TRILOCICLO- triamcinolone acetonide, ciclopirox
PureTek Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Trilociclo
THESE PRODUCTS ARE INTENDED ONLY FOR USE AS DESCRIBED. The individual products contained in this package should not be used alone or in combination for other purposes. The information described in this labeling concerns only the use of these products as indicated in this administration package.
DESCRIPTION
The topical corticosteroids constitute a class of primarily synthetic steroids used as anti- inflammatory and antipruritic agents. The steroids in this class include triamcinolone acetonide. Triamcinolone acetonide is designated chemically as 9-Fluoro-11β, 16α, 17,21-tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16,17-acetal with acetone. C 24H 31FO 6. M.W. 434.51; CAS Reg. No. 76-25-5.
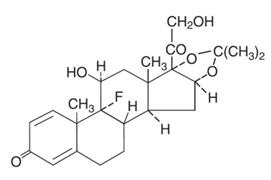
Each gram of Triamcinolone Acetonide Ointment USP, 0.1% contains 1mg triamcinolone acetonide in an ointment base of light mineral oil and white petrolatum.
CLINICAL PHARMACOLOGY
Topical corticosteroids share anti- inflammatory, antipruritic and vasoconstrictive actions. The mechanism of anti- inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Pharmacokinetics -
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids.Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses (see DOSAGE AND ADMINISTRATION).
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids.
Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
INDICATIONS AND USAGE
Triamcinolone Acetonide Ointment is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid responsive dermatoses.
CONTRAINDICATIONS
Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparations.
PRECAUTIONS
General
Systemic absorption of topical corticosteroids has produced reversible hypothalamic pituitary- adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria in some patients.
Conditions which augment systemic absorption include the application of the more potent steroids, use over large surface areas, prolonged use, and the addition of occlusive dressings.
Therefore, patients receiving a large dose of any potent topical steroid applied to a large surface area or under an occlusive dressing should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests, and for impairment of thermal homeostasis. If HPA axis suppression or elevation of the body temperature occurs, an attempt should be made to withdraw the drug, to reduce the frequency of application, substitute a less potent steroid, or use a sequential approach when utilizing the occlusive technique. Recovery of HPA axis function and thermal homeostasis are generally prompt and complete upon discontinuation of the drug. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids. Occasionally, a patient may develop a sensitivity reaction to a particular occlusive dressing material or adhesive and a substitute material may be necessary.
Children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity (see PRECAUTIONS, Pediatric Use). If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted.
In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, the corticosteroid should be discontinued until the infection has been adequately controlled.
These preparations are not for ophthalmic use.
Information for Patients
Patients using topical corticosteroids should receive the following information and instructions:
1. This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
2. Patients should be advised not to use this medication for any disorder other than for which it was prescribed.
3. The treated skin area should not be bandaged or otherwise covered or wrapped as to be occlusive unless directed by the physician.
4. Patients should report any signs of local adverse reactions especially under occlusive dressing.
5. Parents of pediatric patients should be advised not to use tight-fitting diapers or plastic pants on a child being treated in the diaper area, as these garments may constitute occlusive dressings.
Laboratory Tests
A urinary free cortisol test and ACTH stimulation test may be helpful in evaluating HPA axis suppression.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of topical corticosteroids.
Studies to determine mutagenicity with prednisolone and hydrocortisone showed negative results.
Pregnancy:
Teratogenic Effects:
Pregnancy Category C -
Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. The more potent corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well- controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids.Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
Nursing Mothers
Itis not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, caution should be exercised when topical corticosteroids are administered to a nursing woman.
Pediatric Use
Pediatric patients may demonstrate greater susceptibility to topical corticosteroid induced HPA axis suppression and Cushing’s syndrome than mature patients because of a larger skin surface area to body weight ratio.
HPA axis suppression, Cushing’s syndrome, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Administration of topical corticosteroids to children should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of children.
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings (reactions are listed in an approximate decreasing order of occurrence): burning, itching, irritation,dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria.
To report SUSPECTED ADVERSE REACTIONS, contact Teligent Pharma, Inc. at 1-856-697-1441, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
OVERDOSAGE
Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects (see PRECAUTIONS, General).
DOSAGE AND ADMINISTRATION
Apply a thin film to the affected area two to three times daily.
Occlusive Dressing Technique
Occlusive dressings may be used for the management of psoriasis or other recalcitrant conditions. Apply a thin film of ointment to the lesion, cover with a pliable nonporous film, and seal the edges. If needed, additional moisture may be provided by covering the lesion with a dampened clean cotton cloth before the nonporous film is applied or by briefly wetting the affected area with water immediately prior to applying the medication.
The frequency of changing dressings is best determined on an individual basis. It may be convenient to apply Triamcinolone Acetonide Ointment under an occlusive dressing in the evening and to remove the dressing in the morning (i.e., 12-hour occlusion). When utilizing the 12-hour occlusion regimen, additional ointment should be applied, without occlusion, during the day. Reapplication is essential at each dressing change.
If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
HOW SUPPLIED
Triamcinolone Acetonide Ointment USP, 0.1% is supplied in the following sizes:
15 g tube – NDC 52565-014-15
80 g tube – NDC 52565-014-80
1 lb (454 g) jar – NDC 52565-014-26
Ciclopirox Topical Solution, 8%
Nail Lacquer
Rx Only
For use on fingernails and toenails and immediately adjacent skin only. Not for use in eyes.
DESCRIPTION
Ciclopirox Topical Solution, 8% contains a synthetic antifungal agent, ciclopirox. It is intended for topical use on fingernails and toenails and immediately adjacent skin.
Each gram of Ciclopirox Topical Solution, 8% contains 80 mg ciclopirox in a solution base consisting of butyl ester of poly [vinylmethylether/maleic acid copolymer] in isopropyl alcohol, ethyl acetate, and isopropyl alcohol. Ethyl acetate and isopropyl alcohol are solvents that vaporize after application.
Ciclopirox Topical Solution, 8% is a clear, colorless to the slightly yellowish solution.
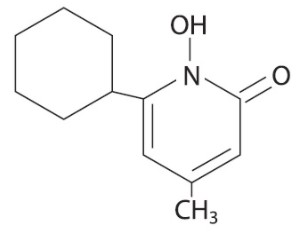
The chemical name for ciclopirox is 6-cyclohexyl-1-hydroxy-4-methyl-2(1H)-pyridone, with the empirical formula C 12H 17NO 2 and a molecular weight of 207.27.
The CAS Registry Number is [29342-05-0]. The chemical structure is:
CLINICAL PHARMACOLOGY
Microbiology
Mechanism of Action - The mechanism of action of ciclopirox has been investigated using various in vitro and in vivo infection models. One in vitro study suggested that ciclopirox acts by chelation of polyvalent cations (Fe +3 or Al +3) resulting in the inhibition of the metal-dependent enzymes that are responsible for the degradation of peroxides within the fungal cell. The clinical significance of this observation is not known.
Activity in vitro and ex vivo - In vitro methodologies employing various broth or solid media with and without additional nutrients have been utilized to determine ciclopirox minimum inhibitory concentration (MIC) values for the dermatophytic molds. (1-2) As a consequence, a broad range of MIC values, 1-20 ug/mL, were obtained for Trichophyton rubrum and Trichophyton mentagrophytes species. Correlation between in vitro MIC results and clinical outcome has yet to be established for ciclopirox.
One ex vivo study was conducted evaluating 8% ciclopirox against new and established Trichophyton rubrum and Trichophyton mentagrophytes infections in ovine hoof material. (3) After 10 days of treatment the growth of T. rubrum and T. mentagrophytes in the established infection model was very minimally affected. Elimination of the molds from hoof material was not achieved in either the new or established infection models.
Susceptibility testing for Trichophyton rubrum species - In vitro susceptibility testing methods for determining ciclopirox MIC values against the dermatophytic molds, including Trichophyton rubrum species, have not been standardized or validated. Ciclopirox MIC values will vary depending on the susceptibility testing method employed, composition and pH of media and the utilization of nutritional supplements. Breakpoints to determine whether clinical isolates of Trichophyton rubrum are susceptible or resistant to ciclopirox have not been established.
Resistance - Studies have not been conducted to evaluate drug resistance development in T. rubrum species exposed to 8% ciclopirox topical solution. Studies assessing cross-resistance to ciclopirox and other known antifungal agents have not been performed.
Antifungal Drug Interactions - No studies have been conducted to determine whether ciclopirox might reduce the effectiveness of systemic antifungal agents for onychomycosis. Therefore, the concomitant use of 8% ciclopirox topical solution and systemic antifungal agents for onychomycosis is not recommended.
Pharmacokinetics - As demonstrated in pharmacokinetic studies in animals and man, ciclopirox olamine is rapidly absorbed after oral administration and completely eliminated in all species via feces and urine. Most of the compound is excreted either unchanged or as glucuronide. After oral administration of 10 mg of radiolabeled drug (14C-ciclopirox) to healthy volunteers, approximately 96% of the radioactivity was excreted renally within 12 hours of administration. Ninety-four percent of the renally excreted radioactivity was in the form of glucuronides. Thus, glucuronidation is the main metabolic pathway of this compound.
Systemic absorption of ciclopirox was determined in five patients with dermatophytic onychomycoses, after application of ciclopirox topical solution, 8%, to all 20 digits and adjacent 5 mm of skin once daily for six months. Random serum concentrations and 24 hour urinary excretion of ciclopirox were determined at two weeks and at 1, 2, 4 and 6 months after initiation of treatment and four weeks post-treatment. In this study, ciclopirox serum levels ranged from 12-80 ng/mL. Based on urinary data, mean absorption of ciclopirox from the dosage form was <5% of the applied dose. One month after cessation of treatment, serum and urine levels of ciclopirox were below the limit of detection.
In two vehicle-controlled trials, patients applied ciclopirox topical solution, 8%, to all toenails and affected fingernails. Out of a total of 66 randomly selected patients on active treatment, 24 had detectable serum ciclopirox concentrations at some point during the dosing interval (range 10.0-24.6 ng/mL). It should be noted that 11 of these 24 patients took concomitant medication containing ciclopirox as ciclopirox olamine.
The penetration of the ciclopirox topical solution, 8% was evaluated in an in vitro investigation. Radiolabeled ciclopirox applied once to onychomycotic toenails that were avulsed demonstrated penetration up to a depth of approximately 0.4 mm. As expected, nail plate concentrations decreased as a function of nail depth. The clinical significance of these findings in nail plates is unknown. Nail bed concentrations were not determined.
INDICATIONS AND USAGE
(To understand fully the indication for this product, please read the entire INDICATIONS AND USAGE section of the labeling.)
Ciclopirox Topical Solution, 8% as a component of a comprehensive management program, is indicated as topical treatment in immunocompetent patients with mild to moderate onychomycosis of fingernails and toenails without lunula involvement, due to Trichophyton rubrum. The comprehensive management program includes removal of the unattached, infected nails as frequently as monthly, by a health care professional who has special competence in the diagnosis and treatment of nail disorders, including minor nail procedures.
- No studies have been conducted to determine whether ciclopirox might reduce the effectiveness of systemic antifungal agents for onychomycosis. Therefore, the concomitant use of 8% ciclopirox topical solution and systemic antifungal agents for onychomycosis, is not recommended.
- Ciclopirox Topical Solution, 8% should be used only under medical supervision as described above.
- The effectiveness and safety of ciclopirox topical solution, 8% in the following populations has not been studied. The clinical trials with use of ciclopirox topical solution, 8% excluded patients who: were pregnant or nursing, planned to become pregnant, had a history of immunosuppression (e.g., extensive, persistent, or unusual distribution of dermatomycoses, extensive seborrheic dermatitis, recent or recurring herpes zoster, or persistent herpes simplex), were HIV seropositive, received organ transplant, required medication to control epilepsy, were insulin dependent diabetics or had diabetic neuropathy. Patients with severe plantar (moccasin) tinea pedis were also excluded.
- The safety and efficacy of using Ciclopirox Topical Solution, 8% daily for greater than 48 weeks have not been established.
Clinical Trials Data -
The results of use of ciclopirox topical solution, 8% in treatment of onychomycosis of the toenail without lunula involvement were obtained from two double-blind, placebo-controlled studies conducted in the United States. In these studies, patients with onychomycosis of the great toenails without lunula involvement were treated with ciclopirox topical solution, 8% in conjunction with monthly removal of the unattached, infected toenail by the investigator. Ciclopirox topical solution, 8%, was applied for 48 weeks. At baseline, patients had 20–65% involvement of the target great toenail plate. Statistical significance was demonstrated in one of two studies for the endpoint "complete cure" (clear nail and negative mycology), and in two studies for the endpoint "almost clear" (≤10% nail involvement and negative mycology) at the end of study. These results are presented below.
|
At Week 48 (plus Last Observation Carried Forward) for the Intent-to-Treat (ITT) Population |
||||
|
Study 312 |
Study 313 |
|||
|
Active |
Vehicle |
Active |
Vehicle |
|
|
Complete Cure* |
6/110 (5.5%) |
1/109 (0.9%) |
10/118 (8.5%) |
0/117 (0%) |
|
Almost Clear** |
7/107 (6.5%) |
1/108 (0.9%) |
14/116 (12%) |
1/115 (0.9%) |
|
Negative Mycology Alone*** |
30/105 (29%) |
12/106 (11%) |
41/115 (36%) |
10/114 (9%) |
|
* Clear nail and negative mycology ** ≤ 10% nail involvement and negative mycology *** Negative KOH and negative culture |
||||
The summary of reported patient outcomes for the ITT population at 12 weeks following the end of treatment are presented below. Note that post-treatment efficacy assessments were scheduled only for patients who achieved a complete cure.
|
Post-Treatment Week 12 Data for Patients Who Achieved Complete Cure at Week 48 |
||||
|
Study 312 |
Study 313 |
|||
|
Active |
Vehicle |
Active |
Vehicle |
|
|
Number of Treated Patients |
112 |
111 |
119 |
118 |
|
Complete Cure at Week 48 |
6 |
1 |
10 |
0 |
|
Post-treatment Week 12 Outcomes: | ||||
|
Patients Missing All Week 12 Assessments |
2 |
0 |
2 |
0 |
|
Patients with Week 12 Assessments |
4 |
1 |
8 |
0 |
|
Complete Cure |
3 |
1 |
4 |
0 |
|
Almost Clear |
2* |
1 |
1* |
0 |
|
Negative Mycology |
3 |
1 |
5 |
0 |
*Four patients (from studies 312 and 313) who were completely cured did not have post-treatment Week 12 planimetry data.
CONTRAINDICATIONS
Ciclopirox Topical Solution, 8% is contraindicated in individuals who have shown hypersensitivity to any of its components.
WARNINGS
Ciclopirox Topical Solution, 8% is not for ophthalmic, oral, or intravaginal use. It is for use on nails and immediately adjacent skin only.
PRECAUTIONS
If a reaction suggesting sensitivity or chemical irritation should occur with the use of Ciclopirox Topical Solution, 8% treatment should be discontinued and appropriate therapy instituted. So far there is no relevant clinical experience with patients with insulin-dependent diabetes or who have diabetic neuropathy. The risk of removal of the unattached, infected nail, by the health care professional and trimming by the patient should be carefully considered before prescribing to patients with a history of insulin-dependent diabetes mellitus or diabetic neuropathy.
Information for Patients
Patients should have detailed instructions regarding the use of Ciclopirox Topical Solution, 8% as a component of a comprehensive management program for onychomycosis in order to achieve maximum benefit with the use of this product.
The patient should be told to:
1. Use Ciclopirox Topical Solution, 8% as directed by a health care professional. Avoid contact with the eyes and mucous membranes. Contact with skin other than skin immediately surrounding the treated nail(s) should be avoided. Ciclopirox Topical Solution, 8% is for external use only.
2. Ciclopirox Topical Solution, 8% should be applied evenly over the entire nail plate and 5 mm of surrounding skin. If possible, Ciclopirox Topical Solution, 8% should be applied to the nail bed, hyponychium, and the undersurface of the nail plate when it is free of the nail bed (e.g., onycholysis). Contact with the surrounding skin may produce mild, transient irritation (redness).
3. Removal of the unattached, infected nail, as frequently as monthly, by a health care professional is needed with use of this medication. Inform a health care professional if they have diabetes or problems with numbness in the toes or fingers for consideration of the appropriate nail management program.
4. Inform a health care professional if the area of application shows signs of increased irritation (redness, itching, burning, blistering, swelling, or oozing).
5. Up to 48 weeks of daily applications with Ciclopirox Topical Solution, 8% and professional removal of the unattached, infected nail, as frequently as monthly, are considered the full treatment needed to achieve a clear or almost clear nail (defined as 10% or less residual nail involvement).
6. Six months of therapy with professional removal of the unattached, infected nail may be required before initial improvement of symptoms is noticed.
7. A completely clear nail may not be achieved with use of this medication. In clinical studies less than 12% of patients were able to achieve either a completely clear or almost clear toenail.
8. Do not use the medication for any disorder other than that for which it is prescribed.
9. Do not use nail polish or other nail cosmetic products on the treated nails.
10. Avoid use near heat or open flame, because product is flammable.
Carcinogenesis, Mutagenesis, Impairment of Fertility -
No carcinogenicity study was conducted with ciclopirox topical solution, 8% formulation. A carcinogenicity study of ciclopirox (1% and 5% solutions in polyethylene glycol 400) in female mice dosed topically twice per week for 50 weeks followed by a six-month drug-free observation period prior to necropsy revealed no evidence of tumors at the application sites. In human systemic tolerability studies following daily application (~340 mg of ciclopirox topical solution, 8%) in subjects with distal subungual onychomycosis, the average maximal serum level of ciclopirox was 31±28 ng/mL after two months of once daily applications. This level was 159 times lower than the lowest toxic dose and 115 times lower than the highest nontoxic dose in rats and dogs fed 7.7 and 23.1 mg ciclopirox (as ciclopirox olamine)/kg/day.
The following in vitro genotoxicity tests have been conducted with ciclopirox: evaluation of gene mutation in Ames Salmonella and E. coli assays (negative); chromosome aberration assays in V79 Chinese hamster lung fibroblasts, with and without metabolic activation (positive); gene mutation assay in the HGPRT-test with V79 Chinese hamster lung fibroblasts (negative); unscheduled DNA synthesis in human A549 cells (negative); and BALB/c3T3 cell transformation assay (negative). In an in vivo Chinese hamster bone marrow cytogenetic assay, ciclopirox was negative for chromosome aberrations at 5,000 mg/kg.
The following in vitro genotoxicity tests were conducted with ciclopirox topical solution, 8%: Ames Salmonella test (negative); unscheduled DNA synthesis in the rat hepatocytes (negative); cell transformation assay in BALB/c3T3 cell assay (positive). The positive response of the lacquer formulation in the BALB/c3T3 test was attributed to its butyl monoester of poly[methylvinyl ether/maleic acid] resin component (Gantrez® ES-435), which also tested positive in this test. The cell transformation assay may have been confounded because of the film-forming nature of the resin.
Gantrez® ES-435 tested nonmutagenic in both the in vitro mouse lymphoma forward mutation assay with or without activation and unscheduled DNA synthesis assay in rat hepatocytes.
Oral reproduction studies in rats at doses up to 3.85 mg ciclopirox (as ciclopirox olamine)/kg/day [equivalent to approximately 1.4 times the potential exposure at the maximum recommended human topical dose (MRHTD)] did not reveal any specific effects on fertility or other reproductive parameters. MRHTD (mg/m2) is based on the assumption of 100% systemic absorption of 27.12 mg ciclopirox (~340 mg ciclopirox topical solution, 8%) that will cover all the fingernails and toenails including 5 mm proximal and lateral fold area plus onycholysis to a maximal extent of 50%.
Pregnancy:
Teratogenic Effects:
Pregnancy Category B -
Teratology studies in mice, rats, rabbits, and monkeys at oral doses of up to 77, 23, 23, or 38.5 mg, respectively, of ciclopirox as ciclopirox olamine/kg/day (14, 8, 17, and 28 times MRHTD), or in rats and rabbits receiving topical doses of up to 92.4 and 77 mg/kg/day, respectively (33 and 55 times MRHTD), did not indicate any significant fetal malformations.
There are no adequate or well-controlled studies of topically applied ciclopirox in pregnant women. Ciclopirox Topical Solution, 8% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers -
It is not known whether this drug is excreted in human milk. Since many drugs are excreted in human milk, caution should be exercised when Ciclopirox Topical Solution, 8% is administered to a nursing woman.
Pediatric Use -
Based on the safety profile in adults, Ciclopirox Topical Solution, 8% is considered safe for use in children 12 years and older. No clinical trials have been conducted in the pediatric population.
Geriatric Use -
In clinical studies of ciclopirox topical solution, 8% did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients.
ADVERSE REACTIONS
In the vehicle-controlled clinical trials conducted in the United States, 9% (30/327) of patients treated with ciclopirox topical solution, 8%, and 7% (23/328) of patients treated with vehicle reported treatment-emergent adverse events (TEAE) considered by the investigator to be causally related to the test material.
The incidence of these adverse events, within each body system, was similar between the treatment groups except for skin and appendages: 8% (27/327) and 4% (14/328) of subjects in the ciclopirox and vehicle groups reported at least one adverse event, respectively. The most common were rash-related adverse events: periungual erythema and erythema of the proximal nail fold were reported more frequently in patients treated with ciclopirox topical solution, 8%, (5% [16/327]) than in patients treated with vehicle (1% [3/328]). Other TEAEs thought to be causally related included nail disorders such as shape change, irritation, ingrown toenail, and discoloration.
The incidence of nail disorders was similar between the treatment groups (2% [6/327] in the ciclopirox topical solution, 8%, group and 2% [7/328] in the vehicle group). Moreover, application site reactions and/or burning of the skin occurred in 1% of patients treated with ciclopirox topical solution, 8%, (3/327) and vehicle (4/328).
A 21-Day Cumulative Irritancy study was conducted under conditions of semi-occlusion. Mild reactions were seen in 46% of patients with the ciclopirox topical solution, 8%, 32% with the vehicle and 2% with the negative control, but all were reactions of mild transient erythema. There was no evidence of allergic contact sensitization for either the ciclopirox topical solution, 8% or the vehicle base. In a separate study of the photosensitization potential of ciclopirox topical solution, 8% in a maximized test design that included the occluded application of sodium lauryl sulfate, no photoallergic reactions were noted. In four subjects localized allergic contact reactions were observed. In the vehicle-controlled studies, one patient treated with ciclopirox topical solution, 8% discontinued treatment due to a rash, localized to the palm (causal relation to test material undetermined).
Use of ciclopirox topical solution, 8% for 48 additional weeks was evaluated in an open-label extension study conducted in patients previously treated in the vehicle-controlled studies. Three percent (9/281) of subjects treated with ciclopirox topical solution, 8% experienced at least one TEAE that the investigator thought was causally related to the test material. Mild rash in the form of periungual erythema (1% [2/281]) and nail disorders (1% [4/281]) were the most frequently reported. Four patients discontinued therapy because of TEAEs. Two of the four had events considered to be related to test material: one patient's great toenail "broke away" and another had an elevated creatine phosphokinase level on Day 1 (after 48 weeks of treatment with the vehicle in the previous vehicle-controlled study).
DOSAGE AND ADMINISTRATION
Ciclopirox Topical Solution, 8% should be used as a component of a comprehensive management program for onychomycosis. Removal of the unattached, infected nail, as frequently as monthly, by a health care professional, weekly trimming by the patient, and daily application of the medication are all integral parts of this therapy. Careful consideration of the appropriate nail management program should be given to patients with diabetes (see PRECAUTIONS).
Nail Care By Health Care Professionals - Removal of the unattached, infected nail, as frequently as monthly, trimming of onycholytic nail, and filing of excess horny material should be performed by professionals trained in the treatment of nail disorders.
Nail Care By Patient – Patients should file away (with an emery board) loose nail material and trim nails, as required, or as directed by the health care professional, every seven days after Ciclopirox Topical Solution, 8% is removed with alcohol.
Ciclopirox Topical Solution, 8% should be applied once daily (preferably at bedtime or eight hours before washing) to all affected nails with the applicator brush provided. Ciclopirox Topical Solution, 8% should be applied evenly over the entire nail plate.
If possible, Ciclopirox Topical Solution, 8% should be applied to the nail bed, hyponychium, and the undersurface of the nail plate when it is free of the nail bed (e.g., onycholysis).
Ciclopirox Topical Solution, 8% should not be removed on a daily basis. Daily applications should be made over the previous coat and removed with alcohol every seven days. This cycle should be repeated throughout the duration of therapy.
HOW SUPPLIED
Ciclopirox Topical Solution, 8% is available as follows:
6.6 mL glass bottle with a screw cap fitted with a brush (NDC 45802-141-67)
References
1.Dittmar W., Lohaus G. 1973. HOE296, A new antimycotic compound with a broad antimicrobial spectrum. Arzneim-Forsch./ Drug Res. 23:670-674.
2. Niewerth et. al., 1998. Antimicrobial susceptibility testing of dermatophytes:
Comparison of the agar macrodilution and broth microdilution tests. Chemotherapy. 44:31-35.
3. Yang et. al. 1997. A new simulation model for studying in vitro topical penetration of antifungal drugs into hard keratin. J. Mycol. Med. 7:195-98.
Patient Instructions
Ciclopirox Topical Solution, 8%
Nail Lacquer
Rx Only
Patient Information and Instructions
Patients should have detailed instructions regarding the use of Ciclopirox Topical Solution, 8% as a component of a comprehensive management program for onychomycosis in order to achieve maximum benefit with the use of this product. Discuss your treatment plan with your health care professional for regular removal of the unattached, infected nail.
Before using this medication, tell your doctor if you:
- Are pregnant or nursing
- Are you an insulin-dependent diabetic or have diabetic neuropathy
- Have a history of immunosuppression
- Are immunocompromised (e.g., received an organ transplant, etc.)
- Require medication to control epilepsy
- Use or require topical corticosteroids on a repeated monthly basis
- Use steroid inhalers on a regular basis
Patient Information:
- Use Ciclopirox Topical Solution, 8% as directed by your health care professional.
- Ciclopirox Topical Solution, 8% is for external use only.
- Contact with skin other than skin immediately surrounding the treated nail(s) should be avoided.
- Avoid contact with the eyes and mucous membranes.
- Removal of the unattached, infected nail, as frequently as monthly, by your health care professional is needed with the use of this medication to obtain maximal benefit with the use of this product. If you have diabetes or problems with numbness in your toes or fingers, talk to your health care provider before trimming your nails or removing any nail material.
- Inform your health care professional if the area of application shows signs of increased irritation (redness, itching, burning, blistering, swelling, or oozing).
- Up to 48 weeks of daily applications with Ciclopirox Topical Solution, 8% and professional removal, as frequently as monthly, of the unattached, infected nail are considered the full treatment time to achieve a clear or almost clear nail (defined as 10% or less residual nail involvement). Six months of therapy with professional removal of the unattached, infected nail may be required before initial improvement of symptoms is noticed.
- A completely clear nail may not be achieved with the use of this medication. In clinical studies, less than 12% of patients were able to achieve either a clear or almost clear toenail.
- Do not use nail polish or other nail cosmetic products on the treated nails.
- Avoid use near heat or open flame, because the product is flammable.
Patient Instructions
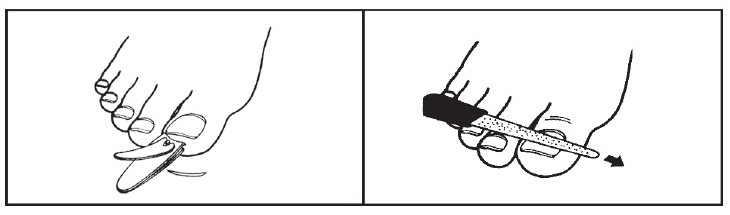
1- Before starting treatment, remove any loose nail or nail material using scissors, nail clippers or a nail file. If you have diabetes or problems with numbness in your toes or fingers, talk to your health care provider before trimming your nails or removing any nail material.
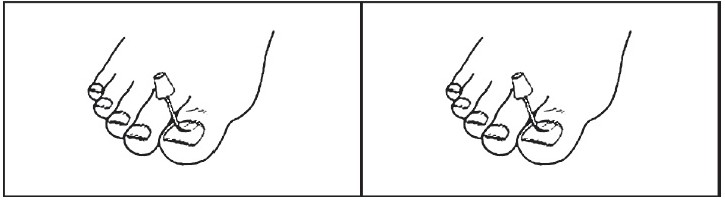
2- Apply Ciclopirox Topical Solution, 8% once daily (preferably at bedtime) to all affected nails with the applicator brush provided. Apply the lacquer evenly over the entire nail. Where possible, nail lacquer should also be applied to the underside of the nail and to the skin beneath it. Allow lacquer to dry (approximately 30 seconds) before putting on socks or stockings. After applying the medication, wait eight hours before taking a bath or shower.
3- Apply Ciclopirox Topical Solution, 8% daily over the previous coat.
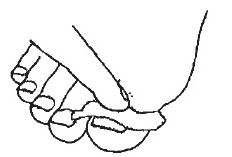
4- Once a week, remove the Ciclopirox Topical Solution, 8% with alcohol. Remove as much of the damaged nail as possible using scissors, nail clippers, or a nail file.
5- Repeat process (steps 2 through 4).
Please Note:
- To prevent the screw cap from sticking to the bottle, do not allow the solution to get into the bottle threads.
- To prevent the solution from drying out, the bottle should be closed tightly after every use.
- To protect from light, replace the bottle in the carton after each use.
Manufactured By
Perrigo
Bronx, NY 10457
Distributed By
Perrigo®
Allegan, MI 49010
Rev 02-17
: 3C000 RC J6
| TRILOCICLO
triamcinolone acetonide, ciclopirox kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - PureTek Corporation (785961046) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Perrigo New York Inc | 078846912 | manufacture(45802-141) | |
