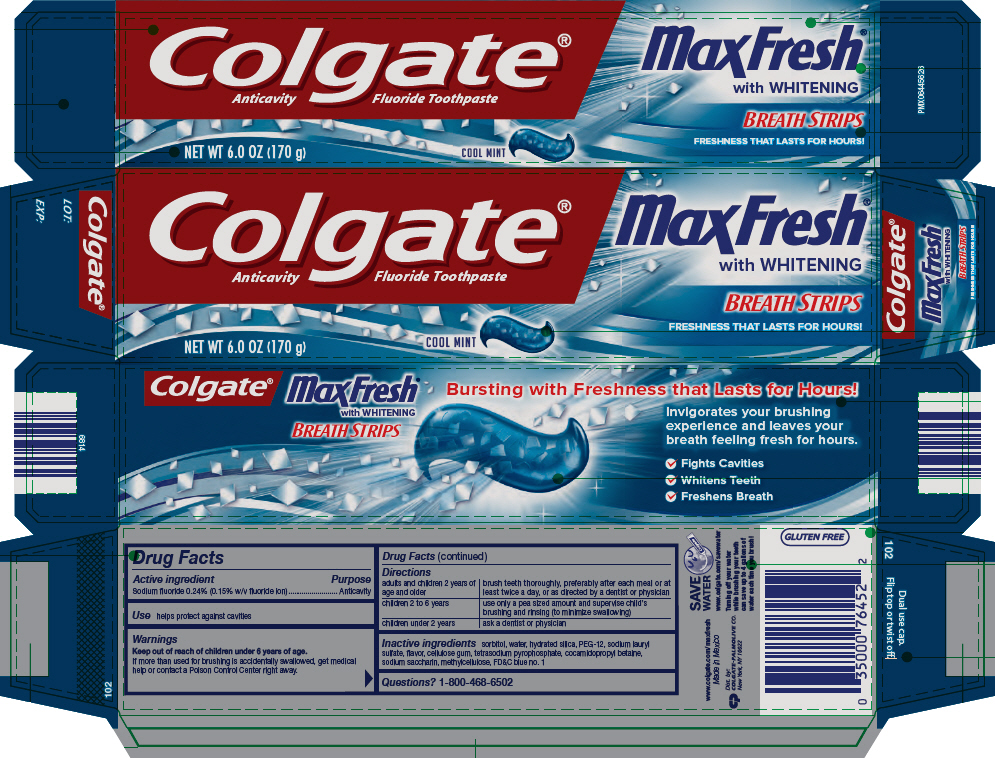
COLGATE MAX FRESH COOL MINT- sodium fluoride gel, dentifrice
Mission Hills S.A de C.V
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Colgate® Max Fresh® Cool Mint Fluoride Toothpaste
Directions
| adults and children 2 years of age and older | brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician |
| children 2 to 6 years | use only a pea sized amount and supervise child's brushing and rinsing (to minimize swallowing) |
| children under 2 years | ask a dentist or physician |
| COLGATE
MAX FRESH COOL MINT
sodium fluoride gel, dentifrice |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Mission Hills S.A de C.V (812312122) |
Revised: 4/2019
Document Id: 4093df3a-15d5-4455-b9ef-9f70b45257c0
Set id: d142e43f-415a-41c5-a7db-8b18a039a323
Version: 8
Effective Time: 20190409
Mission Hills S.A de C.V