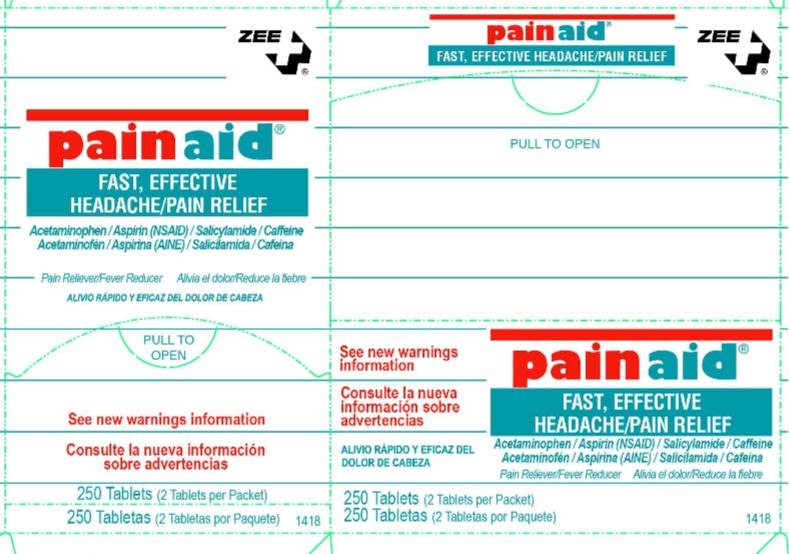
ZEE PAIN AID- acetaminophen salicylamide aspirin tablet
Cintas Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Zee Pain Aid
Active Ingredient/TabletAcetaminophen 110mg, Aspirin 162 mg-Salicylamide 152 mg,Pain Reviever Aid, Caffeine 32.5 mg-Pain Reliever Aid
Adults: Take 2 caplets every 6 hours, not to exceed 8 caplets in 24 hours
Children under 12 years, ask a doctor
Liver Warning: This product contains acetaminophen. Severe liver damage
may occur if you take:
• more than 8 tablets in 24 hours
• with other drugs containing acetaminophen (prescription or nonprescription). Ask a doctor or pharmacist before using with other drugs if you are not sure.
• 3 or more alcoholic drinks every day while using this product
______________________________________________________________
Do not use:
• with any other product containing acetaminophen this will provide more
than the recommended dose (overdose) of acetaminophen and could
cause serious health concerns.
• for more than 10 days for pain unless directed by a doctor
• for more than 3 days for fever unless directed by a doctor
• When using this product do not exceed recommended dose.
_______________________________________________________________
Stop use and ask a doctor if:
• symptoms do not improve
• pain or fever persists or gets worse
• new symptoms occur
• redness or swelling is present
KEEP OUT OF REACH OF CHILDREN. In case of overdose,
get medical help or contact a Poison Control Center right away.
Prompt medical attention is critical for adults as well as for
children even if you do not notice any signs or symptoms.
| ZEE PAIN AID
acetaminophen salicylamide aspirin tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Cintas Corporation (056481716) |
| Registrant - Cintas Corporation (056481716) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ultratab Laboratories, Inc. | 151051757 | manufacture(42961-042) | |