Label: KETOCONAZOLE cream
- NDC Code(s): 68071-2309-3
- Packager: NuCare Pharmaceuticals,Inc.
- This is a repackaged label.
- Source NDC Code(s): 0093-3219
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated January 14, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Ketoconazole Cream, 2% contains the broad-spectrum synthetic antifungal agent, ketoconazole 2%. Each gram, for topical administration, contains ketoconazole 20 mg and is formulated in an aqueous cream vehicle consisting of propylene glycol, purified water, cetyl alcohol, stearyl alcohol, isopropyl myristate, sorbitan monostearate, polysorbate 60, polysorbate 80, and sodium sulfite, anhydrous:
Ketoconazole is cis-1-acetyl-4-[4-[[2-(2,4-dichlorophenyl)-2-(1 H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazine and has the following structural formula:
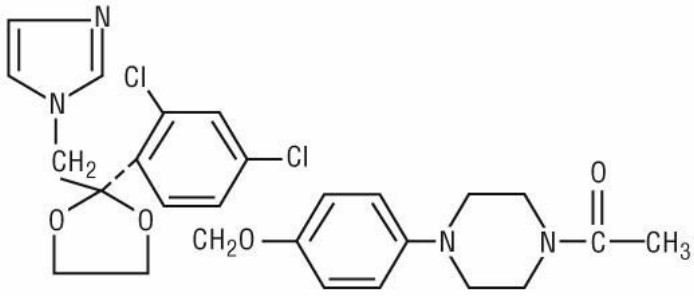
C 26H 28Cl 2N 4O 4 M.W. 531.44
-
CLINICAL PHARMACOLOGY
When ketoconazole cream, 2% was applied dermally to intact or abraded skin of Beagle dogs for 28 consecutive days at a dose of 80 mg, there were no detectable plasma levels using an assay method having a lower detection limit of 2 ng/ml.
After a single topical application to the chest, back and arms of normal volunteers, systemic absorption of ketoconazole was not detected at the 5 ng/ml level in blood over a 72-hour period.
Two dermal irritancy studies, a human sensitization test, a phototoxicity study and a photoallergy study conducted in 38 male and 62 female volunteers showed no contact sensitization of the delayed hypersensitivity type, no irritation, no phototoxicity and no photoallergenic potential due to ketoconazole cream, 2%.
Microbiology
Ketoconazole is a broad spectrum synthetic antifungal agent which inhibits the in vitro growth of the following common dermatophytes and yeasts by altering the permeability of the cell membrane: dermatophytes: Trichophyton rubrum, T. mentagrophytes, T. tonsurans, Microsporum canis, M. audouini, M. gypseum and Epidermophyton floccosum; yeasts: Candida albicans, Malassezia ovale (Pityrosporum ovale) and C. tropicalis; and the organism responsible for tinea versicolor, Malassezia furfur (Pityrosporum orbiculare). Only those organisms listed in the INDICATIONS AND USAGE Section have been proven to be clinically affected. Development of resistance to ketoconazole has not been reported.
-
INDICATIONS AND USAGE
Ketoconazole Cream, 2% is indicated for the topical treatment of tinea corporis, tinea cruris and tinea pedis caused by Trichophyton rubrum, T. mentagrophytes and Epidermophyton floccosum; in the treatment of tinea (pityriasis) versicolor caused by Malassezia furfur (Pityrosporum orbiculare); and in the treatment of cutaneous candidiasis caused by Candida spp.
- CONTRAINDICATIONS
-
WARNINGS
Ketoconazole Cream, 2% is not for opthalmic use.
Ketoconazole Cream, 2% contains sodium sulfite anhydrous, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
-
PRECAUTIONS
General
If a reaction suggesting sensitivity or chemical irritation should occur, use of the medication should be discontinued. Hepatitis (1:10,000 reported incidence) and, at high doses, lowered testosterone and ACTH induced corticosteroid serum levels have been seen with orally administered ketoconazole; these effects have not been seen with topical ketoconazole.
Carcinogenesis, Mutagenesis, Impairment of Fertility
A long-term feeding study in Swiss Albino mice and in Wistar rats showed no evidence of oncogenic activity. The dominant lethal mutation test in male and female mice revealed that single oral doses of ketoconazole as high as 80 mg/kg produced no mutation in any stage of germ cell development. The Ames’ Salmonella microsomal activator assay was also negative.
Pregnancy
Teratogenic effects
Pregnancy Category C
Ketoconazole has been shown to be teratogenic (syndactylia and oligodactylia) in the rat when given orally in the diet at 80 mg/kg/day, (10 times the maximum recommended human oral dose). However, these effects may be related to maternal toxicity, which was seen at this and higher dose levels.
There are no adequate and well-controlled studies in pregnant women. Ketoconazole should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
It is not known whether ketoconazole cream, 2% administered topically could result in sufficient systemic absorption to produce detectable quantities in breast milk. Nevertheless, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
-
ADVERSE REACTIONS
During clinical trials 45 (5%) of 905 patients treated with ketoconazole cream, 2% and 5 (2.4%) of 208 patients treated with placebo reported side effects consisting mainly of severe irritation, pruritus and stinging. One of the patients treated with ketoconazole cream developed a painful allergic reaction.
In worldwide postmarketing experience, rare reports of contact dermatitis have been associated with ketoconazole cream or one of its excipients, namely sodium sulfite or propylene glycol.
-
DOSAGE AND ADMINISTRATION
Cutaneous candidiasis, tinea corporis, tinea cruris, tinea pedis, and tinea (pityriasis) versicolor: It is recommended that ketoconazole cream, 2% be applied once daily to cover the affected and immediate surrounding area. Clinical improvement may be seen fairly soon after treatment is begun; however, candidal infections and tinea cruris and corporis should be treated for two weeks in order to reduce the possibility of recurrence. Patients with tinea versicolor usually require two weeks of treatment. Patients with tinea pedis require six weeks of treatment.
If a patient shows no clinical improvement after the treatment period, the diagnosis should be redetermined.
-
HOW SUPPLIED
Ketoconazole Cream, 2% is supplied in 15, 30, and 60 gram tubes.
30 g tube (NDC 68071-2309-3 )
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Manufactured In Croatia By:
Pliva Hrvatska d.o.o.
Zagreb, Croatia
Manufactured For:
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454
Rev. A 9/2018
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
KETOCONAZOLE
ketoconazole creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68071-2309(NDC:0093-3219) Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength KETOCONAZOLE (UNII: R9400W927I) (KETOCONAZOLE - UNII:R9400W927I) KETOCONAZOLE 20 mg in 1 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) CETYL ALCOHOL (UNII: 936JST6JCN) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) POLYSORBATE 60 (UNII: CAL22UVI4M) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SODIUM SULFITE (UNII: VTK01UQK3G) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68071-2309-3 1 in 1 CARTON 12/07/2020 1 30 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075581 02/08/2019 Labeler - NuCare Pharmaceuticals,Inc. (010632300) Establishment Name Address ID/FEI Business Operations NuCare Pharmaceuticals,Inc. 010632300 relabel(68071-2309)


