FUNGI NAIL TOE AND FOOT- undecylenic acid liquid
Kramer Laboratories
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
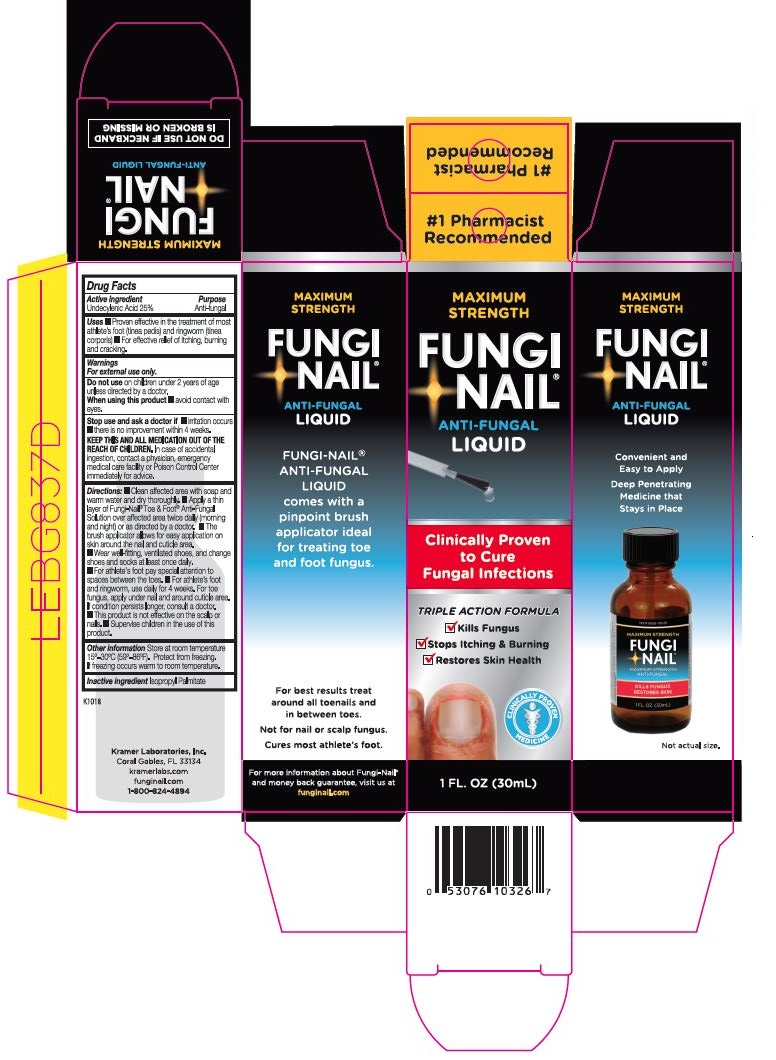
Drug Facts
Uses ■ Proven effective in the treatment of most athlete’s foot (tinea pedis) and ringworm (tinea corporis) ■ For effective relief of itching, burning and cracking.
Do not use on children under 2 years of age unless directed by a doctor.
When using this product ■ avoid contact with eyes.
KEEP THIS AND ALL MEDICATON OUT OF REACH OF CHILDREN. In case of accidental ingestion, contact a physician, emergency medical care facility or Poison Control Center immediately for advice.
Directions: ■ Clean affected area with soap and warm water and dry thoroughly. ■ Apply a thin layer of Fungi-Nail® Toe & Foot® Anti-Fungal Solution over affected area twice daily (morning and night) or as directed by a doctor. ■ The brush applicator allows for easy application on skin around the nail and cuticle area. ■ Wear well-fitting, ventilated shoes, and change shoes and socks at least once daily. ■ For athlete’s foot pay special attention to spaces between the toes. ■ For athlete’s foot and ringworm, use daily for 4 weeks. For toe fungus, apply under nail and around cuticle area. If condition persists longer, consult a doctor. ■ This product is not effective on the scalp or nails. ■ Supervise children in the use of this product.
Other information Store at controlled room temperature 15°-30°C (59°-86°F). Protect from freezing. If freezing occurs warm to room temperature.
PRINCIPAL DISPLAY PANEL
#1 Pharmacist Recommended
MAXIMUM STRENGTH
FUNGI-NAIL®
ANTI-FUNGAL
LIQUID
Clinically Proven to Cure Fungal Infections
TRIPLE ACTION FORMULA
✓ Kills Fungus
✓ Stops Itching & Burning
✓ Restores Skin Health
CLINICALLY PROVEN MEDICINE
1 FL. OZ (30 ML)
Convenient and Easy to Apply
Deep Penetrating Medicine that Stays in Place
Not actual size.
FUNGI-NAIL® ANTI FUNGAL LIQUID
comes with a pinpoint brush applicator ideal for treating toe and foot fungus.
For best results treat around all toenails and in between toes.
Not for nail or scalp fungus.
Cures most athlete's foot.
For more information about Fungi-Nail® and money back guarantee, visit us at funginail.com
K1018
Kramer Laboratories, Inc.
Coral Gables, FL 33134
kramerlabs.com
funginail.com
1-800-824-4894
NDC#: 55505-103-26
THE ORIGINAL
FUNGI-NAIL®
TOE & FOOT BRAND
MAXIMUM STRENGTH ANTI-FUNGAL
KILLS FUNGUS
RESTORES SKIN
1 FL. OZ. (30mL)
Distributed By:
KRAMER
LABORATORIES
Kramer Laboratories
Coral Gables, FL 33134 U.S.A.
kramerlabs.com
fungi-nail.com
1-800-824-4894
K0318

| FUNGI NAIL TOE AND FOOT
undecylenic acid liquid |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Kramer Laboratories (122720675) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Dension Pharmecuticals | 001207208 | manufacture(55505-103) | |