MUCINEX FAST-MAX COLD AND SINUS- acetaminophen, guaifenesin, and phenylephrine hydrochloride tablet, coated
RB Health (US) LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Mucinex® Fast-Max
®
Cold & Sinus
Uses
- temporarily relieves these common cold and flu symptoms:
- nasal congestion
- fever
- headache
- minor aches and pains
- sinus congestion and pressure
- temporarily promotes nasal and/or sinus drainage
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Warnings
Liver warning
This product contains acetaminophen.
Severe liver damage may occur if you take:
- more than 12 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks daily while using this product
Allergy alert
Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease
- heart disease
- diabetes
- high blood pressure
- thyroid disease
- trouble urinating due to an enlarged prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough that occurs with too much phlegm (mucus)
Stop use and ask a doctor if
- nervousness, dizziness or sleeplessness occur
- pain, nasal congestion or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back, or occurs with rash or headache that lasts. These could be signs of a serious condition.
Directions
- do not take more than directed (see Overdose warning)
- do not take more than 12 caplets in any 24-hour period
- adults and children 12 years and older: take 2 caplets every 4 hours
- children under 12 years of age: do not use
Other information
- tamper evident: do not use if outer package is opened or blister is torn or broken
- store between 20-25°C (68-77°F)
Inactive ingredients
croscarmellose sodium, crospovidone, FD&C red no. 40 aluminum lake, FD&C yellow no. 6 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, talc, titanium dioxide
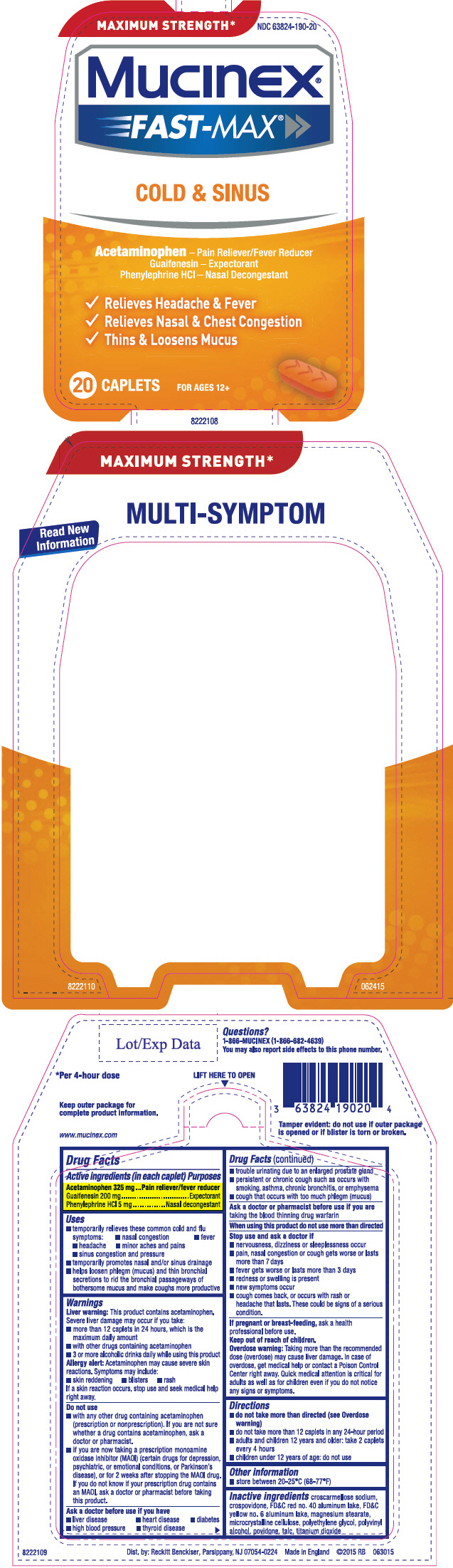
PRINCIPAL DISPLAY PANEL - 20 Caplet Blister Pack Carton
MAXIMUM STRENGTH*
NDC 63824-190-20
Read New
Information
Mucinex®
FAST-MAX ®
COLD & SINUS
Acetaminophen – Pain Reliever/Fever Reducer
Guaifenesin
– Expectorant
Phenylephrine HCl
– Nasal Decongestant
- Relieves Headache & Fever
- Relieves Nasal & Chest Congestion
- Thins & Loosens Mucus
20 CAPLETS
FOR AGES 12+
| MUCINEX FAST-MAX
COLD AND SINUS
acetaminophen, guaifenesin, and phenylephrine hydrochloride tablet, coated |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - RB Health (US) LLC (081049410) |