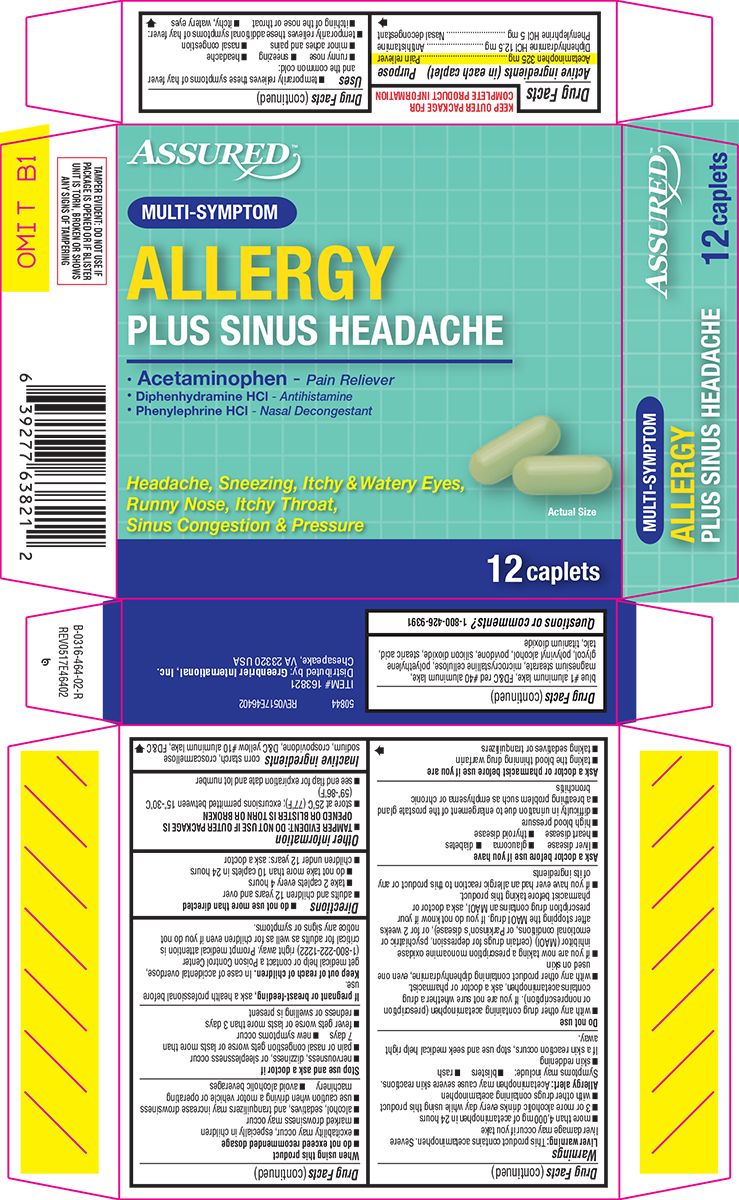
ALLERGY PLUS SINUS HEADACHE- acetaminophen, diphenhydramine hcl and phenylephrine hcl tablet, film coated
GREENBRIER INTERNATIONAL, INC.
----------
Assured 44-464 Delisted
Active ingredients (in each caplet)
Acetaminophen 325 mg
Diphenhydramine HCl 12.5 mg
Phenylephrine HCl 5 mg
Uses
- temporarily relieves these symptoms of hay fever and the common cold:
- runny nose
- sneezing
- headache
- minor aches and pains
- nasal congestion
- temporarily relieves these additional symptoms of hay fever:
- itching of the nose or throat
- itchy, watery eyes
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- 3 or more alcoholic drinks every day while using this product
- with other drugs containing acetaminophen
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- blisters
- rash
- skin reddening
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- with any other product containing diphenhydramine, even one used on skin
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- liver disease
- glaucoma
- diabetes
- heart disease
- thyroid disease
- high blood pressure
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- do not exceed recommended dosage
- excitability may occur, especially in children
- marked drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
- avoid alcoholic beverages
Directions
- do not use more than directed
- adults and children 12 years and over
- take 2 caplets every 4 hours
- do not take more than 10 caplets in 24 hours
- children under 12 years: ask a doctor
Other information
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, croscarmellose sodium, crospovidone, D&C yellow #10 aluminum lake, FD&C blue #1 aluminum lake, FD&C red #40 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, stearic acid, talc, titanium dioxide
Principal Display Panel
ASSURED™
MULTI-SYMPTOM
ALLERGY
PLUS SINUS HEADACHE
• Acetaminophen - Pain Reliever
• Diphenhydramine HCl - Antihistamine
• Phenylephrine HCl - Nasal Decongestant
Headache, Sneezing, Itchy & Watery Eyes,
Runny Nose, Itchy Throat,
Sinus Congestion & Pressure
Actual Size
12 caplets
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERING
50844 REV0517E46402
ITEM# 163821
Distributed by: Greenbrier International, Inc.
Chesapeake, VA 23320 USA
Assured 44-464
| ALLERGY PLUS SINUS HEADACHE
acetaminophen, diphenhydramine hcl and phenylephrine hcl tablet, film coated |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| Labeler - GREENBRIER INTERNATIONAL, INC. (610322518) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | pack(33992-0464) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | manufacture(33992-0464) | |