POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE- dextrose monohydrate, sodium chloride, and potassium chloride injection, solution
Hospira, Inc.
----------
INTRAVENOUS SOLUTIONS
WITH POTASSIUM CHLORIDE
Rx only
Potassium Chloride in 5% Dextrose and Sodium Chloride Injection, USP
Flexible Plastic Container
DESCRIPTION
Intravenous solutions with potassium chloride (I.V. solutions with KCl) are sterile and nonpyrogenic solutions in water for injection. They are for administration by intravenous infusion only.
See Tables for summary of content and characteristics of these solutions.
The solutions contain no bacteriostat, antimicrobial agent or added buffer and each is intended only for use as a single-dose injection. When smaller doses are required the unused portion should be discarded.
These solutions are parenteral fluid, nutrient and/or electrolyte replenishers.
Dextrose, USP is chemically designated D-glucose, monohydrate (C6H12O6 • H2O), a hexose sugar freely soluble in water. It has the following structural formula:
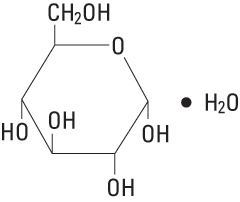
Potassium Chloride, USP is chemically designated KCl, a white granular powder freely soluble in water.
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials. Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in the moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
CLINICAL PHARMACOLOGY
When administered intravenously, these solutions provide a source of water and potassium chloride with carbohydrate (dextrose) and sodium chloride. See HOW SUPPLIED section for specific concentrations of these various solutions.
Solutions containing carbohydrate in the form of dextrose restore blood glucose levels and provide calories. Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein-sparing action. Dextrose injected parenterally undergoes oxidation to carbon dioxide and water.
Intravenous solutions containing potassium chloride are particularly intended to provide needed potassium cation (K+). Potassium is the chief cation of body cells (160 mEq/liter of intracellular water). It is found in low concentration in plasma and extracellular fluids (3.5 to 5.0 mEq/liter in a healthy adult). Potassium plays an important role in electrolyte balance. Normally about 80 to 90% of the potassium intake is excreted in the urine; the remainder in the stools and to a small extent, in the perspiration. The kidney does not conserve potassium well so that during fasting or in patients on a potassium-free diet, potassium loss from the body continues resulting in potassium depletion. A deficiency of either potassium or chloride will lead to a deficit of the other.
Sodium chloride in water dissociates to provide sodium (Na+) and chloride (Cl−) ions. Sodium (Na+) is the principal cation of the extracellular fluid and plays a large part in the therapy of fluid and electrolyte disturbances. Chloride (Cl−) has an integral role in buffering action when oxygen and carbon dioxide exchange occurs in the red blood cells. The distribution and excretion of sodium (Na+) and chloride (Cl−) are largely under the control of the kidney which maintains a balance between intake and output.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
INDICATIONS AND USAGE
These solutions are indicated in patients requiring parenteral administration of potassium chloride with minimal carbohydrate calories and sodium chloride.
CONTRAINDICATIONS
Solutions containing potassium chloride are contraindicated in diseases where high potassium levels may be encountered.
WARNINGS
Solutions which contain potassium ions should be used with great care, if at all, in patients with hyperkalemia, severe renal failure and in conditions in which potassium retention is present.
To avoid potassium intoxication, do not infuse these solutions rapidly. In patients with severe renal insufficiency or adrenal insufficiency, administration of potassium chloride may cause potassium intoxication.
Solutions containing sodium ions should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency and in clinical states in which there exists edema with sodium retention.
In patients with diminished renal function, administration of solutions containing sodium or potassium ions may result in sodium or potassium retention.
The intravenous administration of these solutions can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema.
The risk of dilutional states is inversely proportional to the electrolyte concentration of administered parenteral solutions. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of such solutions.
PRECAUTIONS
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
Solutions containing dextrose should be used with caution in patients with known subclinical or overt diabetes mellitus.
Caution must be exercised in the administration of parenteral fluids, especially those containing sodium ions, to patients receiving corticosteroids or corticotropin.
Potassium replacement therapy should be guided primarily by serial electrocardiograms. Plasma potassium levels are not necessarily indicative of tissue potassium levels.
High plasma concentrations of potassium may cause death through cardiac depression, arrhythmias or arrest.
Potassium-containing solutions should be used with caution in the presence of cardiac disease, particularly in digitalized patients or in the presence of renal disease.
Care should be exercised to insure that the needle (or catheter) is well within the lumen of the vein and that extravasation does not occur.
Do not administer unless solution is clear and container is undamaged. Discard unused portion.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Studies with solutions from flexible plastic containers have not been performed to evaluate carcinogenic potential, mutagenic potential or effects on fertility.
Pregnancy Category C. Animal reproduction studies have not been conducted with dextrose, potassium chloride or sodium chloride. It is also not known whether dextrose, potassium chloride or sodium chloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Dextrose, potassium chloride or sodium chloride should be given to a pregnant woman only if clearly needed.
Nursing Mothers:
Caution should be exercised when solutions from flexible plastic containers are administered to a nursing mother.
Pediatric Use:
The safety and effectiveness in the pediatric population are based on the similarity of the clinical conditions of the pediatric and adult populations. In neonates or very small infants the volume of fluid may affect fluid and electrolyte balance.
Frequent monitoring of serum glucose concentrations is required when dextrose is prescribed to pediatric patients, particularly neonates and low birth weight infants.
In very low birth weight infants, excessive or rapid administration of dextrose injection may result in increased serum osmolality and possible intracerebral hemorrhage.
Geriatric Use:
An evaluation of current literature revealed no clinical experience identifying differences in response between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. Sodium and potassium ions are known to be substantially excreted by the kidney, and the risk of toxic reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
ADVERSE REACTIONS
Reactions which may occur because of the solutions or technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
Nausea, vomiting, abdominal pain and diarrhea have been reported with potassium therapy. The signs and symptoms of potassium intoxication include paresthesias of the extremities, flaccid paralysis, listlessness, mental confusion, weakness and heaviness of the legs, hypotension, cardiac arrhythmias, heart block, electrocardiographic abnormalities such as disappearance of P waves, spreading and slurring of the QRS complex with development of a biphasic curve and cardiac arrest.
Potassium-containing solutions are intrinsically irritating to tissues. Therefore, extreme care should be taken to avoid perivascular infiltration. Local tissue necrosis and subsequent sloughing may result if extravasation occurs. Chemical phlebitis and venospasm have also been reported.
Should perivascular infiltration occur, I.V. administration at that site should be discontinued at once. Local infiltration of the affected area with procaine hydrochloride, 1%, to which hyaluronidase may be added, will often reduce venospasm and dilute the potassium remaining in the tissues locally. Local application of heat may also be helpful.
OVERDOSAGE
In the event of potassium overdosage, discontinue the infusion immediately and institute intensive corrective therapy to reduce serum potassium levels. See WARNINGS and PRECAUTIONS.
DOSAGE AND ADMINISTRATION
These solutions should be administered only by intravenous infusion and as directed by the physician. The dose and rate of injection are dependent upon the age, weight and clinical condition of the patient. If the serum potassium level is greater than 2.5 mEq/liter, potassium should be given at a rate not to exceed 10 mEq/hour in a concentration less than 30 mEq/liter. Somewhat faster rates and greater concentrations (usually up to 40 mEq/liter) of potassium may be indicated in patients with more severe potassium deficiency. The total 24-hour dose should not generally exceed 200 mEq of potassium.
As reported in the literature, the dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low birth weight infants, because of the increased risk of hyperglycemia/hypoglycemia.
Drug Interactions
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See PRECAUTIONS.
INSTRUCTIONS FOR USE
To Open
Tear outer wrap at notch and remove solution container. If supplemental medication is desired, follow directions below before preparing for administration. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Add Medication
- Prepare additive port.
- Using aseptic technique and an additive delivery needle of appropriate length, puncture resealable additive port at target area, inner diaphragm and inject. Withdraw needle after injecting medication.
- The additive port may be protected by covering with an additive cap.
- Mix container contents thoroughly.
Preparation for Administration
(Use aseptic technique)
- Close flow control clamp of administration set.
- Remove cover from outlet port at bottom of container.
- Insert piercing pin of administration set into port with a twisting motion until the set is firmly seated. NOTE: See full directions on administration set carton.
- Suspend container from hanger.
- Squeeze and release drip chamber to establish proper fluid level in chamber.
- Open flow control clamp and clear air from set. Close clamp.
- Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
- Regulate rate of administration with flow control clamp.
WARNING: Do not use flexible container in series connections.
HOW SUPPLIED
Intravenous solutions with potassium chloride (I.V. solution with KCl) are supplied in single-dose flexible plastic containers. See Tables:
|
TABLE 1 |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Potassium Chloride in 5% Dextrose and 0.225% Sodium Chloride Inj., USP |
COMPOSITION (g/L) |
Approx. Ionic Concentrations (mEq/L) | |||||||||
|
mEq Potassium |
Size (mL) |
Dextrose, Hydrous |
Sodium Chloride |
Potassium Chloride |
Calculated Osmolarity (mOsmol/L) |
pH |
Sodium (Na+) |
Potassium (K+) |
Chloride (Cl–) |
Approximate kcal/L |
NDC NO. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 mEq |
1000 |
50 |
2.25 |
0.745 |
349 |
4.2 (3.5 to 6.5) |
38.5 |
10 |
48.5 |
170 |
0409-7997-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 mEq |
500 |
50 |
2.25 |
1.49 |
370 |
4.2 (3.5 to 6.5) |
38.5 |
20 |
58.5 |
170 |
0409-7901-03 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20 mEq |
1000 |
50 |
2.25 |
1.49 |
370 |
4.2 (3.5 to 6.5) |
38.5 |
20 |
58.5 |
170 |
0409-7901-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
30 mEq |
1000 |
50 |
2.25 |
2.24 |
389 |
4.2 (3.5 to 6.5) |
38.5 |
30 |
68.5 |
170 |
0409-7991-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
40 mEq |
1000 |
50 |
2.25 |
2.98 |
409 |
4.2 (3.5 to 6.5) |
38.5 |
40 |
78.5 |
170 |
0409-7992-09 |
|
TABLE 2 |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Potassium Chloride in 5% Dextrose and 0.3% Sodium Chloride Inj., USP |
COMPOSITION (g/L) |
Approx. Ionic Concentrations (mEq/L) | |||||||||
|
mEq Potassium |
Size (mL) |
Dextrose, Hydrous |
Sodium Chloride |
Potassium Chloride |
Calculated Osmolarity (mOsmol/L) |
pH |
Sodium (Na+) |
Potassium (K+) |
Chloride (Cl–) |
Approximate kcal/L |
NDC NO. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 mEq |
500 |
50 |
3.0 |
1.49 |
395 |
4.2 (3.5 to 6.5) |
51 |
20 |
71 |
170 |
0409-7998-03 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20 mEq |
1000 |
50 |
3.0 |
1.49 |
395 |
4.2 (3.5 to 6.5) |
51 |
20 |
71 |
170 |
0409-7998-09 |
|
TABLE 3 |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Potassium Chloride in 5% Dextrose and 0.45% Sodium Chloride Inj., USP |
COMPOSITION (g/L) |
Approx. Ionic Concentrations (mEq/L) | |||||||||
|
mEq Potassium |
Size (mL) |
Dextrose, Hydrous |
Sodium Chloride |
Potassium Chloride |
Calculated Osmolarity (mOsmol/L) |
pH |
Sodium (Na+) |
Potassium (K+) |
Chloride (Cl–) |
Approximate kcal/L |
NDC NO. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 mEq |
1000 |
50 |
4.5 |
0.745 |
426 |
4.2 (3.5 to 6.5) |
77 |
10 |
87 |
170 |
0409-7993-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10 mEq |
500 |
50 |
4.5 |
1.49 |
447 |
4.2 (3.5 to 6.5) |
77 |
20 |
97 |
170 |
0409-7902-03 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20 mEq |
1000 |
50 |
4.5 |
1.49 |
447 |
4.2 (3.5 to 6.5) |
77 |
20 |
97 |
170 |
0409-7902-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
30 mEq |
1000 |
50 |
4.5 |
2.24 |
467 |
4.2 (3.5 to 6.5) |
77 |
30 |
107 |
170 |
0409-7903-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
40 mEq |
1000 |
50 |
4.5 |
2.98 |
487 |
4.2 (3.5 to 6.5) |
77 |
40 |
117 |
170 |
0409-7904-09 |
|
TABLE 4 |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Potassium Chloride in 5% Dextrose and 0.9% Sodium Chloride Inj., USP |
COMPOSITION (g/L) |
Approx. Ionic Concentrations (mEq/L) | |||||||||
|
mEq Potassium |
Size (mL) |
Dextrose, Hydrous |
Sodium Chloride |
Potassium Chloride |
Calculated Osmolarity (mOsmol/L) |
pH |
Sodium (Na+) |
Potassium (K+) |
Chloride (Cl–) |
Approximate kcal/L |
NDC NO. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20 mEq |
1000 |
50 |
9 |
1.49 |
600 |
4.2 (3.5 to 6.5) |
154 |
20 |
174 |
170 |
0409-7107-09 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
40 mEq |
1000 |
50 |
9 |
2.98 |
640 |
4.2 (3.5 to 6.5) |
154 |
40 |
194 |
170 |
0409-7109-09 |
May contain HCl for pH adjustment.
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Protect from freezing.
Revised: April, 2009
Printed in USA EN-2100
Hospira, Inc., Lake Forest, IL 60045 USA
PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0031
20 mEq POTASSIUM
1000 mL
NDC 0409-7107-09
20
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.9% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM
CHLORIDE 149 mg; SODIUM CHLORIDE 900 mg;
DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH
ADJUSTMENT. ELECTROLYTES PER 1000 mL
(NOT INCLUDING IONS FOR pH ADJUSTMENT):
POTASSIUM 20 mEq; SODIUM 154 mEq;
CHLORIDE 174 mEq.
600 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF
AVAILABLE. WHEN INTRODUCING
ADDITIVES, USE ASEPTIC TECHNIQUE,
MIX THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
Rx only
3
v
CONTAINS DEHP
©HOSPIRA 2004
IM-0031 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0034
40 mEq POTASSIUM
1000 mL
NDC 0409-7109-09
40
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.9% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM
CHLORIDE 298 mg; SODIUM CHLORIDE 900 mg;
DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH
ADJUSTMENT. ELECTROLYTES PER 1000 mL
(NOT INCLUDING IONS FOR pH ADJUSTMENT):
POTASSIUM 40 mEq; SODIUM 154 mEq;
CHLORIDE 194 mEq.
640 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF
AVAILABLE. WHEN INTRODUCING
ADDITIVES, USE ASEPTIC TECHNIQUE,
MIX THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
©HOSPIRA 2004
IM-0034 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0086
30 mEq POTASSIUM
1000 mL
NDC 0409-7903-09
30
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.45% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM
CHLORIDE 224 mg; SODIUM CHLORIDE 450 mg;
DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH
ADJUSTMENT. ELECTROLYTES PER 1000 mL
(NOT INCLUDING IONS FOR pH ADJUSTMENT):
POTASSIUM 30 mEq; SODIUM 77 mEq;
CHLORIDE 107 mEq.
467 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF
AVAILABLE. WHEN INTRODUCING
ADDITIVES, USE ASEPTIC TECHNIQUE,
MIX THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
©HOSPIRA 2004
IM-0086 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0087
40 mEq POTASSIUM
1000 mL
NDC 0409-7904-09
40
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.45% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM
CHLORIDE 298 mg; SODIUM CHLORIDE 450 mg;
DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH
ADJUSTMENT. ELECTROLYTES PER 1000 mL
(NOT INCLUDING IONS FOR pH ADJUSTMENT):
POTASSIUM 40 mEq; SODIUM 77 mEq;
CHLORIDE 117 mEq.
487 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF
AVAILABLE. WHEN INTRODUCING
ADDITIVES, USE ASEPTIC TECHNIQUE,
MIX THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
©HOSPIRA 2004
IM-0087 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0088
30 mEq POTASSIUM
1000 mL
NDC 0409-7991-09
30
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.225% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM CHLORIDE
224 mg; SODIUM CHLORIDE 225 mg; DEXTROSE,
HYDROUS 5 g IN WATER FOR INJECTION. MAY
CONTAIN HCl FOR pH ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING
IONS FOR pH ADJUSTMENT): POTASSIUM
30 mEq; SODIUM 38.5 mEq; CHLORIDE 68.5 mEq.
389 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF AVAILABLE.
WHEN INTRODUCING ADDITIVES,
USE ASEPTIC TECHNIQUE, MIX THOROUGHLY
AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS CLEAR
AND CONTAINER IS UNDAMAGED. MUST NOT BE
USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
©HOSPIRA 2004
IM-0088 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0089
40 mEq POTASSIUM
1000 mL
NDC 0409-7992-09
40
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.225% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM CHLORIDE
298 mg; SODIUM CHLORIDE 225 mg; DEXTROSE,
HYDROUS 5 g IN WATER FOR INJECTION. MAY
CONTAIN HCl FOR pH ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING
IONS FOR pH ADJUSTMENT): POTASSIUM
40 mEq; SODIUM 38.5 mEq; CHLORIDE 78.5 mEq.
409 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF AVAILABLE.
WHEN INTRODUCING ADDITIVES,
USE ASEPTIC TECHNIQUE, MIX THOROUGHLY
AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS CLEAR
AND CONTAINER IS UNDAMAGED. MUST NOT BE
USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
©HOSPIRA 2004
IM-0089 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0090
10 mEq POTASSIUM
1000 mL
NDC 0409-7993-09
10
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.45% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM
CHLORIDE 74.5 mg; SODIUM CHLORIDE 450 mg;
DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH
ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING
IONS FOR pH ADJUSTMENT): POTASSIUM
10 mEq; SODIUM 77 mEq; CHLORIDE 87 mEq.
426 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF AVAILABLE.
WHEN INTRODUCING ADDITIVES,
USE ASEPTIC TECHNIQUE, MIX THOROUGHLY
AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS
DEHP
PRINTED IN USA
©HOSPIRA 2004
IM-0090 (4/04)
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - IM-0057
10 mEq POTASSIUM
500 mL
NDC 0409-7901-03
10
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.225% Sodium Chloride Inj., USP
EACH 100 mL CONTAINS POTASSIUM CHLORIDE 149 mg; SODIUM
CHLORIDE 225 mg; DEXTROSE, HYDROUS 5 g IN WATER FOR INJECTION.
MAY CONTAIN HCl FOR pH ADJUSTMENT. ELECTROLYTES PER 1000 mL
(NOT INCLUDING IONS FOR pH ADJUSTMENT): POTASSIUM 20 mEq;
SODIUM 38.5 mEq; CHLORIDE 58.5 mEq. 370 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE. CONSULT WITH
PHARMACIST, IF AVAILABLE. WHEN INTRODUCING
ADDITIVES, USE ASEPTIC TECHNIQUE, MIX
THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE. USUAL DOSAGE: SEE INSERT.
STERILE, NONPYROGENIC. USE ONLY IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED. MUST NOT BE USED IN SERIES
CONNECTIONS.
©HOSPIRA 2004
IM-0057 (4/04)
PRINTED IN USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
3
v
CONTAINS DEHP
Rx ONLY
Hospira

PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - IM-0096
10 mEq POTASSIUM
500 mL
NDC 0409-7998-03
10
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.3% Sodium Chloride Inj., USP
EACH 100 mL CONTAINS POTASSIUM CHLORIDE 149 mg; SODIUM
CHLORIDE 300 mg; DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING IONS FOR pH
ADJUSTMENT): POTASSIUM 20 mEq; SODIUM 51 mEq; CHLORIDE 71
mEq. 395 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF AVAILABLE.
WHEN INTRODUCING ADDITIVES, USE ASEPTIC TECHNIQUE, MIX
THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE. USUAL DOSAGE: SEE
INSERT. STERILE, NONPYROGENIC. USE
ONLY IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED. MUST NOT
BE USED IN SERIES CONNECTIONS.
3
v
CONTAINS DEHP
PRINTED IN USA
Rx ONLY
©HOSPIRA 2004
IM-0096 (4/04)
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - IM-0065
10 mEq POTASSIUM
500 mL
NDC 0409-7902-03
10
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.45% Sodium Chloride Inj., USP
EACH 100 mL CONTAINS POTASSIUM CHLORIDE 149 mg; SODIUM
CHLORIDE 450 mg; DEXTROSE, HYDROUS 5 g IN WATER
FOR INJECTION. MAY CONTAIN HCl FOR pH ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING IONS FOR pH
ADJUSTMENT): POTASSIUM 20 mEq; SODIUM 77 mEq; CHLORIDE 97 mEq.
447 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE. CONSULT WITH
PHARMACIST, IF AVAILABLE. WHEN INTRODUCING
ADDITIVES, USE ASEPTIC TECHNIQUE, MIX
THOROUGHLY AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE. USUAL DOSAGE: SEE INSERT.
STERILE, NONPYROGENIC. USE ONLY IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED. MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
PRINTED IN USA
©HOSPIRA 2004
IM-0065 (4/04)
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - IM-0091
10 mEq POTASSIUM
1000 mL
NDC 0409-7997-09
10
mEq
POTASSIUM
CHLORIDE
in 5% Dextrose and
0.225% Sodium Chloride
Injection, USP
EACH 100 mL CONTAINS POTASSIUM
CHLORIDE 74.5 mg; SODIUM CHLORIDE 225 mg;
DEXTROSE, HYDROUS 5 g IN WATER FOR
INJECTION. MAY CONTAIN HCl FOR pH
ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING
IONS FOR pH ADJUSTMENT): POTASSIUM
10 mEq; SODIUM 38.5 mEq; CHLORIDE 48.5 mEq.
349 mOsmol/LITER (CALC.)
pH 4.2 (3.5 to 6.5)
ADDITIVES MAY BE INCOMPATIBLE.
CONSULT WITH PHARMACIST, IF AVAILABLE.
WHEN INTRODUCING ADDITIVES,
USE ASEPTIC TECHNIQUE, MIX THOROUGHLY
AND DO NOT STORE.
SINGLE-DOSE CONTAINER. FOR I.V. USE.
USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.
Rx ONLY
3
v
CONTAINS DEHP
PRINTED IN USA
©HOSPIRA 2004
IM-0091 (4/04)
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Hospira

PRINCIPAL DISPLAY PANEL - Overwrap
TO OPEN TEAR AT NOTCH
2
HDPE
DO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER FIRMLY.
IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE IMPAIRED.
RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID EXCESSIVE
HEAT. PROTECT FROM FREEZING. SEE INSERT.
98-4321-R14-3/98

| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| POTASSIUM CHLORIDE IN DEXTROSE AND SODIUM CHLORIDE
dextrose monohydrate, sodium chloride, and potassium chloride injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 827731089 | ANALYSIS(0409-7107, 0409-7109, 0409-7901, 0409-7902, 0409-7903, 0409-7904, 0409-7991, 0409-7992, 0409-7993, 0409-7997, 0409-7998) | |