ALBUMINAR-25- albumin (human) solution
CSL Behring LLC
----------
Albumin (Human) USP, 25%
ALBUMINAR®-25
DESCRIPTION
Albumin (Human) 25%, ALBUMINAR®-25 is a sterile aqueous solution of albumin obtained from large pools of adult human venous plasma by low temperature controlled fractionation according to the Cohn process. It is stabilized with 0.02 M sodium acetyltryptophanate and 0.02 M sodium caprylate and pasteurized at 60°C for 10 hours.
All Source Plasma used in the manufacture of this product was tested by FDA-licensed Nucleic Acid Testing (NAT) for HBV, HCV, and HIV-1 and found to be nonreactive (negative).
ALBUMINAR®-25 is a solution containing in each 100 mL, 25 grams of serum albumin, osmotically equivalent to 500 mL of normal human plasma. The pH of the solution is adjusted with sodium bicarbonate, sodium hydroxide, or acetic acid. Approximate concentrations of significant electrolytes per liter are: sodium - 130-160 mEq; and potassium - n.m.t. 1 mEq. The solution contains no preservative. This product has been prepared in accordance with the requirements established by the Food and Drug Administration and is in compliance with the standards of the United States Pharmacopeia.
ALBUMINAR®-25 is to be administered by the intravenous route.
The heat treatment step employed in the manufacture of ALBUMINAR®-25 pasteurization of the final container at 60°C for 10 hours, has been validated in a series of in vitro experiments for its capacity to inactivate Human Immunodeficiency Virus type 1 (HIV-1), and the following model viruses: Bovine Viral Diarrhea Virus (BVDV - an enveloped virus used as a model for hepatitis C virus), Pseudorabies (PrV - a large, enveloped virus), and Encephalomyocarditis Virus (EMC - a small non-enveloped virus). For each virus studied, three independent experiments were conducted using Albumin (Human) 5%, ALBUMINAR®-5 and ALBUMINAR®-25 with the results provided in Table 1.1
| Pasteurization (60°C for 10 hours) Viral Reduction Studies (log10 reduction) |
|
| Virus | Albumin (Human) 5%, ALBUMINAR®-5 |
| HIV-1 | >5.44, >6.38 and >6.31 |
| BVDV | >6.01, >6.76 and >6.55 |
| PrV | >7.30, >7.68 and >7.63 |
| EMC | >7.38, >7.97 and >7.97 |
| Virus | Albumin (Human) 25%, ALBUMINAR®-25 |
| HIV-1 | >5.50, >6.57 and >6.64 |
| BVDV | >5.99, >5.81 and >5.32 |
| PrV | >7.32, >7.20 and >7.42 |
| EMC | >7.10, >7.89 and >7.87 |
CLINICAL PHARMACOLOGY
ALBUMINAR®-25 is active osmotically and is therefore important in regulating the volume of circulating blood. When injected intravenously, 50 mL of 25% albumin draws approximately 175 mL of additional fluid into the circulation within 15 minutes, except in the presence of marked dehydration. This extra fluid reduces hemoconcentration and blood viscosity. The degree of volume expansion is dependent on the initial blood volume. When the circulating blood volume has been depleted, the hemodilution following albumin administration persists for many hours. In individuals with normal blood volume, it usually lasts only a few hours.
Albumin, unlike whole blood or plasma, is considered free of the danger of homologous serum hepatitis. ALBUMINAR®-25 may be given in conjunction with other parenteral fluids such as saline, dextrose or sodium lactate. It is convenient to use since no cross-matching is required and the absence of cellular elements removes the danger of sensitization with repeated infusions.
INDICATIONS AND USAGE
SHOCK
Albumin is indicated in the emergency treatment of shock and in other similar conditions where the restoration of blood volume is urgent. If there has been considerable loss of red blood cells, transfusion with packed red blood cells is indicated.
BURNS
Albumin or Albumin in either normal saline or dextrose is indicated to prevent marked hemoconcentration and to maintain appropriate electrolyte balance.
HYPOPROTEINEMIA with or without edema
Albumin is indicated in those clinical situations usually associated with a low concentration of plasma protein and a resulting decreased circulating blood volume. Although diuresis may occur soon after albumin administration has been instituted, best results are obtained if albumin is continued until the normal serum protein level is regained.
CONTRAINDICATIONS
ALBUMINAR®-25 may be contraindicated in patients with severe anemia or cardiac failure and in patients with a history of allergic reactions to human albumin.
WARNINGS
Infusion of protein-containing solutions such as ALBUMINAR®-25 that have been excessively or inappropriately diluted with hypotonic solutions such as sterile water for injection may result in severe hemolysis and acute renal failure. Please refer to the DOSAGE AND ADMINISTRATION section for information about the recommended diluents for ALBUMINAR®-25, which are normal saline and 5% dextrose.
Do not use if the solution is turbid. Since this product contains no antimicrobial preservative, do not begin administration more than 4 hours after the container has been entered. Discard partially used vials.
ALBUMINAR®-25 is made from human plasma. Products made from human plasma may contain infectious agents such as viruses, that can cause disease. The risk that such products will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain viruses, by testing for the presence of certain current virus infections, and by inactivating and/or removing certain viruses during manufacture. The manufacturing procedure for ALBUMINAR®-25 includes processing steps designed to reduce further the risk of viral transmission. Stringent procedures utilized at plasma collection centers, plasma testing laboratories, and fractionation facilities are designed to reduce the risk of viral transmission. ALBUMINAR®-25 is pasteurized in the final container at 60.0 +/- 0.5°C for 10-11 hours. Virus elimination/inactivation is also achieved by the cold alcohol fractionation process. (See DESCRIPTION section for further information on viral reduction measures.) Despite these measures, such products may still potentially contain human pathogenic agents, including those not yet known or identified. Thus the risk of transmission of infectious agents cannot be totally eliminated. Any infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to CSL Behring at 1-866-915-6958. The physician should discuss the risks and benefits of this product with the patient.
Albumin is a derivative of human blood. Based on effective donor screening and product manufacturing processes, it carries an extremely remote risk for transmission of viral diseases. A theoretical risk for transmission of Creutzfeldt-Jakob disease (CJD) also is considered extremely remote. No cases of transmission of viral diseases or CJD have ever been identified for albumin.
PRECAUTIONS
GENERAL
If dehydration is present additional fluids must accompany or follow the administration of albumin. Administration of large quantities of albumin should be supplemented with or replaced by packed red blood cells to combat the relative anemia which would follow such use. The quick response of blood pressure which may follow the rapid administration of concentrated albumin necessitates careful observation of the injured patient to detect bleeding points which failed to bleed at lower blood pressure. ALBUMINAR®-25 should be administered with caution to patients with low cardiac reserve or with no albumin deficiency because a rapid increase in plasma volume may cause circulatory compromise (e.g. hypertension, hypotension, or pulmonary edema). In cases of hypertension, a slower rate of administration is desired - 200 mL of albumin solution may be mixed with 300 mL of 10% dextrose solution and administered at a rate of 10 grams of albumin (100 mL) per hour.
If anaphylactic or severe anaphylactoid reactions occur, discontinue infusion immediately. Infusion rates and the patient's clinical state should be monitored closely during infusion.
INFORMATION FOR PATIENT
Some viruses, such as parvovirus B19 or hepatitis A are particularly difficult to remove or inactivate at this time. Parvovirus B19 may most seriously affect pregnant women, or immune-compromised individuals. The majority of parvovirus B19 and hepatitis A infections are acquired by environmental (community acquired) sources.
ADVERSE REACTIONS
The incidence of untoward reactions to ALBUMINAR®-25 is low. Reports have been received of anaphylaxis, which may be severe, and hypersensitivity reactions (including urticaria, skin rash, pruritus, edema, erythema, hypotension and bronchospasm). Nausea, vomiting, increased salivation, chills and febrile reactions have also been reported (see also PRECAUTIONS).
DOSAGE AND ADMINISTRATION
ALBUMINAR®-25 may be given intravenously without dilution or it may be diluted with normal saline or 5% dextrose before administration. 200 mL per liter gives a solution which is approximately isotonic and iso-osmotic with citrated plasma.
When undiluted albumin solution is administered in patients with normal blood volume, the rate of infusion should be slow enough (1 mL per minute) to prevent too rapid expansion of plasma volume.
In the treatment of shock the amount of albumin and duration of therapy must be based on the responsiveness of the patient as indicated by blood pressure, degree of pulmonary congestion, and hematocrit. The initial dose may be followed by additional albumin within 15-30 minutes if the response is deemed inadequate. If there is continued loss of protein, it may be desirable to give packed red blood cells.
In the treatment of burns an optimal regimen involving use of albumin, crystalloids, electrolytes and water has not been established. Suggested therapy during the first 24 hours includes administration of large volumes of crystalloid solution to maintain an adequate plasma volume. Continuation of therapy beyond 24 hours usually requires more albumin and less crystalloid solution to prevent marked hemoconcentration and maintain electrolyte balance. Duration of treatment varies depending upon the extent of protein loss through renal excretion, denuded areas of skin and decreased albumin synthesis. Attempts to raise the albumin level above 4.0 g/100 mL may only result in an increased rate of catabolism.
In the treatment of hypoproteinemia, 200 to 300 mL of 25% albumin may be required to reduce edema and to bring serum protein values to normal. Since such patients usually have approximately normal blood volume, doses of more than 100 mL of 25% albumin should not be given faster than 100 mL in 30 to 45 minutes to avoid circulatory embarrassment. If slower administration is desired, 200 mL of 25% albumin may be mixed with 300 mL of 10% dextrose solution and administered by continuous drip at a rate of 100 mL of this dextrose solution an hour.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
HOW SUPPLIED
ALBUMINAR®-25 is supplied as a 25% solution. Each product presentation includes a package insert and the components listed in Table 2 below.
| Presentation | Carton NDC Number | Component |
|---|---|---|
| 20 mL | 0053-7680-01 | One single-dose vial containing 5 grams of albumin [NDC 0053-7680-90] |
| 50 mL | 0053-7680-32 | One single-dose vial containing 12.5 grams of albumin [NDC 0053-7680-91] |
| 100 mL | 0053-7680-33 | One single-dose vial containing 25 grams of albumin [NDC 0053-7680-92] |
BIBLIOGRAPHY
Finlayson, J.S.: Albumin Products. Seminars in Thrombosis and Hemostasis 6:85-120, 1980.
Tullis, J.L.: Albumin. JAMA 237: 355-360 and 460-463, 1977.
Rudolph, A.M.: Pediatrics. 18th ED., p. 1839, Appleton and Lange, 1987.
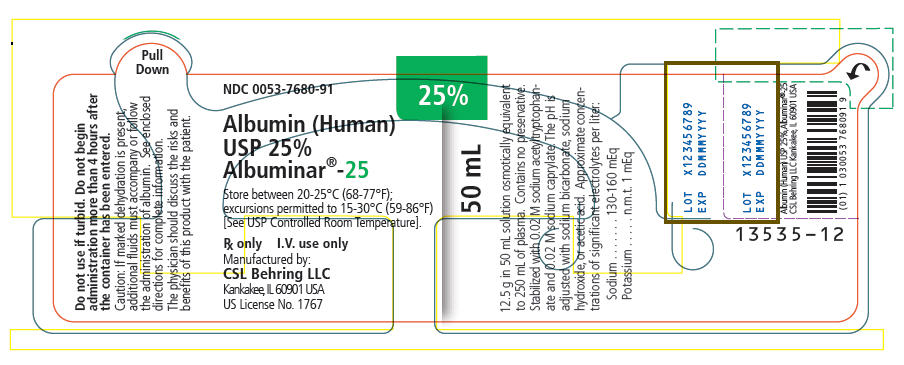
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label
NDC 0053-7680-91
25%
50 mL
Albumin (Human)
USP 25%
Albuminar®-25
Store between 20-25°C (68-77°F);
excursions permitted to 15-30°C (59-86°F)
[See USP Controlled Room Temperature].
Rx only
I.V. use only
Manufactured by:
CSL Behring LLC
Kankakee, IL 60901 USA
US License No. 1767
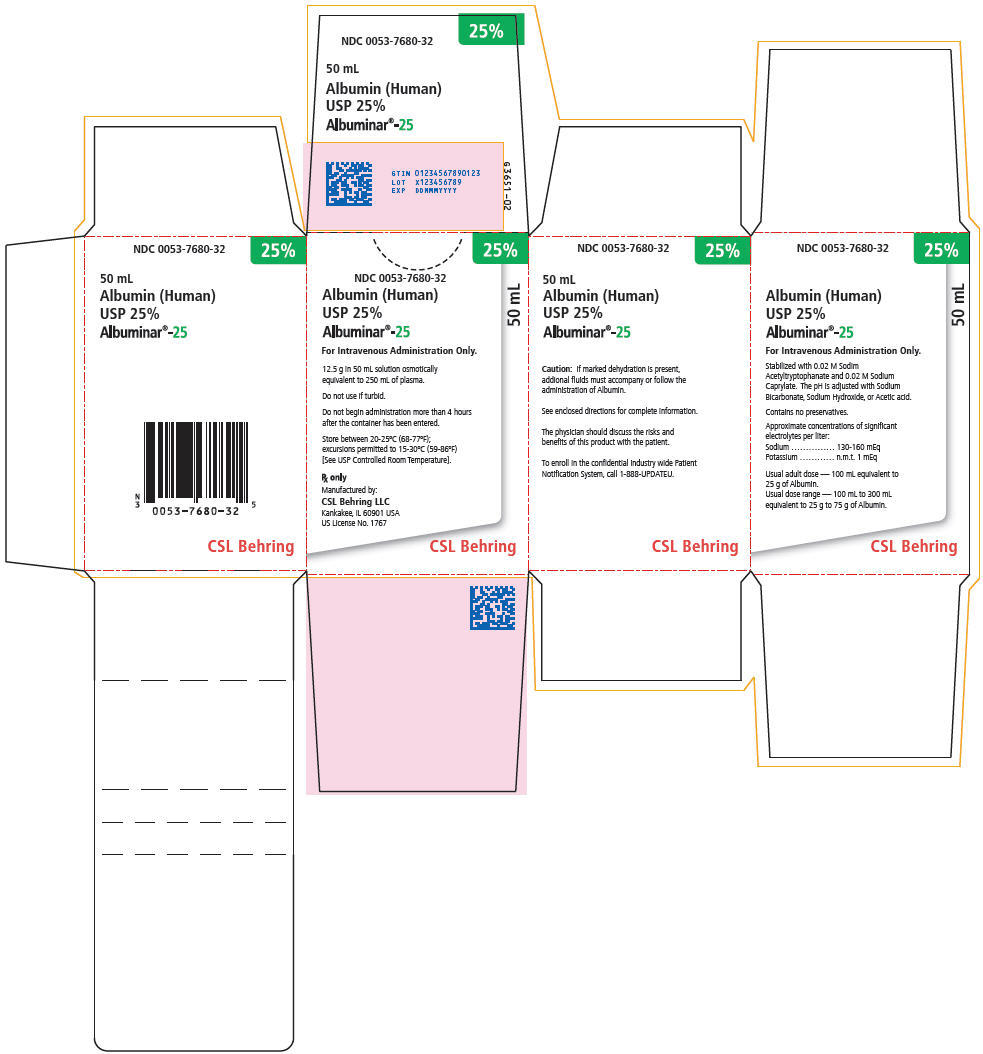
PRINCIPAL DISPLAY PANEL - 50 mL Vial Carton
25%
NDC 0053-7680-32
50 mL
Albumin (Human)
USP 25%
Albuminar®-25
For Intravenous Administration Only.
12.5 g in 50 mL solution osmotically
equivalent to 250 mL of plasma.
Do not use if turbid.
Do not begin administration more than 4 hours
after the container has been entered.
Store between 20-25°C (68-77°F);
excursions permitted to 15-30°C (59-86°F)
[See USP Controlled Room Temperature].
Rx only
Manufactured by:
CSL Behring LLC
Kankakee, IL 60901 USA
US License No. 1767
CSL Behring
| ALBUMINAR-25
albumin (human) solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - CSL Behring LLC (058268293) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| CSL Behring LLC | 058268293 | MANUFACTURE | |