HYPERLYTE CR- multi electrolyte concentrate injection, solution, concentrate
B. Braun Medical Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Hyperlyte CR
Hyperlyte® CR
Multi-Electrolyte Concentrate (20 mL/dose)
PHARMACY BULK PACKAGE
NOT FOR DIRECT INFUSION
FOR INTRAVENOUS INFUSION ONLY. MUST BE DILUTED PRIOR TO INJECTION. Contains no phosphate.
DESCRIPTION
A pharmacy bulk package is a container of a sterile preparation for parenteral use that contains many single doses. The contents are intended for use in a pharmacy admixture service and are restricted to the preparation of admixtures for intravenous infusion.
Hyperlyte® CR (Multi-Electrolyte Concentrate) is a sterile, nonpyrogenic, concentrated solution of intra- and extracellular electrolytes, excluding phosphate. No bacteriostatic or antimicrobial agent has been added.
Each 20 mL contains: Potassium Acetate USP 1.96 g; Sodium Chloride USP 1.17 g; Sodium Acetate Trihydrate USP 0.68 g; Magnesium Chloride Hexahydrate USP 0.51 g; Calcium Chloride Dihydrate USP 0.37 g; Water for Injection USP qs
pH adjusted with Glacial Acetic Acid USP as needed
pH: 5.2 (5.0-5.4); Calculated Osmolarity: 5500 mOsmol/liter
Concentration of Electrolytes (mEq/20 mL dose): Sodium 25; Potassium 20; Calcium 5; Magnesium 5; Chloride 30; Acetate 30
The formulas of the active ingredients are:
| Ingredients | Molecular Formula | Molecular Weight |
| Potassium Acetate USP | CH3COOK | 98.14 |
| Sodium Chloride USP | NaCl | 58.44 |
| Sodium Acetate Trihydrate USP | CH3COONa•3H2O | 136.08 |
| Magnesium Chloride Hexahydrate USP | MgCl2•6H2O | 203.30 |
| Calcium Chloride Dihydrate USP | CaCl2•2H2O | 147.02 |
CLINICAL PHARMACOLOGY
Hyperlyte® CR provides a source of intra- and extracellular electrolytes in suitable amounts, to help maintain normal cellular metabolism during total parenteral nutrition (TPN) and other parenteral therapy in adults. Use of properly diluted Hyperlyte® CR meets most adult daily electrolyte requirements and provides adjunctive therapy for replenishment of depleted electrolyte stores.
Sodium, the major cation of the extracellular fluid, functions primarily in the control of water distribution, fluid balance, and osmotic pressure of body fluids. Sodium is also associated with chloride and bicarbonate in the regulation of the acid-base equilibrium of body fluid.
Potassium is the chief cation of body cells (160 mEq/liter of intracellular water) and is concerned with the maintenance of body fluid composition and electrolyte balance. Potassium participates in carbohydrate utilization and protein synthesis, and is critical in the regulation of nerve conduction and muscle contraction, particularly in the heart.
Chloride, the major extracellular anion, closely follows the metabolism of sodium, and changes in the acid-base balance of the body are reflected by changes in the chloride concentration.
Calcium, an important cation, provides the framework of bones and teeth in the form of calcium phosphate and calcium carbonate. In the ionized form, calcium is essential for the functional mechanism of the clotting of blood, normal cardiac function, and regulation of neuromuscular irritability.
Magnesium, a principal cation of soft tissue, is primarily involved in enzyme activity associated with the metabolism of carbohydrates and protein. Magnesium is also involved in neuromuscular irritability.
Acetate is an organic ion which is a hydrogen ion acceptor, contributes bicarbonate during its metabolism to carbon dioxide and water, and serves as an alkalinizing agent.
INDICATIONS AND USAGE
Hyperlyte® CR is indicated for use as a supplement to parenteral nutritional solutions containing amino acids, dextrose and/or other calorie sources delivered by central venous or peripheral infusion, to facilitate amino acid utilization and maintain electrolyte balance in adults.
Hyperlyte® CR (Multi-Electrolyte Concentrate) is also indicated as a source of replacement electrolytes for the depleted adult patient during parenteral therapy.
CONTRAINDICATIONS
Hyperlyte® CR is contraindicated in clinical conditions where the administration of potassium, sodium, calcium, magnesium, or chloride could be clinically detrimental.
Hyperlyte® CR is not intended for pediatric use.
WARNINGS
Strongly hypertonic solution. Must be properly diluted and thoroughly mixed before injection.
WARNING: This product contains aluminum that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration if kidney function is impaired. Premature neonates are particularly at risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum.
Research indicates that patients with impaired kidney function, including premature neonates, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
Contains no phosphate. Patients receiving TPN solutions containing concentrated dextrose may also require phosphate in addition to Hyperlyte® CR.
Each 20 mL dose contains 20 mEq of potassium. Patient requirements should be carefully evaluated prior to the addition of any potassium salt to a solution containing Hyperlyte® CR.
Solutions containing sodium ions should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency, and in clinical states in which there is sodium retention with edema.
Solutions containing potassium should be used with caution in the presence of cardiac disease, particularly in the presence of renal disease, and in such instances, cardiac monitoring is recommended.
In patients with diminished renal function, administration of solutions containing sodium or potassium ions may result in sodium or potassium retention.
Solutions containing calcium ions should not be administered through the same administration set as blood because of the likelihood of coagulation.
Solutions containing acetate should be used with great care in patients with metabolic or respiratory alkalosis. The administration of acetate should be done with great care in those conditions in which there is an increased level or an impaired utilization of acetate, such as severe hepatic insufficiency.
PRECAUTIONS
General
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation. Significant deviations from normal concentrations may require the use of additional electrolyte supplements, or the use of electrolyte-free dextrose solutions to which individualized electrolyte supplements may be added.
This solution should be used with care in patients with hypervolemia, renal insufficiency, urinary tract obstruction, or impending or frank cardiac decompensation.
Blood levels of sodium, potassium, calcium, magnesium, phosphorus and chloride should be monitored frequently during parenteral nutrition or intravenous therapy, and the daily dosage of electrolytes may require tailoring to meet individual needs. In tissue electrolyte depletion, addition of certain electrolytes may be required to meet individual patient needs.
Sodium-containing solutions should be administered with caution to patients receiving corticosteroids or corticotropin, or to other salt-retaining patients.
Care must be exercised in administering solutions containing up to 30 mEq of sodium and 40 mEq of potassium per liter to patients with renal or cardiovascular insufficiency, with or without congestive heart failure, particularly if they are postoperative or elderly.
Potassium therapy should be guided by serial electrocardiograms since plasma levels are not necessarily indicative of tissue potassium levels.
Solutions containing potassium, magnesium or calcium should be used with caution in the presence of cardiac disease, particularly when accompanied by renal disease. Parenteral magnesium or calcium should be administered with extreme caution to patients receiving digitalis preparations.
Extraordinary electrolyte and fluid losses are not necessarily corrected by infusion of solutions containing Hyperlyte® CR. In order to avoid deficits, special consideration must be given to replacement of excessive fluid and electrolyte losses in such conditions as protracted vomiting or diarrhea, nasogastric suction, or fistula drainage.
If both phosphate and Hyperlyte® CR are to be added to the solution for TPN administration, add the Hyperlyte® CR to one container (either to the amino acids or the concentrated dextrose) and add the phosphate to the other to avoid physical incompatibilities between calcium and phosphorus.
Solutions containing acetate should be used with caution. Excess administration may result in metabolic alkalosis.
If the administration is controlled by a pumping device, care must be taken to discontinue pumping action before the container runs dry or air embolism may result.
To minimize the risk of possible incompatibilities arising from mixing this solution with other additives that may be prescribed, the final infusate should be inspected for cloudiness or precipitation immediately after mixing, prior to administration and periodically during administration.
Use only if solution is clear and vacuum is present. Discard the container no later than 4 hours after initial closure puncture.
Laboratory Tests
Periodic laboratory determinations are necessary to monitor changes in electrolyte concentrations, and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
Drug Interactions
Administration of barbiturates, narcotics, hypnotics or systemic anesthetics should be adjusted and used with caution in patients also receiving magnesium-containing solutions because of an additive central depressive effect.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No in vitro or in vivo carcinogenesis, mutagenesis, or fertility studies have been conducted with Hyperlyte® CR (Multi-Electrolyte Concentrate).
Pregnancy
Teratogenic Effects
Animal reproduction studies have not been conducted with Hyperlyte® CR (Multi-Electrolyte Concentrate). It is also not known whether Hyperlyte® CR can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Hyperlyte® CR should be given to a pregnant woman only if clearly needed.
Labor and Delivery
As reported in the literature, electrolyte containing solutions have been administered during labor and delivery. Caution should be exercised, and the fluid balance, glucose and electrolyte concentrations and acid-base balance, of both mother and fetus should be evaluated periodically or whenever warranted by the condition of the patient or fetus.
Nursing Mothers
Because many drugs are excreted in human milk, caution should be exercised when solutions containing electrolytes are administered to a nursing woman.
Pediatric Use
Safety and effectiveness in pediatric populations have not been established.
See WARNINGS and CONTRAINDICATIONS .
Geriatric Use
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
See WARNINGS.
ADVERSE REACTIONS
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
Symptoms may result from an excess or deficit of one or more of the ions present in the final infusate. Therefore, frequent monitoring of blood electrolyte levels is essential.
Hypernatremia may be associated with edema and exacerbation of congestive heart failure due to the retention of water, resulting in an expanded extracellular fluid volume.
Reactions reported with the use of potassium-containing solutions include nausea, vomiting, abdominal pain and diarrhea.
The signs and symptoms of potassium intoxication include paresthesias of the extremities, areflexia, muscular or respiratory paralysis, mental confusion, weakness, hypotension, cardiac arrhythmias, heart block, electrocardiographic abnormalities and cardiac arrest.
Potassium deficits result in disruption of neuromuscular functions, and intestinal ileus and dilatation.
If infused in large amounts, chloride ions may cause a loss of bicarbonate ions, resulting in an acidifying effect.
Abnormally high plasma levels of calcium can result in depression, amnesia, headaches, drowsiness, disorientation, syncope, hallucinations, hypotonia of both skeletal and smooth muscles, dysphagia, arrhythmias and coma.
Calcium deficits can result in neuromuscular hyperexcitability, including cramps and convulsions.
Abnormally high plasma levels of magnesium can result in flushing, sweating, hypotension, circulatory collapse, and depression of cardiac and central nervous system function. Respiratory depression is the most immediate threat to life.
Magnesium deficits can result in tachycardia, hypertension, hyperirritability and psychotic behavior.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
OVERDOSAGE
In the event of a fluid or solute overload during parenteral therapy, reevaluate the patient’s condition and institute appropriate corrective treatment.
In the event of overdosage with potassium-containing solutions, discontinue the infusion immediately and institute corrective therapy to reduce serum potassium levels.
Treatment of hyperkalemia includes the following:
- Dextrose Injection USP, 10% or 25% containing 10 units of crystalline insulin per 20 grams of dextrose administered intravenously, 300 to 500 mL per hour.
-
Absorption and exchange of potassium using sodium or ammonium cycle cation exchange resin, orally and as retention enema.
-
Hemodialysis and peritoneal dialysis. The use of potassium-containing foods or medications must be eliminated. However, in cases of digitalization, too rapid a lowering of plasma potassium concentration can cause digitalis toxicity.
DOSAGE AND ADMINISTRATION
Not for direct patient injection.
Hyperlyte® CR (Multi-Electrolyte Concentrate) in Pharmacy Bulk Package is for prescription compounding of intravenous admixtures only. Not for direct patient injection. Dilute to appropriate strength with suitable intravenous fluid prior to administration.
For adults, one 20 mL dose of Hyperlyte® CR should be added to each liter of amino acid/dextrose solution (TPN) or other suitable intravenous solution.
Hyperlyte® CR contains no phosphate. It may be admixed with solutions which contain phosphate or which have been supplemented with phosphate. The presence of calcium and magnesium ions in this solution should be considered when phosphate is present in the additive solution in order to avoid precipitation.
Fluid administration should be based on calculated maintenance or replacement fluid requirements for each patient.
Some additives may be incompatible. Consult with pharmacist. When introducing additives, use aseptic techniques. Mix thoroughly. Do not store.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to use, whenever solution and container permit.
HOW SUPPLIED
Hyperlyte® CR (Multi-Electrolyte Concentrate) is supplied sterile and nonpyrogenic in Pharmacy Bulk Package with solid stoppers, packaged 12 per case.
| Canada DIN | NDC | REF | Size |
| Hyperlyte® CR (Multi-Electrolyte Concentrate) | |||
| 01924311 | 0264-1943-20 | S9432 | 250 mL |
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product.
Directions for Use of Pharmacy Bulk Package in B. Braun Glass Containers with Solid Stoppers
Warning: Not for direct infusion. For preparation of admixtures for intravenous infusion.
The pharmacy bulk package is for use in a Pharmacy Admixture Service only. Use of this product is restricted to a suitable work area, such as a laminar flow hood (or an equivalent clean air compounding area).
Additives should not be made to Pharmacy Bulk Packages.
Designed for use with a vented sterile dispensing set.
- Before use, perform the following checks:
• Inspect each container. Read the label. Ensure solution is the one ordered and is within the expiration date.
• Invert container and carefully inspect the solution in good light for cloudiness, haze, or particulate matter; check the bottle for cracks or other damage. In checking for cracks, do not be confused by normal surface marks and seams on bottom and sides of bottle. These are not flaws. Look for bright reflections that have depth and penetrate into the wall of the bottle. Reject any such bottle. -
Remove plastic cap (see Figure 1).
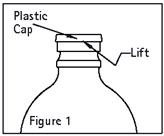
-
Swab exposed stopper surface with a suitable disinfectant.
-
Check for vacuum at first puncture of the stopper. Insert the spike fully into the target area of the rubber stopper (See Figure 2) and promptly invert the bottle. Verify vacuum by observing rising air bubbles. Do not use the bottle if vacuum is not present. Refer to Directions for Use of set to be used.
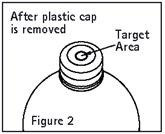
-
If set insertion is not performed immediately following swabbing, swab stopper again with a suitable disinfectant.
The container closure may be penetrated only one time, utilizing a suitable sterile dispensing set which allows measured dispensing of the contents.
Transfer individual dose(s) to appropriate intravenous infusion solutions. Use of a syringe with needle is not recommended. Multiple entries increase the potential of the microbial and particulate contamination.
The withdrawal of container contents should be accomplished without delay using aseptic technique. Discard container no later than 4 hours after initial closure puncture.
The bottle may be stored under laminar flow hood at room temperature (25°C) after the closure has been entered. Date and time of container entry should be noted in the area designated on the container label.
B. Braun Medical Inc.
Bethlehem, PA 18018-3524 USA
1-800-227-2862
In Canada, distributed by:
B. Braun of Canada, Ltd.
Scarborough, Ontario M1H 2W4
Y36-002-971 LD-447-2
Package Insert
Rx only
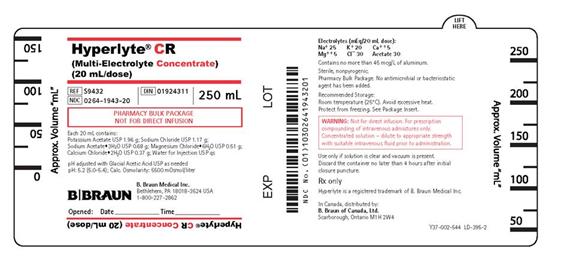
PRINCIPAL DISPLAY PANEL - 250 mL
Hyperlyte® CR
(Multi-Electrolyte Concentrate)
(20 mL/dose)
REF S9432
NDC 0264-1943-20
DIN 01924311
250 mL
PHARMACY BULK PACKAGE
NOT FOR DIRECT INFUSION
Each 20 mL contains:
Potassium Acetate USP 1.96 g; Sodium Chloride USP 1.17 g;
Sodium Acetate•3H2O USP 0.68 g; Magnesium Chloride•6H2O USP 0.51 g;
Calcium Chloride•2H2O USP 0.37 g; Water for Injection USP qs
pH adjusted with Glacial Acetic Acid USP as needed
pH: 5.2 (5.0-5.4); Calc. Osmolarity: 5500 mOsmol/liter
B. Braun Medical Inc.
Bethlehem, PA 18018-3524 USA
1-800-227-2862
Opened: Date Time
Electrolytes (mEq/20 mL dose):
Na+ 25 K+ 20 Ca++ 5
Mg++ 5 Cl– 30 Acetate 30
Contains no more than 45 mcg/L of aluminum.
Sterile, nonpyrogenic.
Pharmacy Bulk Package. No antimicrobial or bacteriostatic
agent has been added.
Recommended Storage:
Room temperature (25°C). Avoid excessive heat.
Protect from freezing. See Package Insert.
WARNING: Not for direct infusion. For prescription
compounding of intravenous admixtures only.
Concentrated solution – dilute to appropriate strength
with suitable intravenous fluid prior to administration.
Use only if solution is clear and vacuum is present.
Discard the container no later than 4 hours after initial
closure puncture.
Rx only
Hyperlyte is a registered trademark of B. Braun Medical Inc.
In Canada, distributed by:
B. Braun of Canada, Ltd.
Scarborough, Ontario M1H 2W4
Y37-002-544 LD-395-2
EXP
LOT
| HYPERLYTE
CR
multi electrolyte concentrate injection, solution, concentrate |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| Labeler - B. Braun Medical Inc. (002397347) |