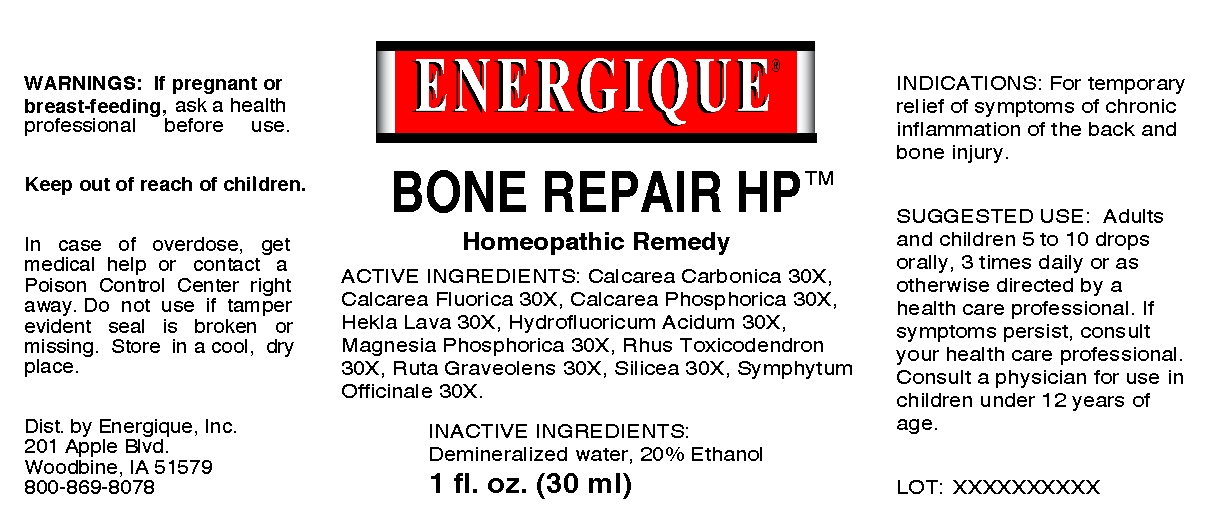
BONE REPAIR HP- calcarea carbonica, calcarea fluorica, calcarea phosphorica, hekla lava, hydrofluoricum acidum, magnesia phosphorica rhus toxicodendron, ruta graveolens, silicea, symphytum officinale liquid
Apotheca Company
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
Bone Repair HP
ACTIVE INGREDIENTS: Calcarea carbonica 30X, Calcarea fluorica 30X, Calcarea phosphorica 30X, Hekla lava 30X, Hydrofluoricum acidum 30X, Magnesia phosphorica 30X, Rhus toxicodendron 30X, Ruta graveolens 30X, Silicea 30X, Symphytum officinale 30X.
WARNINGS: If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away. Do not use if tamper evident seal is broken or missing. Store in a cool, dry place.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
| BONE REPAIR
HP
calcarea carbonica, calcarea fluorica, calcarea phosphorica, hekla lava, hydrofluoricum acidum, magnesia phosphorica rhus toxicodendron, ruta graveolens, silicea, symphytum officinale liquid |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Apotheca Company (844330915) |
| Registrant - Apotheca Company (844330915) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ApothecaCompany | 844330915 | manufacture(57520-0375) , api manufacture(57520-0375) , label(57520-0375) , pack(57520-0375) | |