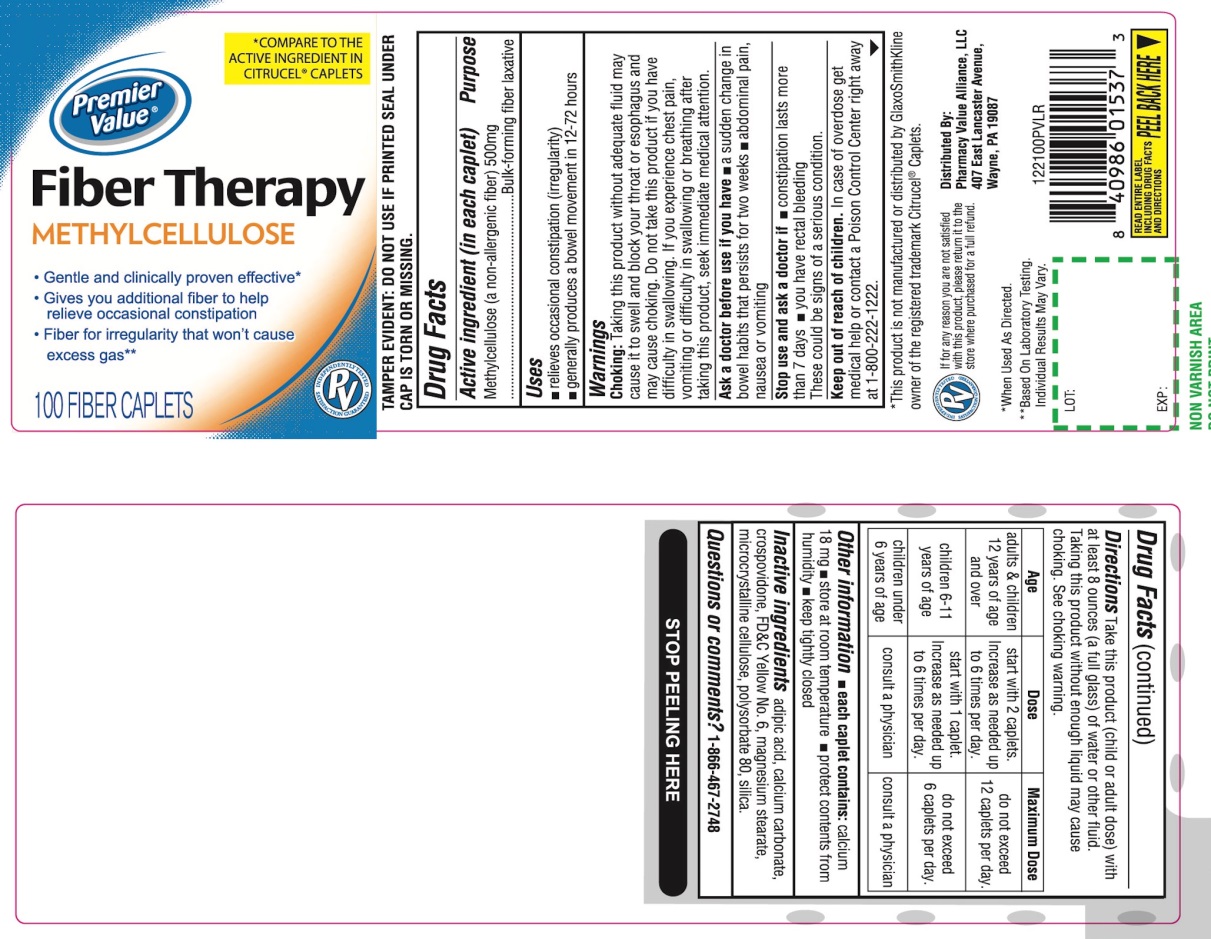
Label: PREMIER VALUE FIBER THERAPY- methylcellulose tablet
- NDC Code(s): 68016-122-10
- Packager: PHARMACY VALUE ALLIANCE, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 3, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each caplet)
- Purpose
-
Uses
- ▪
- relieves occasional constipation (irregularity)
- ▪
- generally produces a bowel movement in 12 -72 hours.
Warnings
Choking: Taking this product without adequate fluid may cause it to swell and block your throat or esophagus and may cause choking. Do not take this product if you have difficulty in swallowing. If you experience chest pain, vomiting or difficulty in swallowing or breathing after taking this product, seek immediate medical attention.
-
Directions
Take this product (child or adult dose) with at least 8 ounces (a full glass) of water or other fluid.
Taking this product without enough liquid may cause choking. See choking warning.
Age
Dose
Maximum Dose
adults & children 12 years of age and over
start with 2 caplets. Increase as needed up to 6 times per day.
Do not exceed 12 caplets per day
Children 6-11 years of age
Start with 1 caplet. Increase as needed up to 6 times per day
Do not exceed 6 caplets per day
children under 6 years of age
Consult a physician
Consult a physician
- Other information
- Inactive ingredients
-
Principal Display Panel
Premier Value®
NDC# 68016-122-10
*COMPARE TO THE ACTIVE INGREDIENTS CITRUCEL® CAPLETS
Fiber Therapy
Fiber Therapy for Regularity
Methylcellulose
- •
- Gentle and clinically proven effective
- •
- Gives you additional fiber to help relieves occasional constipation
- •
- Fiber for irregularity that won’t cause excess gas**
- 100 CAPLETS
If for any reason you are not satisfied with this product, please return it to the store where purchased for a full refund.
*When Used As Directed.
**Based on Laboratory Testing.
Individual Results May Vary.
Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
*This product is not manufactured or distributed by GlaxoSmithKline, owner of the registered trademark Citrucel® Caplets.
-
INGREDIENTS AND APPEARANCE
PREMIER VALUE FIBER THERAPY
methylcellulose tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-122 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLCELLULOSE (4000 MPA.S) (UNII: MRJ667KA5E) (METHYLCELLULOSE (4000 MPA.S) - UNII:MRJ667KA5E) METHYLCELLULOSE (4000 MPA.S) 500 mg Inactive Ingredients Ingredient Name Strength ADIPIC ACID (UNII: 76A0JE0FKJ) CALCIUM CARBONATE (UNII: H0G9379FGK) CROSPOVIDONE (120 .MU.M) (UNII: 68401960MK) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color ORANGE (Light orange) Score no score Shape CAPSULE (Caplet) Size 19mm Flavor Imprint Code RP122 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-122-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/19/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 04/19/2019 Labeler - PHARMACY VALUE ALLIANCE, LLC (101668460)