Label: ELF PERSONAL BLEND FOUNDATION SPF 15- titanium dioxide powder
-
Contains inactivated NDC Code(s)
NDC Code(s): 76166-001-01 - Packager: Shanghai Justking Enterprise Co. Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 1, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- WHEN USING
- ASK DOCTOR
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions:
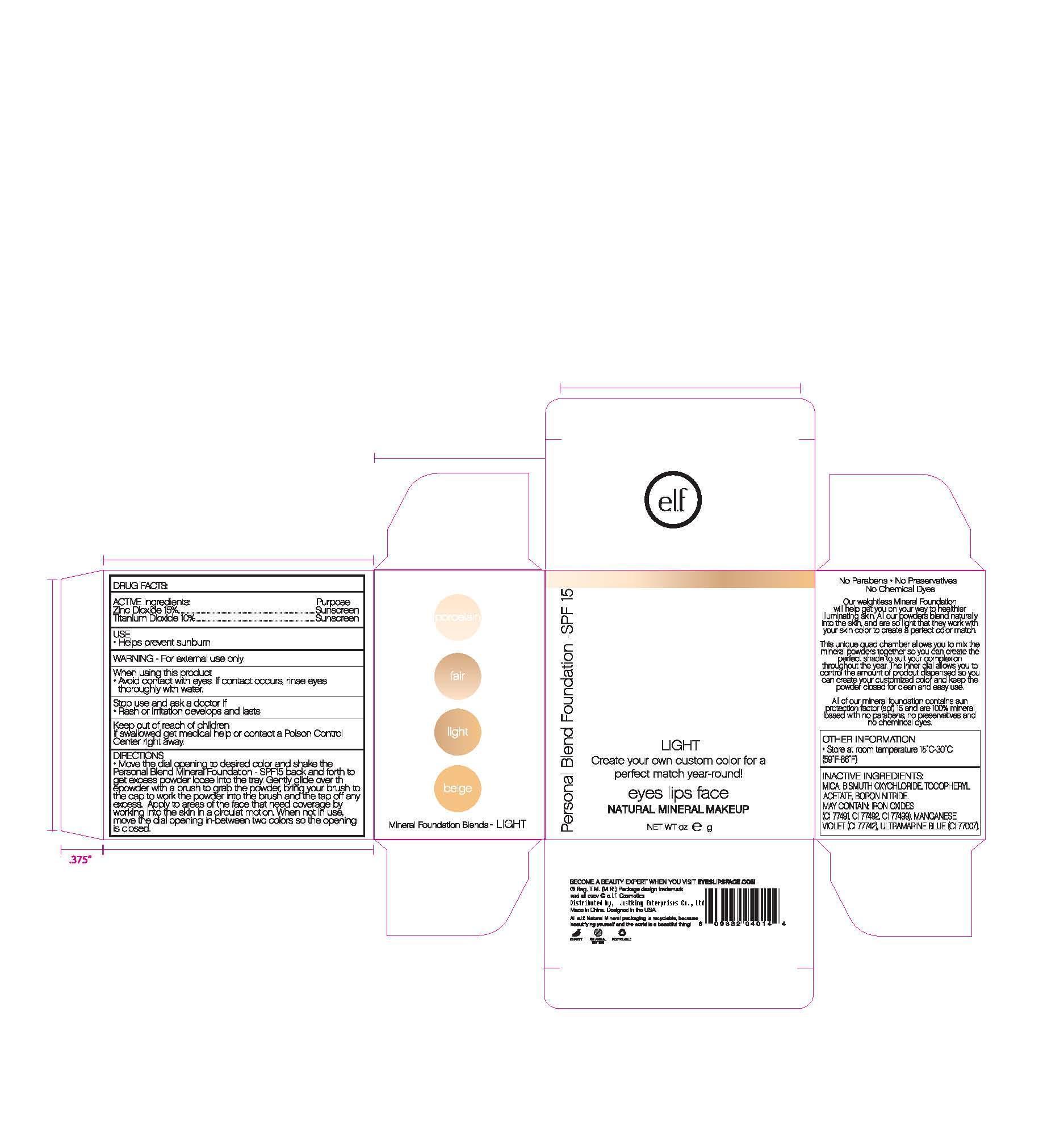
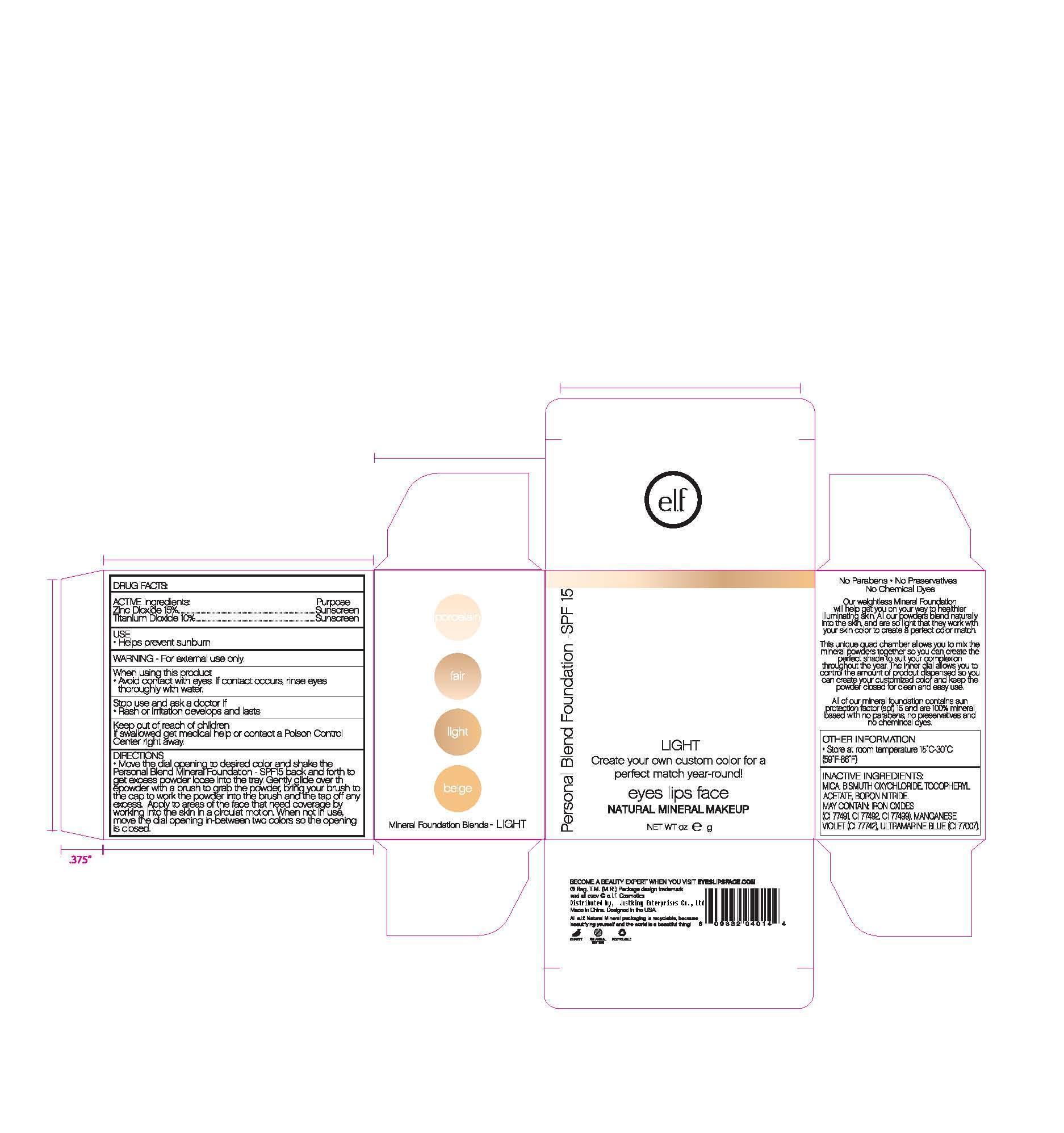
Move the dial opening to desired color and shake the Personal Blend Foundation SPF 15 back and forth to get excess powder loose onto the tray. Gently glide over the powder with a brush to grab the powder, bring your brush to the cap to work in the powder to the brush, then tap off any excess. Apply to the areas of the face that need coverage by working into the skin in a circular motion. When not in use, move the dial opening in between two colors so the dial opening is closed.
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ELF PERSONAL BLEND FOUNDATION SPF 15
titanium dioxide powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76166-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 10 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 15 g in 100 g Inactive Ingredients Ingredient Name Strength BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) MICA (UNII: V8A1AW0880) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76166-001-01 2.8 g in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 05/01/2011 Labeler - Shanghai Justking Enterprise Co. Ltd. (421267459) Establishment Name Address ID/FEI Business Operations Shanghai Justking Enterprise Co. Ltd. 421267459 manufacture