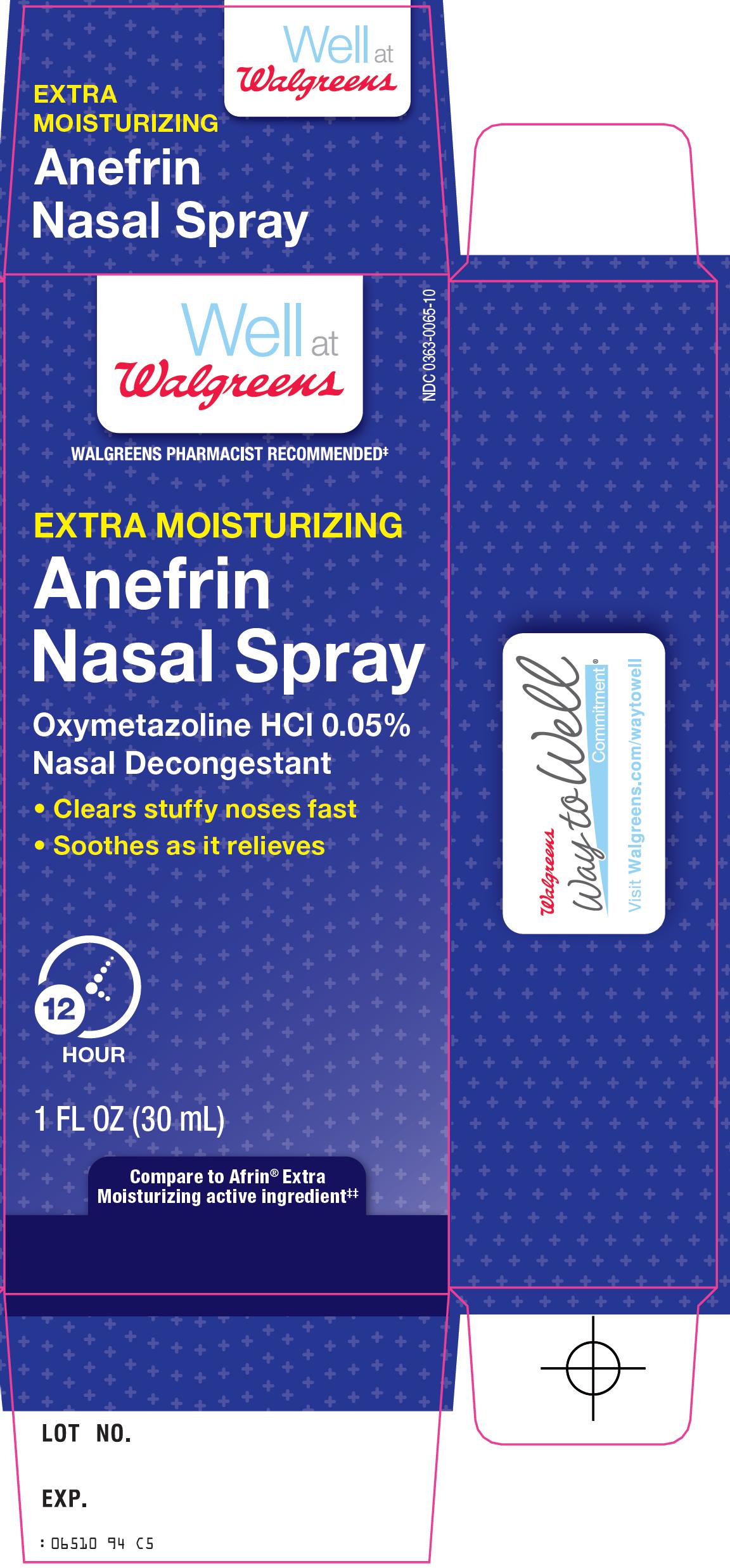
ANEFRIN NASAL EXTRA MOISTURIZING- oxymetazoline hcl spray
Walgreen Company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Walgreen Co. Anefrin Nasal Spray Drug Facts
Uses
- •
- temporarily relieves nasal congestion due to:
- •
- common cold
- •
- hay fever
- •
- upper respiratory allergies
- •
- shrinks swollen nasal membranes so you can breathe more freely
Warnings
Ask a doctor before use if you have
- •
- heart disease
- •
- thyroid disease
- •
- high blood pressure
- •
- diabetes
- •
- trouble urinating due to an enlarged prostate gland
When using this product
- •
- do not use more than directed
- •
- do not use for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen.
- •
- temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
- •
- use of this container by more than one person may spread infection
Directions
- •
- adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period.
- •
- children under 6 years of age: ask a doctor
To spray, squeeze bottle quickly and firmly. Do not tilt head backward while spraying. Wipe nozzle clean after use.
Other information
- •
- store at 20°-25°C (68°-77°F)
- •
- retain carton for future reference on full labeling
| ANEFRIN NASAL
EXTRA MOISTURIZING
oxymetazoline hcl spray |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Walgreen Company (008965063) |
Revised: 11/2017
Document Id: f5da65dc-ba7e-4199-b32e-026b35f22b46
Set id: d6f5e093-997c-4a80-8db3-b3f82eff1b6e
Version: 3
Effective Time: 20171108
Walgreen Company