PYRIDOSTIGMINE BROMIDE - pyridostigmine bromide tablet
Eywa Pharma Inc
----------
Pyridostigmine Bromide Tablets ,USP
DESCRIPTION
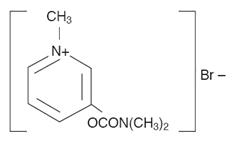
Pyridostigmine bromide tablets, USP (pyridostigmine bromide) are an orally active cholinesterase inhibitor. Chemically, pyridostigmine bromide is 3-hydroxy-1-methylpyridinium bromide dimethylcarbamate. Its structural formula is:
Pyridostigmine bromide tablets,USP are available in the following form:
Tablets containing 60 mg pyridostigmine bromide; each tablet also contains anhydrous lactose, colloidal silicon dioxide and stearic acid.
CLINICAL PHARMACOLOGY
Pyridostigmine bromide tablets inhibit the destruction of acetylcholine by cholinesterase and thereby permits freer transmission of nerve impulses across the neuromuscular junction. Pyridostigmine is an analog of neostigmine (ProstigminTM), but differs from it in certain clinically significant respects; for example, pyridostigmine is characterized by a longer duration of action and fewer gastrointestinal side effects.
INDICATIONS AND USAGE
Pyridostigmine bromide tablets are useful in the treatment of myasthenia gravis.
CONTRAINDICATIONS
Pyridostigmine bromide tablets are contraindicated in mechanical intestinal or urinary obstruction, and particular caution should be used in its administration to patients with bronchial asthma. Care should be observed in the use of atropine for counteracting side effects, as discussed below.
WARNINGS
Although failure of patients to show clinical improvement may reflect under dosage, it can also be indicative of overdosage. As is true of all cholinergic drugs, overdosage of pyridostigmine bromide may result in cholinergic crisis, a state characterized by increasing muscle weakness which, through involvement of the muscles of respiration, may lead to death. Myasthenic crisis due to an increase in the severity of the disease is also accompanied by extreme muscle weakness, and thus may be difficult to distinguish from cholinergic crisis on a symptomatic basis. Such differentiation is extremely important, since increases in doses of pyridostigmine bromide or other drugs of this class in the presence of cholinergic crisis or of a refractory or "insensitive" state could have grave consequences. Osserman and Genkins1 indicate that the differential diagnosis of the two types of crisis may require the use of TensilonTM (edrophonium chloride) as well as clinical judgment. The treatment of the two conditions obviously differs radically. Whereas the presence of myasthenic crisis suggests the need for more intensive anticholinesterase therapy, the diagnosis of cholinergic crisis, according to Osserman and Genkins1, calls for the prompt withdrawal of all drugs of this type. The immediate use of atropine in cholinergic crisis is also recommended.
Atropine may also be used to abolish or obtund gastrointestinal side effects or other muscarinic reactions; but such use, by masking signs of overdosage, can lead to inadvertent induction of cholinergic crisis.
For detailed information on the management of patients with myasthenia gravis, the physician is referred to one of the excellent reviews such as those by Osserman and Genkins2, Grob3 or Schwab4,5
Usage in Pregnancy
The safety of pyridostigmine bromide tablets during pregnancy or lactation in humans has not been established. Therefore, use of pyridostigmine bromide tablets in women who may become pregnant requires weighing the drug's potential benefits against its possible hazards to mother and child.
PRECAUTIONS
Pyridostigmine is mainly excreted unchanged by the kidney.6,7,8 Therefore, lower doses may be required in patients with renal disease, and treatment should be based on titration of drug dosage to effect.6,7
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
ADVERSE REACTIONS
The side effects of pyridostigmine bromide tablets are most commonly related to overdosage and generally are of two varieties, muscarinic and nicotinic. Among those in the former group are nausea, vomiting, diarrhea, abdominal cramps, increased peristalsis, increased salivation, increased bronchial secretions, miosis and diaphoresis. Nicotinic side effects are comprised chiefly of muscle cramps, fasciculation and weakness. Muscarinic side effects can usually be counteracted by atropine, but for reasons shown in the preceding section the expedient is not without danger. As with any compound containing the bromide radical, a skin rash may be seen in an occasional patient. Such reactions usually subside promptly upon discontinuance of the medication.
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DOSAGE AND ADMINISTRATION
Pyridostigmine bromide tablets are available in following dosage form:
Conventional Tablets
each containing 60 mg pyridostigmine bromide.
Dosage
The size and frequency of the dosage must be adjusted to the needs of the individual patient.
Conventional Tablets
The average dose is ten 60 mg tablets, spaced to provide maximum relief when maximum strength is needed. In severe cases as many as 25 tablets a day may be required, while in mild cases one to six tablets a day may suffice.
NOTE: For information on a diagnostic test for myasthenia gravis, and for the evaluation and stabilization of therapy, please see product literature on Tensilon (edrophonium chloride).
HOW SUPPLIED
Tablets, are available as white, flat-faced tablets containing 60 mg pyridostigmine bromide in bottles of 30 (NDC 71930-028-30) and 90 (NDC 71930-028-90). Each tablet is white to off-white, round, flat-faced tablet debossed with "C 60" on one side and quadrisect scored on the other side.
Store at 25˚C (77˚F); excursions permitted between 15˚ to 30˚C (59˚ to 86˚F). [See USP Controlled Room Temperature].
Preserve in a tight, light-resistant container. Keep pyridostigmine bromide tablets, USP in a dry place with the silica gel enclosed.
REFERENCES
1. Osserman KE, Genkins G. Studies in myasthenia gravis: Reduction in mortality rate after crisis. JAMA. Jan 1963; 183:97-101.
2. Osserman KE, Genkins G. Studies in myasthenia gravis. NY State J Med. June 1961; 61:2076-2085.
3. Grob D. Myasthenia gravis. A review of pathogenesis and treatment. Arch Intern Med. Oct 1961;108:615-638.
4. Schwab RS. Management of myasthenia gravis. New Eng J Med. Mar 1963; 268:596-597.
5. Schwab RS. Management of myasthenia gravis. New Eng J Med. Mar 1963; 268:717-719.
6. Cronnelly R, Stanski DR, Miller RD, Sheiner LB. Pyridostigmine kinetics with and without renal function. Clin Pharmacol Ther. 1980; 28:No. 1,78-81.
7. Miller RD. Pharmacodynamics and pharmacokinetics of anticholinesterase. In: Ruegheimer E, Zindler M, ed. Anaesthesiology. (Hamburg, Germany: Congress; Sep 14- 21, 1980; 222-223.) (Int Congr. No. 538), Amsterdam, Netherlands: Excerpta Medica; 1981.
8. Breyer-Pfaff U, Maier U, Brinkmann AM, Schumm F. Pyridostigmine kinetics in healthy subjects and patients with myasthenia gravis. Clin Pharmacol Ther. 1985; 5:495- 501.
All Product/Brand names are the trademarks of their respective owners.
Manufactured by:
Centaur Pharmaceuticals Private Limited
Plot no. 4, International Biotech Park,
Phase-II, Hinjewadi, Pune,
Maharashtra 411057, India
Distributed by:
Eywa Pharma Inc
2 Research Way, Floor 3
Princeton, NJ 08540
Revised: 02/2020
| PYRIDOSTIGMINE BROMIDE
pyridostigmine bromide tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Eywa Pharma Inc (080465609) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Centaur Pharmaceuticals Private Limited | 675596622 | ANALYSIS(71930-028) , MANUFACTURE(71930-028) , PACK(71930-028) | |
