O3S M.W. 547.6
Doxazosin mesylate is freely soluble in dimethylsulfoxide, soluble in dimethylformamide, slightly soluble in methanol, ethanol, and water (0.8% at 25°C), and very slightly soluble in acetone and methylene chloride. Each doxazosin tablet USP, for oral administration, contains 1 mg, 2 mg, 4 mg, or 8 mg of doxazosin as the free base.
The inactive ingredients for all tablets are: microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, magnesium stearate, pregelatinized starch, and sodium lauryl sulfate.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Benign Prostatic Hyperplasia (BPH)
Benign prostatic hyperplasia (BPH) is a common cause of urinary outflow obstruction in aging males. Severe BPH may lead to urinary retention and renal damage. A static and a dynamic component contribute to the symptoms and reduced urinary flow rate associated with BPH. The static component is related to an increase in prostate size caused, in part, by a proliferation of smooth muscle cells in the prostatic stroma. However, the severity of BPH symptoms and the degree of urethral obstruction do not correlate well with the size of the prostate. The dynamic component of BPH is associated with an increase in smooth muscle tone in the prostate and bladder neck. The degree of tone in this area is mediated by the alpha1 adrenoceptor, which is present in high density in the prostatic stroma, prostatic capsule and bladder neck. Blockade of the alpha1 receptor decreases urethral resistance and may relieve the obstruction and BPH symptoms. In the human prostate, doxazosin mesylate antagonizes phenylephrine (alpha1 agonist)-induced contractions, in vitro, and binds with high affinity to the alpha1c adrenoceptor. The receptor subtype is thought to be the predominant functional type in the prostate. Doxazosin mesylate acts within 1 to 2 weeks to decrease the severity of BPH symptoms and improve urinary flow rate. Since alpha1 adrenoceptors are of low density in the urinary bladder (apart from the bladder neck), doxazosin mesylate should maintain bladder contractility.
The efficacy of doxazosin mesylate was evaluated extensively in over 900 patients with BPH in double-blind, placebo-controlled trials. Doxazosin mesylate treatment was superior to placebo in improving patient symptoms and urinary flow rate. Significant relief with doxazosin mesylate was seen as early as one week into the treatment regimen, with doxazosin mesylate-treated patients (N = 173) showing a significant (p < 0.01) increase in maximum flow rate of 0.8 mL/sec compared to a decrease of 0.5 mL/sec in the placebo group (N = 41). In long-term studies, improvement was maintained for up to 2 years of treatment. In 66 to 71% of patients, improvements above baseline were seen in both symptoms and maximum urinary flow rate.
In three placebo-controlled studies of 14 to 16 weeks' duration, obstructive symptoms (hesitation, intermittency, dribbling, weak urinary stream, incomplete emptying of the bladder) and irritative symptoms (nocturia, daytime frequency, urgency, burning) of BPH were evaluated at each visit by patient-assessed symptom questionnaires. The bothersomeness of symptoms was measured with a modified Boyarsky questionnaire. Symptom severity/frequency was assessed using a modified Boyarsky questionnaire or an AUA-based questionnaire. Uroflowmetric evaluations were performed at times of peak (2 to 6 hours post-dose) and/or trough (24 hours post-dose) plasma concentrations of doxazosin mesylate.
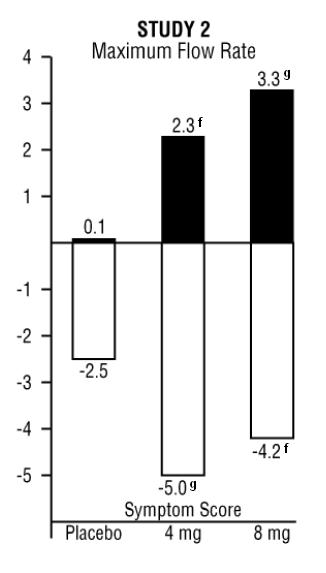
The results from the three placebo-controlled studies (N = 609) showing significant efficacy with 4 mg and 8 mg doxazosin are summarized in TABLE 1. In all three studies, doxazosin mesylate resulted in statistically significant relief of obstructive and irritative symptoms compared to placebo. Statistically significant improvements of 2.3 to 3.3 mL/sec in maximum flow rate were seen with doxazosin mesylate in STUDIES 1 and 2, compared to 0.1 to 0.7 mL/sec with placebo.
|
||||||
|
|
SYMPTOM SCORE* |
MAXIMUM FLOW RATE (mL/sec) |
||||
|
N |
MEAN BASELINE |
MEANCHANGE† |
N |
MEAN BASELINE |
MEANCHANGE‡ |
|
|
STUDY 1 (Titration to maximum dose of 8 mg)§ |
||||||
|
Placebo |
47 |
15.6 |
-2.3 |
41 |
9.7 |
+0.7 |
|
Doxazosin mesylate |
49 |
14.5 |
-4.9¶ |
41 |
9.8 |
+2.9¶ |
|
STUDY 2 (Titration to fixed dose-14 weeks)# |
||||||
|
Placebo |
37 |
20.7 |
-2.5 |
30 |
10.6 |
+0.1 |
|
Doxazosin mesylate 4 mg |
38 |
21.2 |
-5.0¶ |
32 |
9.8 |
+2.3Þ |
|
Doxazosin mesylate 8 mg |
42 |
19.9 |
-4.2Þ |
36 |
10.5 |
+3.3¶ |
|
STUDY 3 (Titration to fixed dose-12 weeks) |
||||||
|
Placebo |
47 |
14.9 |
-4.7 |
44 |
9.9 |
+2.1 |
|
Doxazosin mesylate 4 mg |
46 |
16.6 |
-6.1Þ |
46 |
9.6 |
+2.6 |
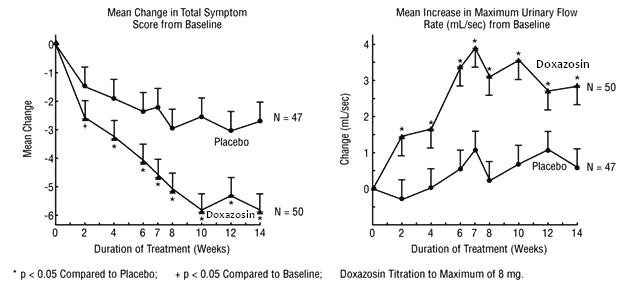
In one fixed-dose study (STUDY 2), doxazosin mesylate therapy (4 to 8 mg, once daily) resulted in a significant and sustained improvement in maximum urinary flow rate of 2.3 to 3.3 mL/sec (TABLE 1) compared to placebo (0.1 mL/sec). In this study, the only study in which weekly evaluations were made, significant improvement with doxazosin mesylate vs. placebo was seen after one week. The proportion of patients who responded with a maximum flow rate improvement of ≥ 3 mL/sec was significantly larger with doxazosin mesylate (34 to 42%) than placebo (13 to 17%). A significantly greater improvement was also seen in average flow rate with doxazosin mesylate (1.6 mL/sec) than with placebo (0.2 mL/sec). The onset and time course of symptom relief and increased urinary flow from STUDY 1 are illustrated in Figure 1.
In BPH patients (N = 450) treated for up to 2 years in open-label studies, doxazosin mesylate therapy resulted in significant improvement above baseline in urinary flow rates and BPH symptoms. The significant effects of doxazosin mesylate were maintained over the entire treatment period.
Although blockade of alpha1 adrenoceptors also lowers blood pressure in hypertensive patients with increased peripheral vascular resistance, doxazosin mesylate treatment of normotensive men with BPH did not result in a clinically significant blood pressure lowering effect (TABLE 2). The proportion of normotensive patients with a sitting systolic blood pressure less than 90 mmHg and/or diastolic blood pressure less than 60 mmHg at any time during treatment with doxazosin mesylate 1 to 8 mg once daily was 6.7% with doxazosin and not significantly different (statistically) from that with placebo (5%).
|
||||
|
PLACEBO (N = 85) |
DOXAZOSIN MESYLATE (N = 183) |
|||
|
Sitting BP (mmHg) |
Baseline |
Change |
Baseline |
Change |
|
Systolic |
128.4 |
-1.4 |
128.8 |
-4.9* |
|
Diastolic |
79.2 |
-1.2 |
79.6 |
-2.4* |
|
Standing BP (mmHg) |
Baseline |
Change |
Baseline |
Change |
|
Systolic |
128.5 |
-0.6 |
128.5 |
-5.3* |
|
Diastolic |
80.5 |
-0.7 |
80.4 |
-2.6* |
Hypertension
The mechanism of action of doxazosin mesylate is selective blockade of the alpha1 (postjunctional) subtype of adrenergic receptors. Studies in normal human subjects have shown that doxazosin competitively antagonized the pressor effects of phenylephrine (an alpha1 agonist) and the systolic pressor effect of norepinephrine. Doxazosin and prazosin have similar abilities to antagonize phenylephrine. The antihypertensive effect of doxazosin mesylate results from a decrease in systemic vascular resistance. The parent compound doxazosin is primarily responsible for the antihypertensive activity. The low plasma concentrations of known active and inactive metabolites of doxazosin (2-piperazinyl, 6΄- and 7΄-hydroxy and 6- and 7-O-desmethyl compounds) compared to parent drug indicate that the contribution of even the most potent compound (6΄-hydroxy) to the antihypertensive effect of doxazosin in man is probably small. The 6΄- and 7΄-hydroxy metabolites have demonstrated antioxidant properties at concentrations of 5 μM, in vitro.
Administration of doxazosin mesylate results in a reduction in systemic vascular resistance. In patients with hypertension, there is little change in cardiac output. Maximum reductions in blood pressure usually occur 2 to 6 hours after dosing and are associated with a small increase in standing heart rate. Like other alpha1-adrenergic blocking agents, doxazosin has a greater effect on blood pressure and heart rate in the standing position.
In a pooled analysis of placebo-controlled hypertension studies with about 300 hypertensive patients per treatment group, doxazosin, at doses of 1 to 16 mg given once daily, lowered blood pressure at 24 hours by about 10/8 mmHg compared to placebo in the standing position and about 9/5 mmHg in the supine position. Peak blood pressure effects (1 to 6 hours) were larger by about 50 to 75% (i.e., trough values were about 55 to 70% of peak effect), with the larger peak-trough differences seen in systolic pressures. There was no apparent difference in the blood pressure response of Caucasians and blacks or of patients above and below age 65. In these predominantly normocholesterolemic patients, doxazosin produced small reductions in total serum cholesterol (2 to 3%), LDL cholesterol (4%), and a similarly small increase in HDL/total cholesterol ratio (4%). The clinical significance of these findings is uncertain. In the same patient population, patients receiving doxazosin mesylate gained a mean of 0.6 kg compared to a mean loss of 0.1 kg for placebo patients.
Pharmacokinetics
After oral administration of therapeutic doses, peak plasma levels of doxazosin mesylate occur at about 2 to 3 hours. Bioavailability is approximately 65%, reflecting first-pass metabolism of doxazosin by the liver. The effect of food on the pharmacokinetics of doxazosin mesylate was examined in a crossover study with twelve hypertensive subjects. Reductions of 18% in mean maximum plasma concentration and 12% in the area under the concentration-time curve occurred when doxazosin mesylate was administered with food. Neither of these differences was statistically or clinically significant.
Doxazosin mesylate is extensively metabolized in the liver, mainly by O-demethylation of the quinazoline nucleus or hydroxylation of the benzodioxan moiety. Although several active metabolites of doxazosin have been identified, the pharmacokinetics of these metabolites have not been characterized. In a study of two subjects administered radiolabelled doxazosin 2 mg orally and 1 mg intravenously on two separate occasions, approximately 63% of the dose was eliminated in the feces and 9% of the dose was found in the urine. On average only 4.8% of the dose was excreted as unchanged drug in the feces and only a trace of the total radioactivity in the urine was attributed to unchanged drug. At the plasma concentrations achieved by therapeutic doses, approximately 98% of the circulating drug is bound to plasma proteins.
Plasma elimination of doxazosin is biphasic, with a terminal elimination half-life of about 22 hours. Steady-state studies in hypertensive patients given doxazosin doses of 2 to 16 mg once daily showed linear kinetics and dose proportionality. In two studies, following the administration of 2 mg orally once daily, the mean accumulation ratios (steady-state AUC vs. first-dose AUC) were 1.2 and 1.7. Enterohepatic recycling is suggested by secondary peaking of plasma doxazosin concentrations.
In a crossover study in 24 normotensive subjects, the pharmacokinetics and safety of doxazosin were shown to be similar with morning and evening dosing regimens. The area under the curve after morning dosing was, however, 11% less than that after evening dosing and the time to peak concentration after evening dosing occurred significantly later than that after morning dosing (5.6 hr vs. 3.5 hr).
The pharmacokinetics of doxazosin mesylate in young (< 65 years) and elderly (≥ 65 years) subjects were similar for plasma half-life values and oral clearance. Pharmacokinetic studies in elderly patients and patients with renal impairment have shown no significant alterations compared to younger patients with normal renal function. Administration of a single 2 mg dose to patients with cirrhosis (Child-Pugh Class A) showed a 40% increase in exposure to doxazosin. There are only limited data on the effects of drugs known to influence the hepatic metabolism of doxazosin [e.g., cimetidine (see PRECAUTIONS, Drug Interactions)]. As with any drug wholly metabolized by the liver, use of doxazosin mesylate in patients with altered liver function should be undertaken with caution.
In two placebo-controlled studies of normotensive and hypertensive BPH patients, in which doxazosin was administered in the morning and the titration interval was two weeks and one week, respectively, trough plasma concentrations of doxazosin mesylate were similar in the two populations. Linear kinetics and dose proportionality were observed.
INDICATIONS AND USAGE
Benign Prostatic Hyperplasia (BPH)
Doxazosin mesylate is indicated for the treatment of both the urinary outflow obstruction and obstructive and irritative symptoms associated with BPH: obstructive symptoms (hesitation, intermittency, dribbling, weak urinary stream, incomplete emptying of the bladder) and irritative symptoms (nocturia, daytime frequency, urgency, burning). Doxazosin mesylate may be used in all BPH patients whether hypertensive or normotensive. In patients with hypertension and BPH, both conditions were effectively treated with doxazosin mesylate monotherapy. Doxazosin mesylate provides rapid improvement in symptoms and urinary flow rate in 66 to 71% of patients. Sustained improvements with doxazosin mesylate were seen in patients treated for up to 14 weeks in double-blind studies and up to 2 years in open-label studies.
CONTRAINDICATIONS
Doxazosin mesylate is contraindicated in patients with a known sensitivity to quinazolines (e.g., prazosin, terazosin), doxazosin, or any of the inert ingredients.
WARNINGS
Syncope and “First-dose” Effect
Doxazosin, like other alpha-adrenergic blocking agents, can cause marked hypotension, especially in the upright position, with syncope and other postural symptoms such as dizziness. Marked orthostatic effects are most common with the first dose but can also occur when there is a dosage increase, or if therapy is interrupted for more than a few days. To decrease the likelihood of excessive hypotension and syncope, it is essential that treatment be initiated with the 1 mg dose. The 2, 4, and 8 mg tablets are not for initial therapy. Dosage should then be adjusted slowly (see DOSAGE AND ADMINISTRATION), with evaluations and increases in dose every two weeks to the recommended dose. Additional antihypertensive agents should be added with caution.
Patients being titrated with doxazosin should be cautioned to avoid situations where injury could result should syncope occur, during both the day and night.
In an early investigational study of the safety and tolerance of increasing daily doses of doxazosin in normotensives beginning at 1 mg/day, only 2 of 6 subjects could tolerate more than 2 mg/day without experiencing symptomatic postural hypotension. In another study of 24 healthy normotensive male subjects receiving initial doses of 2 mg/day of doxazosin, seven (29%) of the subjects experienced symptomatic postural hypotension between 0.5 and 6 hours after the first dose, necessitating termination of the study. In this study, 2 of the normotensive subjects experienced syncope. Subsequent trials in hypertensive patients always began doxazosin dosing at 1 mg/day, resulting in a 4% incidence of postural side effects at 1 mg/day with no cases of syncope.
In multiple-dose clinical trials in hypertension involving over 1500 hypertensive patients with dose titration every one to two weeks, syncope was reported in 0.7% of patients. None of these events occurred at the starting dose of 1 mg, and 1.2% (8/664) occurred at 16 mg/day.
In placebo-controlled clinical trials in BPH, 3 out of 665 patients (0.5%) taking doxazosin reported syncope. Two of the patients were taking 1 mg doxazosin, while one patient was taking 2 mg doxazosin when syncope occurred. In the open-label, long-term extension follow-up of approximately 450 BPH patients, there were 3 reports of syncope (0.7%). One patient was taking 2 mg, one patient was taking 8 mg, and one patient was taking 12 mg when syncope occurred. In a clinical pharmacology study, one subject receiving 2 mg experienced syncope.
If syncope occurs, the patient should be placed in a recumbent position and treated supportively as necessary.
Priapism
Rarely (probably less frequently than once in every several thousand patients), alpha1 antagonists, including doxazosin, have been associated with priapism (painful penile erection, sustained for hours and unrelieved by sexual intercourse or masturbation). Because this condition can lead to permanent impotence if not promptly treated, patients must be advised about the seriousness of the condition (see PRECAUTIONS, Information for Patients).
PRECAUTIONS
General
Prostate Cancer
Carcinoma of the prostate causes many of the symptoms associated with BPH and the two disorders frequently co-exist. Carcinoma of the prostate should therefore be ruled out prior to commencing therapy with doxazosin mesylate.
Cataract Surgery
Intraoperative Floppy Iris Syndrome (IFIS) has been observed during cataract surgery in some patients on or previously treated with alpha1 blockers. This variant of small pupil syndrome is characterized by the combination of a flaccid iris that billows in response to intraoperative irrigation currents, progressive intraoperative miosis despite preoperative dilation with standard mydriatic drugs, and potential prolapse of the iris toward the phacoemulsification incisions. The patient’s surgeon should be prepared for possible modifications to their surgical technique, such as the utilization of iris hooks, iris dilator rings, or viscoelastic substances. There does not appear to be a benefit of stopping alpha1 blocker therapy prior to cataract surgery.
Orthostatic Hypotension
While syncope is the most severe orthostatic effect of doxazosin mesylate, other symptoms of lowered blood pressure, such as dizziness, lightheadedness, or vertigo can occur, especially at initiation of therapy or at the time of dose increases.
Hypertension
These symptoms were common in clinical trials in hypertension, occurring in up to 23% of all patients treated and causing discontinuation of therapy in about 2%.
In placebo-controlled titration trials in hypertension, orthostatic effects were minimized by beginning therapy at 1 mg per day and titrating every two weeks to 2, 4, or 8 mg per day. There was an increased frequency of orthostatic effects in patients given 8 mg or more, 10%, compared to 5% at 1 to 4 mg and 3% in the placebo group.
Benign prostatic hyperplasia
In placebo-controlled trials in BPH, the incidence of orthostatic hypotension with doxazosin was 0.3% and did not increase with increasing dosage (to 8 mg/day). The incidence of discontinuations due to hypotensive or orthostatic symptoms was 3.3% with doxazosin and 1% with placebo. The titration interval in these studies was one to two weeks.
Patients in occupations in which orthostatic hypotension could be dangerous should be treated with particular caution. As alpha1 antagonists can cause orthostatic effects, it is important to evaluate standing blood pressure two minutes after standing, and patients should be advised to exercise care when arising from a supine or sitting position.
If hypotension occurs, the patient should be placed in the supine position and, if this measure is inadequate, volume expansion with intravenous fluids or vasopressor therapy may be used. A transient hypotensive response is not a contraindication to further doses of doxazosin mesylate.
Information for Patients (See Patient Package Insert)
Patients should be made aware of the possibility of syncopal and orthostatic symptoms, especially at the initiation of therapy, and urged to avoid driving or hazardous tasks for 24 hours after the first dose, after a dosage increase, and after interruption of therapy when treatment is resumed. They should be cautioned to avoid situations where injury could result should syncope occur during initiation of doxazosin therapy. They should also be advised of the need to sit or lie down when symptoms of lowered blood pressure occur, although these symptoms are not always orthostatic, and to be careful when rising from a sitting or lying position. If dizziness, lightheadedness, or palpitations are bothersome, they should be reported to the physician, so that dose adjustment can be considered. Patients should also be told that drowsiness or somnolence can occur with doxazosin mesylate or any selective alpha1 adrenoceptor antagonist, requiring caution in people who must drive or operate heavy machinery.
Patients should be advised about the possibility of priapism as a result of treatment with alpha1 antagonists. Patients should know that this adverse event is very rare. If they experience priapism, it should be brought to immediate medical attention, for, if not treated promptly, it can lead to permanent erectile dysfunction (impotence).
Drug/Laboratory Test Interactions
Doxazosin mesylate does not affect the plasma concentration of prostate-specific antigen in patients treated for up to 3 years. Both doxazosin, an alpha1 inhibitor, and finasteride, a 5-alpha reductase inhibitor, are highly protein-bound and hepatically metabolized. There is no definitive controlled clinical experience on the concomitant use of alpha1 inhibitors and 5-alpha reductase inhibitors at this time.
Impaired Liver Function
Doxazosin mesylate should be administered with caution to patients with evidence of impaired hepatic function, or to patients receiving drugs known to influence hepatic metabolism (see CLINICAL PHARMACOLOGY, Pharmacokinetics).
Leukopenia/Neutropenia
Analysis of hematologic data from hypertensive patients receiving doxazosin mesylate in controlled hypertension clinical trials showed that the mean WBC (N = 474) and mean neutrophil counts (N = 419) were decreased by 2.4% and 1.0%, respectively, compared to placebo, a phenomenon seen with other alpha-blocking drugs. In BPH patients, the incidence of clinically significant WBC abnormalities was 0.4% (2/459) with doxazosin mesylate and 0% (0/147) with placebo, with no statistically significant difference between the two treatment groups. A search through a data base of 2400 hypertensive patients and 665 BPH patients revealed 4 hypertensives in which drug-related neutropenia could not be ruled out and one BPH patient in which drug-related leukopenia could not be ruled out. Two hypertensives had a single low value on the last day of treatment. Two hypertensives had stable, non-progressive neutrophil counts in the 1000/mm3 range over periods of 20 and 40 weeks. One BPH patient had a decrease from a WBC count of 4800/mm3 to 2700/mm3 at the end of the study; there was no evidence of clinical impairment. In cases where follow-up was available, the WBCs and neutrophil counts returned to normal after discontinuation of doxazosin mesylate. No patients became symptomatic as a result of the low WBC or neutrophil counts.
Drug Interactions
Most (98%) of plasma doxazosin is protein bound. In vitro data in human plasma indicate that doxazosin mesylate has no effect on protein binding of digoxin, warfarin, phenytoin, or indomethacin. There is no information on the effect of other highly plasma protein-bound drugs on doxazosin binding. Doxazosin mesylate has been administered without any evidence of an adverse drug interaction to patients receiving thiazide diuretics, beta-blocking agents, and non-steroidal anti-inflammatory drugs. In a placebo-controlled trial in normal volunteers, the administration of a single 1 mg dose of doxazosin on day 1 of a four-day regimen of oral cimetidine (400 mg twice daily) resulted in a 10% increase in mean AUC of doxazosin (p = 0.006), and a slight but not statistically significant increase in mean Cmax and mean half-life of doxazosin. The clinical significance of this increase in doxazosin AUC is unknown.
In clinical trials, doxazosin mesylate tablets have been administered to patients on a variety of concomitant medications; while no formal interaction studies have been conducted, no interactions were observed. Doxazosin mesylate tablets have been used with the following drugs or drug classes: 1) analgesic/anti-inflammatory (e.g., acetaminophen, aspirin, codeine and codeine combinations, ibuprofen, indomethacin); 2) antibiotics (e.g., erythromycin, trimethoprim and sulfamethoxazole, amoxicillin); 3) antihistamines (e.g., chlorpheniramine); 4) cardiovascular agents (e.g., atenolol, hydrochlorothiazide, propranolol); 5) corticosteroids; 6) gastrointestinal agents (e.g., antacids); 7) hypoglycemics and endocrine drugs; 8) sedatives and tranquilizers (e.g., diazepam); 9) cold and flu remedies.
Concomitant administration of doxazosin mesylate with a phosphodiesterase-5 (PDE-5) inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension (see DOSAGE AND ADMINISTRATION).
Cardiac Toxicity in Animals
An increased incidence of myocardial necrosis or fibrosis was displayed by Sprague-Dawley rats after 6 months of dietary administration at concentrations calculated to provide 80 mg doxazosin/kg/day, and after 12 months of dietary administration at concentrations calculated to provide 40 mg doxazosin/kg/day (AUC exposure in rats 8 times the human AUC exposure with a 12 mg/day therapeutic dose). Myocardial fibrosis was observed in both rats and mice treated in the same manner with 40 mg doxazosin/kg/day for 18 months (exposure 8 times human AUC exposure in rats and somewhat equivalent to human Cmax exposure in mice). No cardiotoxicity was observed at lower doses (up to 10 or 20 mg/kg/day, depending on the study) in either species. These lesions were not observed after 12 months of oral dosing in dogs at maximum doses of 20 mg/kg/day [maximum plasma concentrations (Cmax) in dogs 14 times the Cmax exposure in humans receiving a 12 mg/day therapeutic dose] and in Wistar rats at doses of 100 mg/kg/day (Cmax exposures 15 times human Cmax exposure with a 12 mg/day therapeutic dose). There is no evidence that similar lesions occur in humans.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Chronic dietary administration (up to 24 months) of doxazosin mesylate at maximally tolerated doses of 40 mg/kg/day in rats and 120 mg/kg/day in mice revealed no evidence of carcinogenic potential. The highest doses evaluated in the rat and mouse studies are associated with AUCs (a measure of systemic exposure) that are 8 times and 4 times, respectively, the human AUC at a dose of 16 mg/day.
Mutagenicity studies revealed no drug- or metabolite-related effects at either chromosomal or subchromosomal levels.
Studies in rats showed reduced fertility in males treated with doxazosin at oral doses of 20 (but not 5 or 10) mg/kg/day, about 4 times the AUC exposures obtained with a 12 mg/day human dose. This effect was reversible within two weeks of drug withdrawal. There have been no reports of any effects of doxazosin on male fertility in humans.
Pregnancy
Teratogenic Effects
Pregnancy category C
Studies in pregnant rabbits and rats at daily oral doses of up to 41 and 20 mg/kg, respectively (plasma drug concentrations 10 and 4 times human Cmax and AUC exposures with a 12 mg/day therapeutic dose), have revealed no evidence of harm to the fetus. A dosage regimen of 82 mg/kg/day in the rabbit was associated with reduced fetal survival. There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, doxazosin mesylate should be used during pregnancy only if clearly needed.
Radioactivity was found to cross the placenta following oral administration of labelled doxazosin to pregnant rats.
Nonteratogenic Effects
In peri-postnatal studies in rats, postnatal development at maternal doses of 40 or 50 mg/kg/day of doxazosin (8 times human AUC exposure with a 12 mg/day therapeutic dose) was delayed, as evidenced by slower body weight gain and slightly later appearance of anatomical features and reflexes.
Nursing Mothers
Studies in lactating rats given a single oral dose of 1 mg/kg of [2-14C]-doxazosin mesylate indicate that doxazosin accumulates in rat breast milk with a maximum concentration about 20 times greater than the maternal plasma concentration. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when doxazosin mesylate is administered to a nursing mother.
Pediatric Use
The safety and effectiveness of doxazosin mesylate as an antihypertensive agent have not been established in children.
Geriatric Use
The safety and effectiveness profile of doxazosin mesylate in BPH was similar in the elderly (age ≥ 65 years) and younger (age < 65 years) patients.
For Hypertension
Clinical studies of doxazosin mesylate did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
ADVERSE REACTIONS
Benign Prostatic Hyperplasia (BPH)
The incidence of adverse events has been ascertained from worldwide clinical trials in 965 BPH patients. The incidence rates presented below (TABLE 3) are based on combined data from seven placebo-controlled trials involving once-daily administration of doxazosin mesylate in doses of 1 to 16 mg in hypertensives and 0.5 to 8 mg in normotensives. The adverse events when the incidence in the doxazosin mesylate group was at least 1% are summarized in TABLE 3. No significant difference in the incidence of adverse events compared to placebo was seen except for dizziness, fatigue, hypotension, edema, and dyspnea. Dizziness and dyspnea appeared to be dose-related.
|
Body System |
DOXAZOSIN MESYLATE (N = 665) |
PLACEBO (N = 300) |
|
BODY AS A WHOLE | ||
|
Back Pain |
1.8% |
2.0% |
|
Chest Pain |
1.2% |
0.7% |
|
Fatigue |
8.0%* |
1.7% |
|
Headache |
9.9% |
9.0% |
|
Influenza-like Symptoms |
1.1% |
1.0% |
|
Pain |
2.0% |
1.0% |
|
CARDIOVASCULAR SYSTEM | ||
|
Hypotension |
1.7%* |
0.0% |
|
Palpitation |
1.2% |
0.3% |
|
DIGESTIVE SYSTEM | ||
|
Abdominal Pain |
2.4% |
2.0% |
|
Diarrhea |
2.3% |
2.0% |
|
Dyspepsia |
1.7% |
1.7% |
|
Nausea |
1.5% |
0.7% |
|
METABOLIC AND NUTRITIONAL DISORDERS | ||
|
Edema |
2.7%* |
0.7% |
|
NERVOUS SYSTEM | ||
|
Dizziness† |
15.6%* |
9.0% |
|
Mouth Dry |
1.4% |
0.3% |
|
Somnolence |
3.0% |
1.0% |
|
RESPIRATORY SYSTEM | ||
|
Dyspnea |
2.6%* |
0.3% |
|
Respiratory Disorder |
1.1% |
0.7% |
|
SPECIAL SENSES | ||
|
Vision Abnormal |
1.4% |
0.7% |
|
UROGENITAL SYSTEM | ||
|
Impotence |
1.1% |
1.0% |
|
Urinary Tract Infection |
1.4% |
2.3% |
|
SKIN & APPENDAGES | ||
|
Sweating Increased |
1.1% |
1.0% |
|
PSYCHIATRIC DISORDERS | ||
|
Anxiety |
1.1% |
0.3% |
|
Insomnia |
1.2% |
0.3% |
In these placebo-controlled studies of 665 doxazosin mesylate patients treated for a mean of 85 days, additional adverse reactions have been reported. These are less than 1% and not distinguishable from those that occurred in the placebo group. Adverse reactions with an incidence of less than 1% but of clinical interest are (doxazosin mesylate vs. placebo): Cardiovascular System: angina pectoris (0.6% vs. 0.7%), postural hypotension (0.3% vs. 0.3%), syncope (0.5% vs. 0.0%), tachycardia (0.9% vs. 0.0%); Urogenital System: dysuria (0.5% vs. 1.3%); and Psychiatric Disorders: libido decreased (0.8% vs. 0.3%). The safety profile in patients treated for up to three years was similar to that in the placebo-controlled studies.
The majority of adverse experiences with doxazosin mesylate were mild.
Hypertension
Doxazosin mesylate has been administered to approximately 4000 hypertensive patients, of whom 1679 were included in the hypertension clinical development program. In that program, minor adverse effects were frequent, but led to discontinuation of treatment in only 7% of patients. In placebo-controlled studies, adverse effects occurred in 49% and 40% of patients in the doxazosin and placebo groups, respectively, and led to discontinuation in 2% of patients in each group. The major reasons for discontinuation were postural effects (2%), edema, malaise/fatigue, and some heart rate disturbance, each about 0.7%.
In controlled hypertension clinical trials directly comparing doxazosin mesylate to placebo, there was no significant difference in the incidence of side effects, except for dizziness (including postural), weight gain, somnolence, and fatigue/malaise. Postural effects and edema appeared to be dose-related. The prevalence rates presented below are based on combined data from placebo-controlled studies involving once-daily administration of doxazosin at doses ranging from 1 to 16 mg. TABLE 4 summarizes those adverse experiences (possibly/probably related) reported for patients in these hypertension studies where the prevalence rate in the doxazosin group was at least 0.5% or where the reaction is of particular interest.
|
|
HYPERTENSION |
|
|
DOXAZOSIN (N = 339) |
PLACEBO (N = 336) |
|
|
CARDIOVASCULAR SYSTEM | ||
|
Dizziness |
19% |
9% |
|
Vertigo |
2% |
1% |
|
Postural Hypotension |
0.3% |
0% |
|
Edema |
4% |
3% |
|
Palpitation |
2% |
3% |
|
Arrhythmia |
1% |
0% |
|
Hypotension |
1% |
0% |
|
Tachycardia |
0.3% |
1% |
|
Peripheral Ischemia |
0.3% |
0% |
|
SKIN & APPENDAGES | ||
|
Rash |
1% |
1% |
|
Pruritus |
1% |
1% |
|
MUSCULOSKELETAL SYSTEM | ||
|
Arthralgia/Arthritis |
1% |
0% |
|
Muscle Weakness |
1% |
0% |
|
Myalgia |
1% |
0% |
|
CENTRAL & PERIPHERAL N.S. | ||
|
Headache |
14% |
16% |
|
Paresthesia |
1% |
1% |
|
Kinetic Disorders |
1% |
0% |
|
Ataxia |
1% |
0% |
|
Hypertonia |
1% |
0% |
|
Muscle Cramps |
1% |
0% |
|
AUTONOMIC | ||
|
Mouth Dry |
2% |
2% |
|
Flushing |
1% |
0% |
|
SPECIAL SENSES | ||
|
Vision Abnormal |
2% |
1% |
|
Conjunctivitis/Eye Pain |
1% |
1% |
|
Tinnitus |
1% |
0.3% |
|
PSYCHIATRIC | ||
|
Somnolence |
5% |
1% |
|
Nervousness |
2% |
2% |
|
Depression |
1% |
1% |
|
Insomnia |
1% |
1% |
|
Sexual Dysfunction |
2% |
1% |
|
GASTROINTESTINAL | ||
|
Nausea |
3% |
4% |
|
Diarrhea |
2% |
3% |
|
Constipation |
1% |
1% |
|
Dyspepsia |
1% |
1% |
|
Flatulence |
1% |
1% |
|
Abdominal Pain |
0% |
2% |
|
Vomiting |
0% |
1% |
|
RESPIRATORY | ||
|
Rhinitis |
3% |
1% |
|
Dyspnea |
1% |
1% |
|
Epistaxis |
1% |
0% |
|
URINARY | ||
|
Polyuria |
2% |
0% |
|
Urinary Incontinence |
1% |
0% |
|
Micturition Frequency |
0% |
2% |
|
GENERAL |
|
|
|
Fatigue/Malaise |
12% |
6% |
|
Chest Pain |
2% |
2% |
|
Asthenia |
1% |
1% |
|
Face Edema |
1% |
0% |
|
Pain |
2% |
2% |
Additional adverse reactions have been reported, but these are, in general, not distinguishable from symptoms that might have occurred in the absence of exposure to doxazosin. The following adverse reactions occurred with a frequency of between 0.5% and 1%: syncope, hypoesthesia, increased sweating, agitation, increased weight. The following additional adverse reactions were reported by < 0.5% of 3960 patients who received doxazosin in controlled or open, short- or long-term clinical studies, including international studies. Cardiovascular System: angina pectoris, myocardial infarction, cerebrovascular accident; Autonomic Nervous System: pallor; Metabolic: thirst, gout, hypokalemia; Hematopoietic: lymphadenopathy, purpura; Reproductive System: breast pain; Skin Disorders: alopecia, dry skin, eczema; Central Nervous System: paresis, tremor, twitching, confusion, migraine, impaired concentration; Psychiatric: paroniria, amnesia, emotional lability, abnormal thinking, depersonalization; Special Senses: parosmia, earache, taste perversion, photophobia, abnormal lacrimation; Gastrointestinal System: increased appetite, anorexia, fecal incontinence, gastroenteritis; Respiratory System: bronchospasm, sinusitis, coughing, pharyngitis; Urinary System: renal calculus; General Body System: hot flushes, back pain, infection, fever/rigors, decreased weight, influenza-like symptoms.
Doxazosin mesylate has not been associated with any clinically significant changes in routine biochemical tests. No clinically relevant adverse effects were noted on serum potassium, serum glucose, uric acid, blood urea nitrogen, creatinine or liver function tests. Doxazosin mesylate has been associated with decreases in white blood cell counts (see PRECAUTIONS, Leukopenia/Neutropenia).
In postmarketing experience, the following additional adverse reactions have been reported: Autonomic Nervous System: priapism; Central Nervous System: hypoesthesia; Endocrine System: gynecomastia; Gastrointestinal System: vomiting; General Body System: allergic reaction; Heart Rate/Rhythm: bradycardia; Hematopoietic: leukopenia, thrombocytopenia; Liver/Biliary System: hepatitis, hepatitis cholestatic; Respiratory System: bronchospasm aggravated; Skin Disorders: urticaria; Special Senses: Intraoperative Floppy Iris Syndrome (see PRECAUTIONS, Cataract Surgery); Urinary System: hematuria, micturition disorder, micturition frequency, nocturia.
OVERDOSAGE
Experience with doxazosin mesylate overdosage is limited. Two adolescents, who each intentionally ingested 40 mg doxazosin mesylate with diclofenac or acetaminophen, were treated with gastric lavage with activated charcoal and made full recoveries. A two-year-old child who accidently ingested 4 mg doxazosin mesylate was treated with gastric lavage and remained normotensive during the five-hour emergency room observation period. A six-month-old child accidentally received a crushed 1 mg tablet of doxazosin mesylate and was reported to have been drowsy. A 32-year-old female with chronic renal failure, epilepsy, and depression intentionally ingested 60 mg doxazosin mesylate (blood level = 0.9 mcg/mL; normal values in hypertensives = 0.02 mcg/mL); death was attributed to a grand mal seizure resulting from hypotension. A 39-year-old female who ingested 70 mg doxazosin mesylate, alcohol, and Dalmane® (flurazepam) developed hypotension which responded to fluid therapy.
The oral LD50 of doxazosin is greater than 1000 mg/kg in mice and rats. The most likely manifestation of overdosage would be hypotension, for which the usual treatment would be intravenous infusion of fluid. As doxazosin is highly protein bound, dialysis would not be indicated.
DOSAGE AND ADMINISTRATION
DOSAGE MUST BE INDIVIDUALIZED. The initial dosage of doxazosin tablets USP in patients with hypertension and/or BPH is 1 mg given once daily in the a.m. or p.m. This starting dose is intended to minimize the frequency of postural hypotension and first-dose syncope associated with doxazosin tablets USP. Postural effects are most likely to occur between 2 and 6 hours after a dose. Therefore, blood pressure measurements should be taken during this time period after the first dose and with each increase in dose. If doxazosin tablet USP administration is discontinued for several days, therapy should be restarted using the initial dosing regimen.
Concomitant administration of doxazosin tablets USP with a PDE-5 inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension; therefore, PDE-5 inhibitor therapy should be initiated at the lowest dose in patients taking doxazosin tablets USP.
Benign Prostatic Hyperplasia 1 to 8 mg once daily
The initial dosage of doxazosin mesylate is 1 mg, given once daily in the a.m. or p.m. Depending on the individual patient’s urodynamics and BPH symptomatology, dosage may then be increased to 2 mg and thereafter to 4 mg and 8 mg once daily, the maximum recommended dose for BPH. The recommended titration interval is 1 to 2 weeks. Blood pressure should be evaluated routinely in these patients.
Hypertension 1 to 16 mg once daily
The initial dosage of doxazosin mesylate is 1 mg given once daily. Depending on the individual patient’s standing blood pressure response (based on measurements taken at 2 to 6 hours post-dose and 24 hours post-dose), dosage may then be increased to 2 mg and thereafter if necessary to 4 mg, 8 mg and 16 mg to achieve the desired reduction in blood pressure. Increases in dose beyond 4 mg increase the likelihood of excessive postural effects, including syncope, postural dizziness/vertigo and postural hypotension. At a titrated dose of 16 mg once daily, the frequency of postural effects is about 12% compared to 3% for placebo.
HOW SUPPLIED
Doxazosin tablets USP, 2 mg (doxazosin as free base), are white to off-white, capsule-shaped, biconvex compressed tablet, debossed modified N over bisect 593 (N/593) on one side and 2 on the other side. They are supplied as follows:
Bottle of 30 - 68788-0320-3
Doxazosin tablets USP, 4 mg (doxazosin as free base), are white to off-white, modified diamond-shaped, biconvex compressed tablet, debossed modified N over bisect 596 (N/596) on one side and 4 on the other side. They are supplied as follows:
Bottle of 30 - 68788-9154
Recommended Storage: Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a well-closed container.
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Teva Pharmaceuticals USA.
Manufactured In Canada By:
NOVOPHARM LIMITED
Toronto, Canada M1B 2K9
Manufactured For:
TEVA PHARMACEUTICALS USA
Sellersville, PA 18960
Rev. K 3/2010
PATIENT INFORMATION
ABOUT DOXAZOSIN TABLETS USP FOR BENIGN PROSTATIC HYPERPLASIA (BPH)
Rx only
Read this leaflet:
- •
- before you start taking doxazosin tablets USP
- •
- each time you get a new prescription
You and your doctor should discuss this treatment and your BPH symptoms before you start taking doxazosin tablets USP and at your regular checkups. This leaflet does NOT take the place of discussions with your doctor.
Doxazosin tablets USP are used to treat both benign prostatic hyperplasia (BPH) and high blood pressure (hypertension). This leaflet describes doxazosin tablets USP as treatment for BPH (although you may be taking doxazosin tablets USP for both your BPH and high blood pressure).
What is BPH?
BPH is an enlargement of the prostate gland. This gland surrounds the tube that drains the urine from the bladder. The symptoms of BPH can be caused by a tensing of the enlarged muscle in the prostate gland which blocks the passage of urine. This can lead to such symptoms as:
- •
- a weak or start-and-stop stream when urinating
- •
- a feeling that the bladder is not completely emptied after urination
- •
- a delay or difficulty in the beginning of urination
- •
- a need to urinate often during the day and especially at night
- •
- a feeling that you must urinate immediately
Treatment Options for BPH
The four main treatment options for BPH are:
- •
- If you are not bothered by your symptoms, you and your doctor may decide on a program of “watchful waiting.” It is not an active treatment like taking medication or surgery but involves having regular checkups to see if your condition is getting worse or causing problems.
- •
- Treatment with doxazosin tablets USP or other similar drugs. Doxazosin tablets USP are the medication your doctor has prescribed for you. See “What Doxazosin Tablets USP Do,” below.
- •
- Treatment with the medication class of 5-alpha reductase inhibitors (e.g., Proscar®). It can cause the prostate to shrink. It may take 6 months or more for the full benefit of finasteride to be seen.
- •
- Various surgical procedures. Your doctor can describe these procedures to you. The best procedure for you depends on your BPH symptoms and medical condition.
What Doxazosin Tablets USP Do
Doxazosin tablets USP work on a specific type of muscle found in the prostate, causing it to relax. This in turn decreases the pressure within the prostate, thus improving the flow of urine and your symptoms.
- •
- Doxazosin tablets USP help relieve the symptoms of BPH (weak stream, start-and-stop stream, a feeling that your bladder is not completely empty, delay in beginning of urination, need to urinate often during the day and especially at night, and feeling that you must urinate immediately). It does not change the size of the prostate. The prostate may continue to grow; however, a larger prostate is not necessarily related to more symptoms or to worse symptoms. Doxazosin tablets USP can decrease your symptoms and improve urinary flow, without decreasing the size of the prostate.
- •
- If doxazosin tablets USP are helping you, you should notice an effect within 1 to 2 weeks after you start your medication. Doxazosin tablets USP have been studied in over 900 patients for up to 2 years and the drug has been shown to continue to work during long-term treatment. Even though you take doxazosin tablets USP and they may help you, doxazosin tablets USP may not prevent the need for surgery in the future.
- •
- Doxazosin tablets USP do not affect PSA levels. PSA is the abbreviation for Prostate Specific Antigen. Your doctor may have done a blood test called PSA. You may want to ask your doctor more about this if you have had a PSA test done.
Other Important Facts
- •
- You should see an improvement of your symptoms within 1 to 2 weeks. In addition to your other regular checkups you will need to continue seeing your doctor regularly to check your progress regarding your BPH and to monitor your blood pressure.
- •
- Doxazosin tablets USP are not a treatment for prostate cancer. Your doctor has prescribed doxazosin tablets USP for your BPH and not for prostate cancer; however, a man can have BPH and prostate cancer at the same time. Doctors usually recommend that men be checked for prostate cancer once a year when they turn 50 (or 40 if a family member has had prostate cancer). A higher incidence of prostate cancer has been noted in men of African-American descent. These checks should continue even if you are taking doxazosin tablets USP.
How To Take Doxazosin Tablets USP and What You Should Know While Taking Doxazosin Tablets USP for BPH
Doxazosin Tablets USP Can Cause a Sudden Drop in Blood Pressure After the VERY FIRST DOSE. You may feel dizzy, faint or “light-headed,” especially after you stand up from a lying or sitting position. This is more likely to occur after you’ve taken the first few doses or if you increase your dose, but can occur at any time while you are taking the drug. It can also occur if you stop taking the drug and then restart treatment. If you feel very dizzy, faint or “light-headed” you should contact your doctor. Your doctor will discuss with you how often you need to visit and how often your blood pressure should be checked.
Your blood pressure should be checked when you start taking doxazosin tablets USP even if you do not have high blood pressure (hypertension). Your doctor will discuss with you the details of how blood pressure is measured.
Blood Pressure Measurement: Whatever equipment is used, it is usual for your blood pressure to be measured in the following way: measure your blood pressure after lying quietly on your back for five minutes. Then, after standing for two minutes measure your blood pressure again. Your doctor will discuss with you what other times during the day your blood pressure should be taken, such as two to six hours after a dose, before bedtime or after waking up in the morning. Note that moderate to high-intensity exercise can, over a period of time, lower your average blood pressure.
You can take doxazosin tablets USP either in the morning or at bedtime and it will be equally effective. If you take doxazosin tablets USP at bedtime but need to get up from bed to go to the bathroom, get up slowly and cautiously until you are sure how the medication affects you. It is important to get up slowly from a chair or bed at any time until you learn how you react to doxazosin tablets USP. You should not drive or do any hazardous tasks until you are used to the effects of the medication. If you begin to feel dizzy, sit or lie down until you feel better.
- •
- You will start with a 1 mg dose of doxazosin tablets USP once daily. Then the once daily dose will be increased as your body gets used to the effects of the medication. Follow your doctor’s instructions about how to take doxazosin tablets USP. You must take it every day at the dose prescribed. Talk with your doctor if you don’t take it for a few days for some reason; you may then need to restart the medication at a 1 mg dose, increase your dose gradually and again be cautious about possible dizziness. Do not share doxazosin tablets USP with anyone else; they were prescribed only for you.
- •
- Other side effects you could have while taking doxazosin tablets USP, in addition to lowering of the blood pressure, include dizziness, fatigue (tiredness), swelling of the feet and shortness of breath. Most side effects are mild. However, you should discuss any unexpected effects you notice with your doctor.
- •
- WARNING: Extremely rarely, doxazosin tablets USP and similar medications have caused painful erection of the penis, sustained for hours and unrelieved by sexual intercourse or masturbation. This condition is serious, and if untreated it can be followed by permanent inability to have an erection. If you have a prolonged abnormal erection, call your doctor or go to an emergency room as soon as possible.
- •
- Tell your surgeon if you take or have taken doxazosin tablets USP if you plan to have surgery for cataracts (clouding of the eye). During cataract surgery, a condition called Intraoperative Floppy Iris Syndrome (IFIS) can happen if you take or have taken doxazosin tablets USP.
- •
- If you use doxazosin tablets USP with an oral erectile dysfunction medicine (phosphodiesterase-5 (PDE-5) inhibitor), it can cause a sudden drop in your blood pressure and you can become dizzy or faint. Talk with your healthcare provider before using PDE-5 inhibitors.
- •
- Keep doxazosin tablets USP and all medicines out of the reach of children.
FOR MORE INFORMATION ABOUT DOXAZOSIN TABLETS USP AND BPH TALK WITH YOUR DOCTOR, NURSE, PHARMACIST OR OTHER HEALTH CARE PROVIDER.
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Teva Pharmaceuticals USA.
Manufactured In Canada By:
NOVOPHARM LIMITED
Toronto, Canada M1B 2K9
Manufactured For:
TEVA PHARMACEUTICALS USA
Sellersville, PA 18960
Rev. B 3/2010
Repackaged By:
Preferred Pharmaceuticals, Inc
Anaheim, CA 92870
| DOXAZOSIN
doxazosin mesylate tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| DOXAZOSIN
doxazosin mesylate tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Preferred Pharmaceuticals, Inc (791119022) |
| Registrant - Preferred Pharmaceuticals, Inc (791119022) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Preferred Pharmaceuticals, Inc | 791119022 | REPACK(68788-0320, 68788-9154) | |