Label: MOEXIPRIL HYDROCHLORIDE tablet, film coated
-
NDC Code(s):
68462-208-01,
68462-208-10,
68462-208-90,
68462-209-01, view more68462-209-10, 68462-209-90
- Packager: Glenmark Pharmaceuticals Inc., USA
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated March 28, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
Rx only
WARNING: FETAL TOXICITY
- •
- When pregnancy is detected, discontinue moexipril hydrochloride as soon as possible.
- •
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. See WARNINGS: Fetal Toxicity.
-
DESCRIPTION
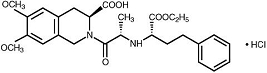
Moexipril hydrochloride USP, the hydrochloride salt of moexipril, has the empirical formula C27H34N2O7•HCl and a molecular weight of 535.04. It is chemically described as [3S-[2[R*(R*)],3R*]]-2-[2-[[1-(ethoxycarbonyl)-3-phenylpropyl]amino]-1-oxopropyl]-1,2,3,4-tetrahydro-6,7-dimethoxy-3-isoquinolinecarboxylic acid, monohydrochloride. It is a non-sulfhydryl containing precursor of the active angiotensin-converting enzyme (ACE) inhibitor moexiprilat and its structural formula is:
Moexipril hydrochloride USP is a fine white to off-white powder. It is soluble (about 10% weight-to-volume) in distilled water at room temperature.
Moexipril hydrochloride tablets USP are supplied as scored, coated tablets containing 7.5 mg and 15 mg of moexipril hydrochloride USP for oral administration. In addition to the active ingredient, moexipril hydrochloride USP, the tablet core contains the following inactive ingredients: crospovidone, lactose monohydrate, magnesium oxide, magnesium stearate and povidone. The film coating contains: hypromellose, hydroxypropyl cellulose, titanium dioxide, polyethylene glycol 6000, magnesium stearate, ferric oxide red, ferric oxide black and ferric oxide yellow (15 mg tablet only).
-
CLINICAL PHARMACOLOGY
Mechanism of Action
Moexipril hydrochloride is a prodrug for moexiprilat, which inhibits ACE in humans and animals. The mechanism through which moexiprilat lowers blood pressure is believed to be primarily inhibition of ACE activity. ACE is a peptidyl dipeptidase that catalyzes the conversion of the inactive decapeptide angiotensin I to the vasoconstrictor substance angiotensin II. Angiotensin II is a potent peripheral vasoconstrictor that also stimulates aldosterone secretion by the adrenal cortex and provides negative feedback on renin secretion. ACE is identical to kininase II, an enzyme that degrades bradykinin, an endothelium-dependent vasodilator. Moexiprilat is about 1000 times as potent as moexipril in inhibiting ACE and kininase II. Inhibition of ACE results in decreased angiotensin II formation, leading to decreased vasoconstriction, increased plasma renin activity, and decreased aldosterone secretion. The latter results in diuresis and natriuresis and a small increase in serum potassium concentration (mean increases of about 0.25 mEq/L were seen when moexipril was used alone, see PRECAUTIONS).
Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of moexipril remains to be elucidated. Although the principal mechanism of moexipril in blood pressure reduction is believed to be through the renin-angiotensin-aldosterone system, ACE inhibitors have some effect on blood pressure even in apparent low-renin hypertension. As is the case with other ACE inhibitors, however, the antihypertensive effect of moexipril is considerably smaller in black patients, a predominantly low-renin population, than in non-black hypertensive patients.
Pharmacokinetics and Metabolism
Pharmacokinetics:
Moexipril’s antihypertensive activity is almost entirely due to its deesterified metabolite, moexiprilat. Bioavailability of oral moexipril is about 13% compared to intravenous (I.V.) moexipril (both measuring the metabolite moexiprilat), and is markedly affected by food, which reduces the peak plasma level (Cmax) and AUC (see Absorption). Moexipril should therefore be taken in a fasting state. The time of peak plasma concentration (Tmax) of moexiprilat is about 11/2 hours and elimination half-life (t1/2) is estimated at 2 to 9 hours in various studies, the variability reflecting a complex elimination pattern that is not simply exponential. Like all ACE inhibitors, moexiprilat has a prolonged terminal elimination phase, presumably reflecting slow release of drug bound to the ACE. Accumulation of moexiprilat with repeated dosing is minimal, about 30%, compatible with a functional elimination t1/2 of about 12 hours. Over the dose range of 7.5 to 30 mg, pharmacokinetics are approximately dose proportional.
Absorption:
Moexipril is incompletely absorbed, with bioavailability as moexiprilat of about 13%. Bioavailability varies with formulation and food intake which reduces Cmax and AUC by about 70% and 40% respectively after the ingestion of a low-fat breakfast or by 80% and 50% respectively after the ingestion of a high-fat breakfast.
Distribution:
The clearance (CL) for moexipril is 441 mL/min and for moexiprilat 232 mL/min with a t½ of 1.3 and 9.8 hours, respectively. Moexiprilat is about 50% protein bound. The volume of distribution of moexiprilat is about 183 liters.
Metabolism and Excretion:
Moexipril is relatively rapidly converted to its active metabolite moexiprilat, but persists longer than some other ACE inhibitor prodrugs, such that its t ½ is over one hour and it has a significant AUC. Both moexipril and moexiprilat are converted to diketopiperazine derivatives and unidentified metabolites. After I.V. administration of moexipril, about 40% of the dose appears in urine as moexiprilat, about 26% as moexipril, with small amounts of the metabolites; about 20% of the I.V. dose appears in feces, principally as moexiprilat. After oral administration, only about 7% of the dose appears in urine as moexiprilat, about 1% as moexipril, with about 5% as other metabolites. Fifty-two percent of the dose is recovered in feces as moexiprilat and 1% as moexipril.
Special Populations:
Decreased Renal Function: The effective elimination t½ and AUC of both moexipril and moexiprilat are increased with decreasing renal function. There is insufficient information available to characterize this relationship fully, but at creatinine clearances in the range of 10 to 40 mL/min, the t½ of moexiprilat is increased by a factor of 3 to 4.
Decreased Hepatic Function: In patients with mild to moderate cirrhosis given single 15 mg doses of moexipril, the Cmax of moexipril was increased by about 50% and the AUC increased by about 120%, while the Cmax for moexiprilat was decreased by about 50% and the AUC increased by almost 300%.
Elderly Patients: In elderly male subjects (65 to 80 years old) with clinically normal renal and hepatic function, the AUC and Cmax of moexiprilat is about 30% greater than those of younger subjects (19 to 42 years old).
Pharmacodynamics and Clinical Effect
Single and multiple doses of 15 mg or more of moexipril hydrochloride gives sustained inhibition of plasma ACE activity of 80 to 90%, beginning within 2 hours and lasting 24 hours (80%).
In controlled trials, the peak effects of orally administered moexipril increased with the dose administered over a dose range of 7.5 to 60 mg, given once a day. Antihypertensive effects were first detectable about 1 hour after dosing, with a peak effect between 3 and 6 hours after dosing. Just before dosing (i.e., at trough), the antihypertensive effects were less prominently related to dose and the antihypertensive effect tended to diminish during the 24-hour dosing interval when the drug was administered once a day.
In multiple dose studies in the dose range of 7.5 to 30 mg once daily, moexipril hydrochloride lowered sitting diastolic and systolic blood pressure effects at trough by 3 to 6 mmHg and 4 to 11 mmHg more than placebo, respectively. There was a tendency toward increased response with higher doses over this range. These effects are typical of ACE inhibitors but, to date, there are no trials of adequate size comparing moexipril with other antihypertensive agents.
The trough diastolic blood pressure effects of moexipril were approximately 3 to 6 mmHg in various studies. Generally, higher doses of moexipril leave a greater fraction of the peak blood pressure effect still present at trough. During dose titration, any decision as to the adequacy of a dosing regimen should be based on trough blood pressure measurements. If diastolic blood pressure control is not adequate at the end of the dosing interval, the dose can be increased or given as a divided (BID) regimen.
During chronic therapy, the antihypertensive effect of any dose of moexipril hydrochloride is generally evident within 2 weeks of treatment, with maximal reduction after 4 weeks. The antihypertensive effects of moexipril hydrochloride have been proven to continue during therapy for up to 24 months.
Moexipril hydrochloride, like other ACE inhibitors, is less effective in decreasing trough blood pressures in blacks than in non-blacks. Placebo-corrected trough group mean diastolic blood pressure effects in blacks in the proposed dose range varied between +1 to -3 mmHg compared with responses in non-blacks of -4 to -6 mmHg.
The effectiveness of moexipril hydrochloride was not significantly influenced by patient age, gender, or weight. Moexipril hydrochloride has been shown to have antihypertensive activity in both pre- and postmenopausal women who have participated in placebo-controlled clinical trials.
Formal interaction studies with moexipril have not been carried out with antihypertensive agents other than thiazide diuretics. In these studies, the added effect of moexipril was similar to its effect as monotherapy. In general, ACE inhibitors have less than additive effects with beta-adrenergic blockers, presumably because both work by inhibiting the renin-angiotensin system.
-
INDICATIONS AND USAGE
Moexipril hydrochloride tablets are indicated for treatment of patients with hypertension. They may be used alone or in combination with thiazide diuretics.
In using moexipril hydrochloride tablets, consideration should be given to the fact that another ACE inhibitor, captopril, has caused agranulocytosis, particularly in patients with renal impairment or collagen-vascular disease. Available data are insufficient to show that moexipril hydrochloride tablets do not have a similar risk (see WARNINGS).
In considering use of moexipril hydrochloride tablets, it should be noted that in controlled trials ACE inhibitors have an effect on blood pressure that is less in black patients than in non-blacks. In addition, ACE inhibitors (for which adequate data are available) cause a higher rate of angioedema in black than in non-black patients (see WARNINGS, Angioedema).
-
CONTRAINDICATIONS
Moexipril hydrochloride is contraindicated in patients who are hypersensitive to this product and in patients with a history of angioedema related to previous treatment with an ACE inhibitor.
Do not co-administer aliskiren with moexipril hydrochloride in patients with diabetes (see PRECAUTIONS, Drug Interactions).
-
WARNINGS
Anaphylactoid and Possibly Related Reactions
Presumably because angiotensin-converting enzyme inhibitors affect the metabolism of eicosanoids and polypeptides, including endogenous bradykinin, patients receiving ACE inhibitors, including moexipril hydrochloride, may be subject to a variety of adverse reactions, some of them serious.
Head and Neck Angioedema:
Angioedema involving the face, extremities, lips, tongue, glottis, and/or larynx has been reported in patients treated with ACE inhibitors, including moexipril hydrochloride. Symptoms suggestive of angioedema or facial edema occurred in < 0.5% of moexipril-treated patients in placebo-controlled trials. None of the cases were considered life-threatening and all resolved either without treatment or with medication (antihistamines or glucocorticoids). One patient treated with hydrochlorothiazide alone experienced laryngeal edema. No instances of angioedema were reported in placebo-treated patients.
In cases of angioedema, treatment should be promptly discontinued and the patient carefully observed until the swelling disappears. In instances where swelling has been confined to the face and lips, the condition has generally resolved without treatment, although antihistamines have been useful in relieving symptoms.
Angioedema associated with involvement of the tongue, glottis, or larynx, may be fatal due to airway obstruction. Appropriate therapy, e.g., subcutaneous epinephrine solution 1:1000 (0.3 to 0.5 mL) and/or measures to ensure a patent airway, should be promptly provided (see ADVERSE REACTIONS).
Intestinal Angioedema:
Intestinal angioedema has been reported in patients treated with ACE inhibitors. These patients presented with abdominal pain (with or without nausea or vomiting); in some cases there was no prior history of facial angioedema and C-1 esterase levels were normal. The angioedema was diagnosed by procedures including abdominal CT scan or ultrasound, or at surgery, and symptoms resolved after stopping the ACE inhibitor. Intestinal angioedema should be included in the differential diagnosis of patients on ACE inhibitors presenting with abdominal pain.
Anaphylactoid Reactions During Desensitization:
Two patients undergoing desensitizing treatment with hymenoptera venom while receiving ACE inhibitors sustained life-threatening anaphylactoid reactions. In the same patients, these reactions did not occur when ACE inhibitors were temporarily withheld, but they reappeared when the ACE inhibitors were inadvertently readministered.
Anaphylactoid Reactions During Membrane Exposure:
Anaphylactoid reactions have been reported in patients dialyzed with high-flux membranes and treated concomitantly with an ACE inhibitor. Anaphylactoid reactions have also been reported in patients undergoing low-density lipoprotein apheresis with dextran sulfate absorption.
Hypotension
Moexipril hydrochloride can cause symptomatic hypotension, although, as with other ACE inhibitors, this is unusual in uncomplicated hypertensive patients treated with moexipril hydrochloride alone. Symptomatic hypotension was seen in 0.5% of patients given moexipril and led to discontinuation of therapy in about 0.25%. Symptomatic hypotension is most likely to occur in patients who have been salt- and volume- depleted as a result of prolonged diuretic therapy, dietary salt restriction, dialysis, diarrhea, or vomiting. Volume- and salt-depletion should be corrected and, in general, diuretics stopped, before initiating therapy with moexipril hydrochloride (see PRECAUTIONS, Drug Interactions, and ADVERSE REACTIONS).
In patients with congestive heart failure, with or without associated renal insufficiency, ACE inhibitor therapy may cause excessive hypotension, which may be associated with oliguria or progressive azotemia, and rarely, with acute renal failure and death. In these patients, moexipril hydrochloride therapy should be started under close medical supervision, and patients should be followed closely for the first two weeks of treatment and whenever the dose of moexipril or an accompanying diuretic is increased. Care in avoiding hypotension should also be taken in patients with ischemic heart disease, aortic stenosis, or cerebrovascular disease, in whom an excessive decrease in blood pressure could result in a myocardial infarction or a cerebrovascular accident.
If hypotension occurs, the patient should be placed in a supine position and, if necessary, treated with an intravenous infusion of normal saline. Moexipril hydrochloride treatment usually can be continued following restoration of blood pressure and volume.
Neutropenia/Agranulocytosis
Another ACE inhibitor, captopril, has been shown to cause agranulocytosis and bone marrow depression, rarely in patients with uncomplicated hypertension, but more frequently in hypertensive patients with renal impairment, especially if they also have a collagen-vascular disease such as systemic lupus erythematosus or scleroderma. Although there were no instances of severe neutropenia (absolute neutrophil count < 500/mm3) among patients given moexipril hydrochloride, as with other ACE inhibitors, monitoring of white blood cell counts should be considered for patients who have collagen-vascular disease, especially if the disease is associated with impaired renal function. Available data from clinical trials of moexipril hydrochloride are insufficient to show that moexipril hydrochloride does not cause agranulocytosis at rates similar to captopril.
Fetal Toxicity
Pregnancy Category D
Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue moexipril hydrochloride as soon as possible. These adverse outcomes are usually associated with use of these drugs in the second and third trimester of pregnancy. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. Appropriate management of maternal hypertension during pregnancy is important to optimize outcomes for both mother and fetus.
In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus. Perform serial ultrasound examinations to assess the intra-amniotic environment. If oligohydramnios is observed, discontinue moexipril hydrochloride, unless it is considered lifesaving for the mother. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to moexipril hydrochloride for hypotension, oliguria, and hyperkalemia (see PRECAUTIONS, Pediatric Use).
No embryotoxic, fetotoxic, or teratogenic effects were seen in rats or in rabbits treated with up to 90.9 and 0.7 times, respectively, the Maximum Recommended Human Dose (MRHD) on a mg/m2 basis.
Hepatic Failure
Rarely, ACE inhibitors have been associated with a syndrome that starts with cholestatic jaundice and progresses to fulminant hepatic necrosis and sometimes death. The mechanism of this syndrome is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevations of hepatic enzymes should discontinue the ACE inhibitor and receive appropriate medical follow-up.
-
PRECAUTIONS
General
Impaired Renal Function:
As a consequence of inhibition of the renin-angiotensin-aldosterone system, changes in renal function may be anticipated in susceptible individuals. There is no clinical experience of moexipril hydrochloride in the treatment of hypertension in patients with renal failure.
Some hypertensive patients with no apparent preexisting renal vascular disease have developed increases in blood urea nitrogen and serum creatinine, usually minor and transient, especially when moexipril hydrochloride has been given concomitantly with a thiazide diuretic. This is more likely to occur in patients with preexisting renal impairment. There may be a need for dose adjustment of moexipril hydrochloride and/or the discontinuation of the thiazide diuretic.
Evaluation of hypertensive patients should always include assessment of renal function (see DOSAGE AND ADMINISTRATION).
Hypertensive Patients With Congestive Heart Failure:
In hypertensive patients with severe congestive heart failure, whose renal function may depend on the activity of the renin-angiotensin-aldosterone system, treatment with ACE inhibitors, including moexipril hydrochloride, may be associated with oliguria and/or progressive azotemia and, rarely, acute renal failure and/or death.
Hypertensive Patients With Renal Artery Stenosis:
In hypertensive patients with unilateral or bilateral renal artery stenosis, increases in blood urea nitrogen and serum creatinine have been observed in some patients following ACE inhibitor therapy. These increases were almost always reversible upon discontinuation of the ACE inhibitor and/or diuretic therapy. In such patients, renal function should be monitored during the first few weeks of therapy.
Hyperkalemia:
In clinical trials, persistent hyperkalemia (serum potassium above 5.4 mEq/L) occurred in approximately 1.3% of hypertensive patients receiving moexipril hydrochloride. Risk factors for the development of hyperkalemia with ACE inhibitors include renal insufficiency, diabetes mellitus, and the concomitant use of potassium-sparing diuretics, potassium supplements, and/or potassium-containing salt substitutes, which should be used cautiously, if at all, with moexipril hydrochloride (see PRECAUTIONS, Drug Interactions).
Surgery/Anesthesia:
In patients undergoing major surgery or during anesthesia with agents that produce hypotension, moexipril may block the effects of compensatory renin release. If hypotension occurs in this setting and is considered to be due to this mechanism, it can be corrected by volume expansion.
Cough:
Presumably due to the inhibition of the degradation of endogenous bradykinin, persistent nonproductive cough has been reported with all ACE inhibitors, always resolving after discontinuation of therapy. ACE inhibitor-induced cough should be considered in the differential diagnosis of cough. In controlled trials with moexipril, cough was present in 6.1% of moexipril patients and 2.2% of patients given placebo.
Information for Patients
Food:
Patients should be advised to take moexipril one hour before meals (see CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION).
Angioedema:
Angioedema, including laryngeal edema, may occur with treatment with ACE inhibitors, usually occurring early in therapy (within the first month). Patients should be so advised and told to report immediately any signs or symptoms suggesting angioedema (swelling of the face, extremities, eyes, lips, tongue, difficulty in breathing) and to take no more moexipril hydrochloride until they have consulted with the prescribing physician.
Symptomatic Hypotension:
Patients should be cautioned that lightheadedness can occur with moexipril hydrochloride, especially during the first few days of therapy. If fainting occurs, the patient should stop taking moexipril hydrochloride and consult the prescribing physician.
All patients should be cautioned that excessive perspiration and dehydration may lead to an excessive fall in blood pressure because of reduction in fluid volume. Other causes of volume depletion such as vomiting or diarrhea may also lead to a fall in blood pressure; patients should be advised to consult their physician if they develop these conditions.
Hyperkalemia:
Patients should be told not to use potassium supplements or salt substitutes containing potassium without consulting their physician.
Drug Interactions
Diuretics:
Excessive reductions in blood pressure may occur in patients on diuretic therapy when ACE inhibitors are started. The possibility of hypotensive effects with moexipril hydrochloride can be minimized by discontinuing diuretic therapy for several days or cautiously increasing salt intake before initiation of treatment with moexipril hydrochloride. If this is not possible, the starting dose of moexipril should be reduced. (See WARNINGS and DOSAGE AND ADMINISTRATION).
Potassium Supplements and Potassium-Sparing Diuretics:
Moexipril hydrochloride can increase serum potassium because it decreases aldosterone secretion. Use of potassium-sparing diuretics (spironolactone, triamterene, amiloride) or potassium supplements concomitantly with ACE inhibitors can increase the risk of hyperkalemia. Therefore, if concomitant use of such agents is indicated, they should be given with caution and the patient’s serum potassium should be monitored.
Oral Anticoagulants:
Interaction studies with warfarin failed to identify any clinically important effect on the serum concentrations of the anticoagulant or on its anticoagulant effect.
Lithium:
Increased serum lithium levels and symptoms of lithium toxicity have been reported in patients receiving ACE inhibitors during therapy with lithium. These drugs should be coadministered with caution, and frequent monitoring of serum lithium levels is recommended. If a diuretic is also used, the risk of lithium toxicity may be increased.
Gold:
Nitritoid reactions (symptoms include facial flushing, nausea, vomiting and hypotension) have been reported rarely in patients on therapy with injectable gold (sodium aurothiomalate) and concomitant ACE inhibitor therapy including moexipril hydrochloride.
Non-Steroidal Anti-Inflammatory Agents including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors):
In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, co-administration of NSAIDS, including selective COX-2 inhibitors, with ACE inhibitors, including moexipril, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving moexipril and NSAID therapy.
The antihypertensive effect of ACE inhibitors, including moexipril, may be attenuated by NSAIDS.
Dual Blockade of the Renin-Angiotensin System (RAS):
Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Closely monitor blood pressure, renal function and electrolytes in patients on moexipril hydrochloride and other agents that affect the RAS.
Do not co-administer aliskiren with moexipril hydrochloride in patients with diabetes. Avoid use of aliskiren with moexipril hydrochloride in patients with renal impairment (GFR <60 mL/min).
Other Agents:
No clinically important pharmacokinetic interactions occurred when moexipril hydrochloride was administered concomitantly with hydrochlorothiazide, digoxin, or cimetidine.
Moexipril hydrochloride has been used in clinical trials concomitantly with calcium-channel-blocking agents, diuretics, H2 blockers, digoxin, oral hypoglycemic agents, and cholesterol-lowering agents. There was no evidence of clinically important adverse interactions.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of carcinogenicity was detected in long-term studies in mice and rats at doses up to 14 or 27.3 times the Maximum Recommended Human Dose (MRHD) on a mg/m2 basis.
No mutagenicity was detected in the Ames test and microbial reverse mutation assay, with and without metabolic activation, or in an in vivo nucleus anomaly test. However, increased chromosomal aberration frequency in Chinese hamster ovary cells was detected under metabolic activation conditions at a 20-hour harvest time.
Reproduction studies have been performed in rabbits at oral doses up to 0.7 times the MRHD on a mg/m2 basis, and in rats up to 90.9 times the MRHD on a mg/m2 basis. No indication of impaired fertility, reproductive toxicity, or teratogenicity was observed.
Nursing Mothers
It is not known whether moexipril hydrochloride is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when moexipril hydrochloride is given to a nursing mother.
Pediatric Use
Neonates with a history of in utero exposure to moexipril hydrochloride: If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and/or substituting for disordered renal function.
Safety and effectiveness of moexipril hydrochloride in pediatric patients have not been established.
Geriatric Use
Clinical studies of moexipril hydrochloride did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
-
ADVERSE REACTIONS
Moexipril hydrochloride has been evaluated for safety in more than 2500 patients with hypertension; more than 250 of these patients were treated for approximately one year. The overall incidence of reported adverse events was only slightly greater in patients treated with moexipril hydrochloride than patients treated with placebo.
Reported adverse experiences were usually mild and transient, and there were no differences in adverse reaction rates related to gender, race, age, duration of therapy, or total daily dosage within the range of 3.75 mg to 60 mg. Discontinuation of therapy because of adverse experiences was required in 3.4% of patients treated with moexipril hydrochloride and in 1.8% of patients treated with placebo. The most common reasons for discontinuation in patients treated with moexipril hydrochloride were cough (0.7%) and dizziness (0.4%).
All adverse experiences considered at least possibly related to treatment that occurred at any dose in placebo-controlled trials of once-daily dosing in more than 1% of patients treated with moexipril hydrochloride alone and that were at least as frequent in the moexipril hydrochloride group as in the placebo group are shown in the following table:
ADVERSE EVENTS IN PLACEBO-CONTROLLED STUDIES ADVERSE EVENT
MOEXIPRIL HYDROCHLORIDE (N=674)
PLACEBO (N=226)
N (%)
N (%)
Cough Increased
41 (6.1)
5 (2.2)
Dizziness
29 (4.3)
5 (2.2)
Diarrhea
21 (3.1)
5 (2.2)
Flu Syndrome
21 (3.1)
0 (0)
Fatigue
16 (2.4)
4 (1.8)
Pharyngitis
12 (1.8)
2 (0.9)
Flushing
11 (1.6)
0 (0)
Rash
11 (1.6)
2 (0.9)
Myalgia
9 (1.3)
0 (0)
Other adverse events occurring in more than 1% of patients on moexipril that were at least as frequent on placebo include: headache, upper respiratory infection, pain, rhinitis, dyspepsia, nausea, peripheral edema, sinusitis, chest pain, and urinary frequency. See WARNINGS and PRECAUTIONS for discussion of anaphylactoid reactions, angioedema, hypotension, neutropenia/agranulocytosis, second and third trimester fetal/neonatal morbidity and mortality, hyperkalemia, and cough.
Other potentially important adverse experiences reported in controlled or uncontrolled clinical trials in less than 1% of moexipril patients or that have been attributed to other ACE inhibitors include the following:
Cardiovascular:
Symptomatic hypotension, postural hypotension, or syncope were seen in 9/1750 (0.51%) patients; these reactions led to discontinuation of therapy in controlled trials in 3/1254 (0.24%) patients who had received moexipril hydrochloride monotherapy and in 1/344 (0.3%) patients who had received moexipril hydrochloride with hydrochlorothiazide (see PRECAUTIONS and WARNINGS). Other adverse events included angina/myocardial infarction, palpitations, rhythm disturbances, and cerebrovascular accident.
Renal:
Of hypertensive patients with no apparent preexisting renal disease, 1% of patients receiving moexipril hydrochloride alone and 2% of patients receiving moexipril hydrochloride with hydrochlorothiazide experienced increases in serum creatinine to at least 140% of their baseline values (see PRECAUTIONS and DOSAGE AND ADMINISTRATION).
Gastrointestinal:
Abdominal pain, constipation, vomiting, appetite/weight change, dry mouth, pancreatitis, hepatitis.
Dermatologic:
Apparent hypersensitivity reactions manifested by urticaria, rash, pemphigus, pruritus, photosensitivity, alopecia.
Other:
Angioedema (see WARNINGS), taste disturbances, tinnitus, sweating, malaise, arthralgia, hemolytic anemia.
Clinical Laboratory Test Findings
Creatinine and Blood Urea Nitrogen:
As with other ACE inhibitors, minor increases in blood urea nitrogen or serum creatinine, reversible upon discontinuation of therapy, were observed in approximately 1% of patients with essential hypertension who were treated with moexipril hydrochloride. Increases are more likely to occur in patients receiving concomitant diuretics and in patients with compromised renal function (see PRECAUTIONS, General).
Other (causal relationship unknown):
Clinically important changes in standard laboratory tests were rarely associated with moexipril hydrochloride administration.
Elevations of liver enzymes and uric acid have been reported. In trials, less than 1% of moexipril-treated patients discontinued moexipril hydrochloride treatment because of laboratory abnormalities. The incidence of abnormal laboratory values with moexipril was similar to that in the placebo-treated group.
-
OVERDOSAGE
Human overdoses of moexipril have not been reported. In case reports of overdoses with other ACE inhibitors, hypotension has been the principal adverse effect noted. Single oral doses of 2 g/kg moexipril were associated with significant lethality in mice. Rats, however, tolerated single oral doses of up to 3 g/kg.
No data are available to suggest that physiological maneuvers (e.g., maneuvers to change the pH of the urine) would accelerate elimination of moexipril and its metabolites. The dialyzability of moexipril is not known.
Angiotensin II could presumably serve as a specific antagonist-antidote in the setting of moexipril overdose, but angiotensin II is essentially unavailable outside of research facilities. Because the hypotensive effect of moexipril is achieved through vasodilation and effective hypovolemia, it is reasonable to treat moexipril overdose by infusion of normal saline solution. In addition, renal function and serum potassium should be monitored.
-
DOSAGE AND ADMINISTRATION
Hypertension
The recommended initial dose of moexipril hydrochloride tablets in patients not receiving diuretics is 7.5 mg, one hour prior to meals, once daily. Dosage should be adjusted according to blood pressure response. The antihypertensive effect of moexipril hydrochloride tablets may diminish towards the end of the dosing interval. Blood pressure should, therefore, be measured just prior to dosing to determine whether satisfactory blood pressure control is obtained. If control is not adequate, increased dose or divided dosing can be tried. The recommended dose range is 7.5 to 30 mg daily, administered in one or two divided doses one hour before meals. Total daily doses above 60 mg a day have not been studied in hypertensive patients.
In patients who are currently being treated with a diuretic, symptomatic hypotension may occasionally occur following the initial dose of moexipril hydrochloride tablets. The diuretic should, if possible, be discontinued for 2 to 3 days before therapy with moexipril hydrochloride tablets is begun, to reduce the likelihood of hypotension (see WARNINGS). If the patient’s blood pressure is not controlled with moexipril hydrochloride tablets alone, diuretic therapy may then be reinstituted. If diuretic therapy cannot be discontinued, an initial dose of 3.75 mg of moexipril hydrochloride tablets should be used with medical supervision until blood pressure has stabilized (see WARNINGS and PRECAUTIONS, Drug Interactions).
-
HOW SUPPLIED
Moexipril hydrochloride tablets USP 7.5 mg are peach, round, biconvex, film coated tablets with ‘G' and breakline engraved on one side and ‘209’ on the other side.
Bottles of 90 NDC 68462-209-90
Bottles of 100 NDC 68462-209-01
Bottles of 1000 NDC 68462-209-10Moexipril hydrochloride tablets USP 15 mg are brown, round, biconvex, film coated tablets with ‘G' and breakline engraved on one side and ‘208’ on the other side.
Bottles of 90 NDC 68462-208-90
Bottles of 100 NDC 68462-208-01
Bottles of 1000 NDC 68462-208-10 - STORAGE AND HANDLING
- Principal Display Panel - 7.5 mg Bottle
- Principal Display Panel - 15 mg Bottle
-
INGREDIENTS AND APPEARANCE
MOEXIPRIL HYDROCHLORIDE
moexipril hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68462-209 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MOEXIPRIL HYDROCHLORIDE (UNII: Q1UMG3UH45) (MOEXIPRILAT - UNII:H3753190JS) MOEXIPRIL HYDROCHLORIDE 7.5 mg Inactive Ingredients Ingredient Name Strength CROSPOVIDONE (120 .MU.M) (UNII: 68401960MK) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM OXIDE (UNII: 3A3U0GI71G) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) Product Characteristics Color PINK (Peach) Score 2 pieces Shape ROUND (biconvex) Size 6mm Flavor Imprint Code G;209 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68462-209-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/31/2010 2 NDC:68462-209-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 12/31/2010 3 NDC:68462-209-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/31/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090416 12/31/2010 MOEXIPRIL HYDROCHLORIDE
moexipril hydrochloride tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68462-208 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MOEXIPRIL HYDROCHLORIDE (UNII: Q1UMG3UH45) (MOEXIPRILAT - UNII:H3753190JS) MOEXIPRIL HYDROCHLORIDE 15 mg Inactive Ingredients Ingredient Name Strength CROSPOVIDONE (120 .MU.M) (UNII: 68401960MK) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM OXIDE (UNII: 3A3U0GI71G) MAGNESIUM STEARATE (UNII: 70097M6I30) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 6000 (UNII: 30IQX730WE) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) Product Characteristics Color BROWN Score 2 pieces Shape ROUND (biconvex) Size 8mm Flavor Imprint Code G;208 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68462-208-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 12/31/2010 2 NDC:68462-208-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 12/31/2010 3 NDC:68462-208-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 12/31/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090416 12/31/2010 Labeler - Glenmark Pharmaceuticals Inc., USA (130597813) Establishment Name Address ID/FEI Business Operations Glenmark Pharmaceuticals Limited 677318665 ANALYSIS(68462-209, 68462-208) , MANUFACTURE(68462-209, 68462-208)