PMS- viburnum opulus bark, caulophyllum thalictroides root, anamirta cocculus seed, and gelsemium sempervirens root tablet, soluble
Hyland's
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
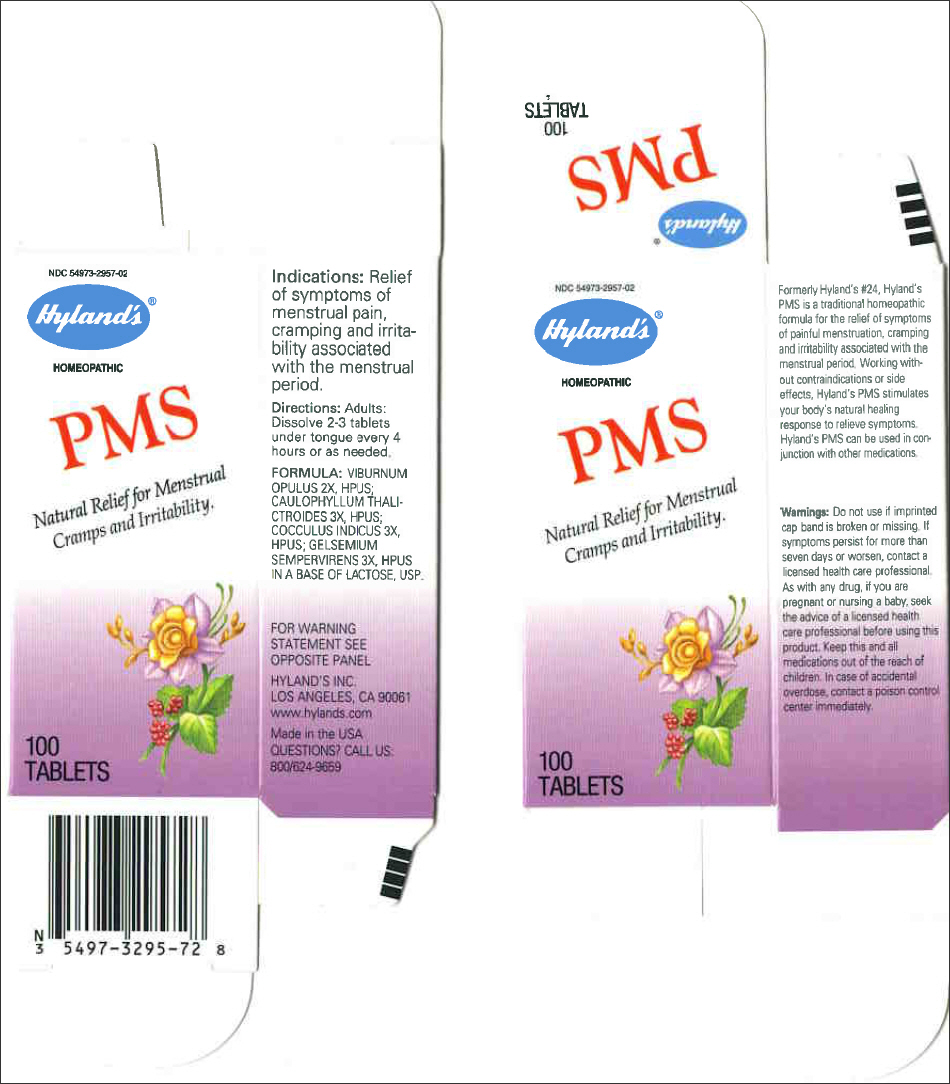
Formerly Hyland's #24, Hyland's PMS is a traditional homeopathic formula for the relief of symptoms of painful menstruation, cramping and irritability associated with the menstrual period. Working without contraindications or side effects, Hyland's PMS stimulates your body's natural healing response to relieve symptoms. Hyland's PMS can be used in conjunction with other medications.
Warnings
Do not use if imprinted cap band is broken or missing.
If symptoms persist for more than seven days or worsen, contact a licensed health care professional.
As with any drug, if you are pregnant or nursing a baby, seek the advice of a licensed health care professional before using this product.
Keep this and all medications out of the reach of children. In case of accidental overdose, contact a poison control center immediately.
Indications
Relief of symptoms of menstrual pain, cramping and irritability associated with the menstrual period.
Directions
Adults: Dissolve 2-3 tablets under tongue every 4 hours or as needed.
FORMULA
VIBURNUM OPULUS 2X, HPUS; CAULOPHYLLUM THALICTROIDES 3X, HPUS; COCCULUS INDICUS 3X, HPUS; GELSEMIUM SEMPERVIRENS 3X, HPUS IN A BASE OF LACTOSE, USP.
QUESTIONS?
CALL US: 800/624-9659
PRINCIPAL DISPLAY PANEL - 100 Tablet Bottle Carton
NDC 54973-2957-02
Hyland's
®
HOMEOPATHIC
PMS
Natural Relief for Menstrual
Cramps and Irritability.
100
TABLETS