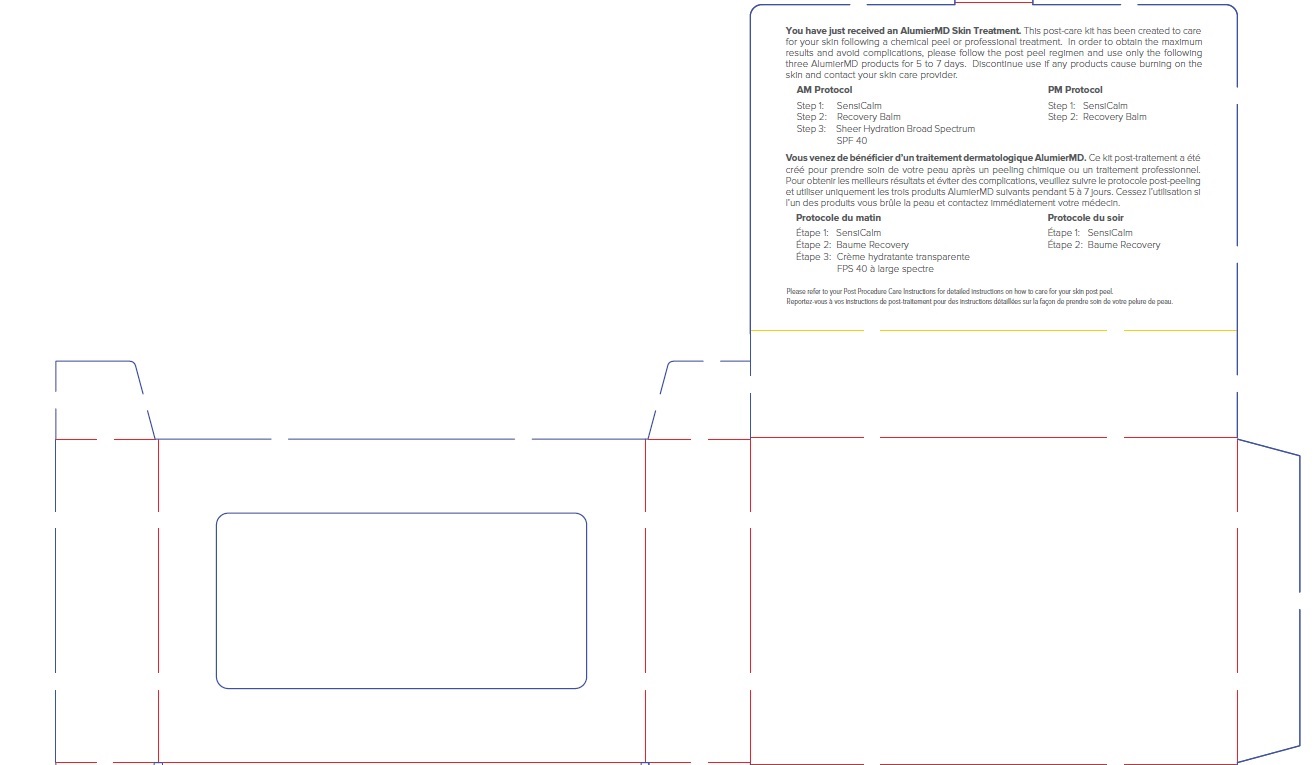
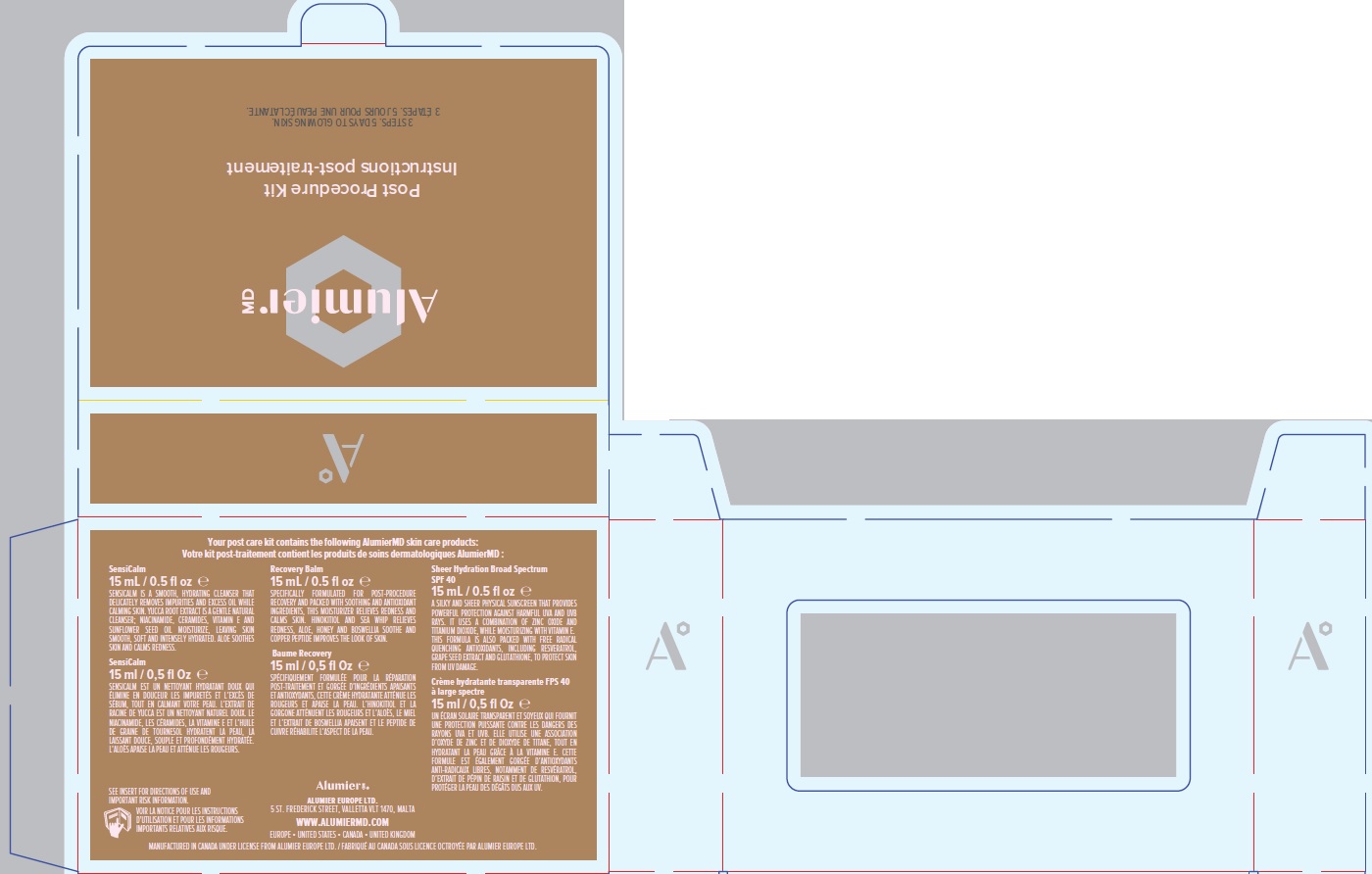
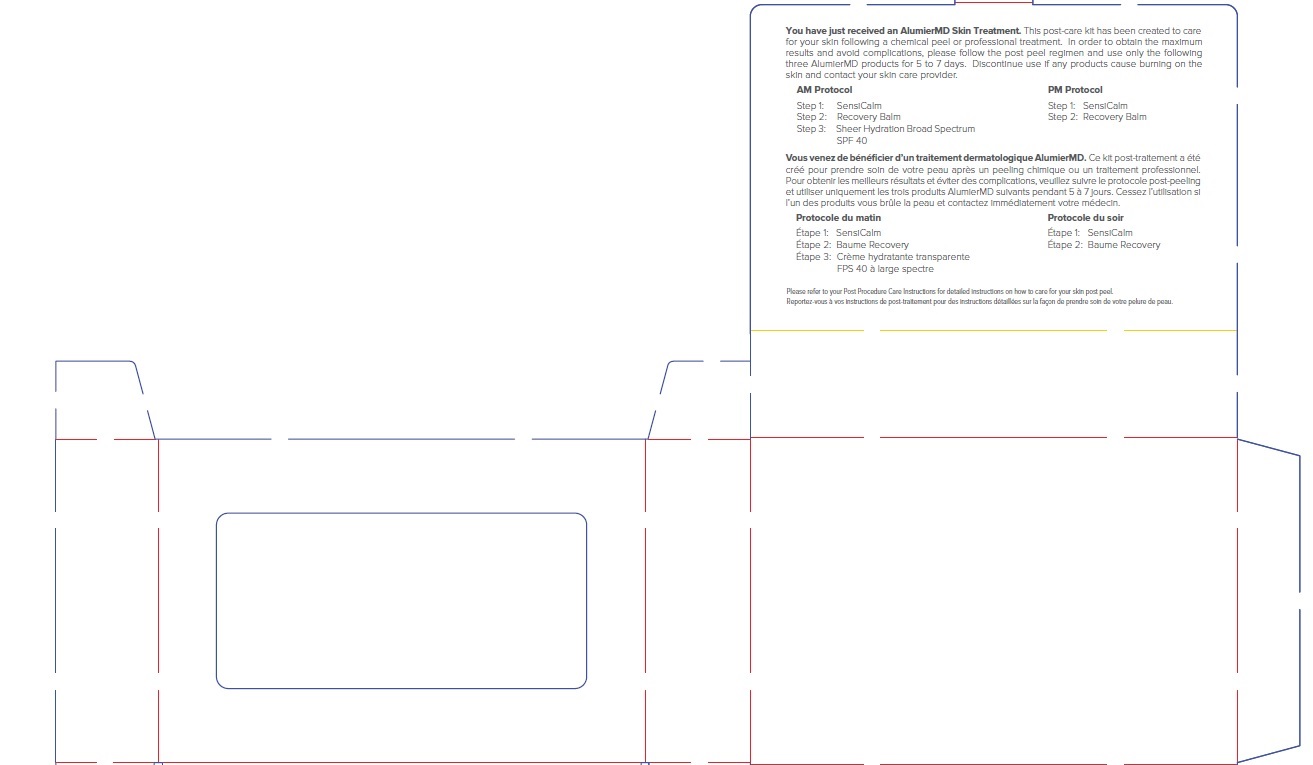
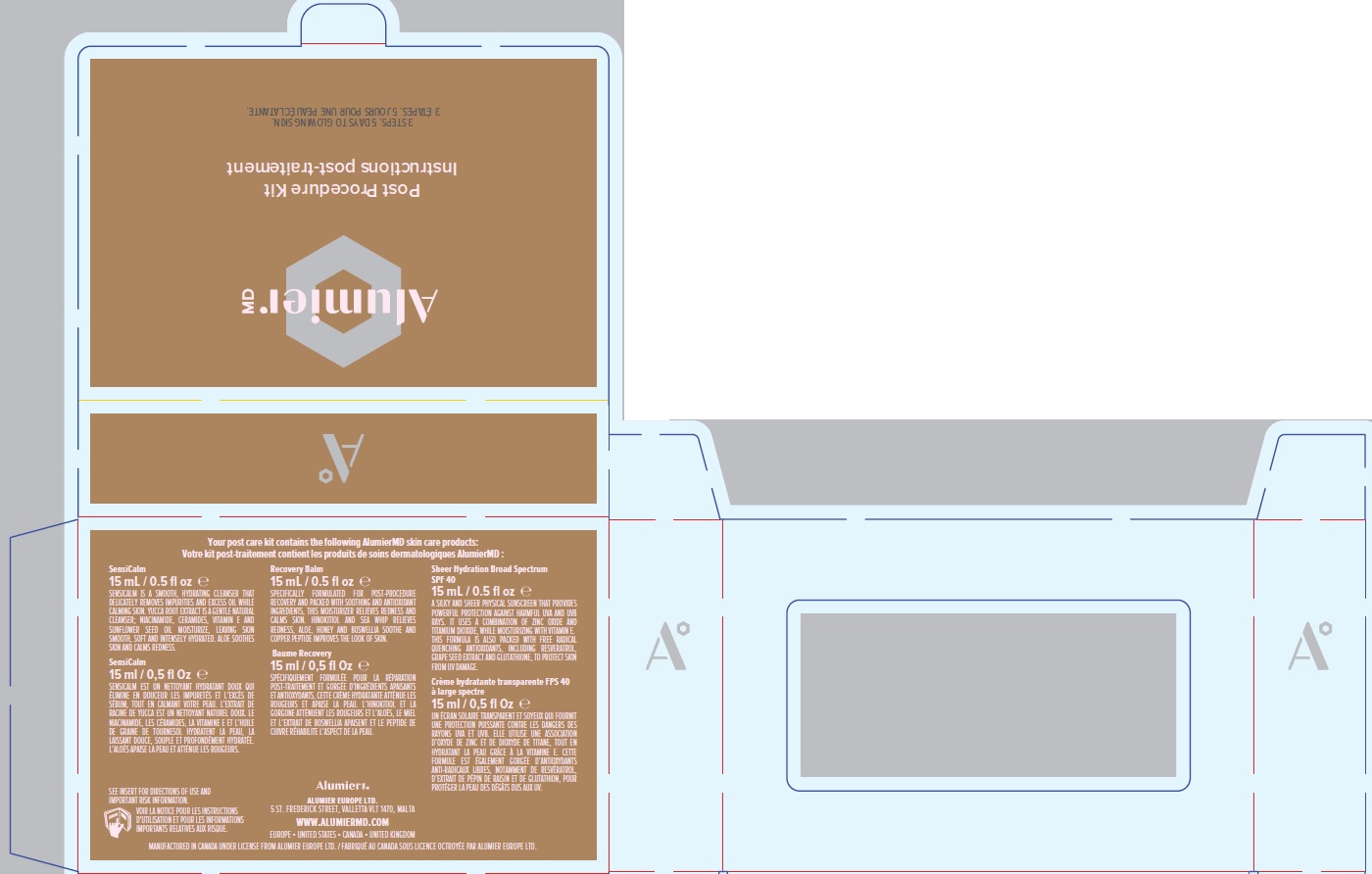
Label: ALUMIER MD POST PROCEDURE KIT- titanium dioxide, zinc oxide kit
- NDC Code(s): 69473-012-01
- Packager: Alumier Labs
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 8, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts - Sheer Hydration
- Active Ingredients
- Purpose
- Use
- Warnings
-
Directions
- apply liberally/generously (and evenly) 15 minutes before sun exposure
- reapply at least every 2 hours
- for use on children less than 6 months of age, consult a health care practitioner
- Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including: - limit time in the sun, especially from 10 a.m. - 2 p.m. - wear long-sleeved shirts, pants, hats and sunglasses
- use a water resistant sunscreen if swimming or sweating
- Other Information
-
Inactive Ingredients
Water/Aqua/Eau, C12-15 Alkyl Benzoate, Caprylic/Capric Triglyceride, Propanediol, Butyloctyl Salicylate, Cetearyl Alcohol, Aluminium Stearate, Glycerin, Glyceryl Stearate, PEG-100 Stearate, Lecithin (Syn. Phosphatidycholine), Vitis Vinifera Seed Extract, Resveratrol, Glutathione, Tocopheryl Acetate, Magnesium Ascorbyl Phosphate, Acacia Senegal Gum, Synthetic Wax, Polyhydroxysteric Acid, Ceteareth-20, Alumina, Microcrystalline Cellulose, Triethoxycaprylylsilane, Xanthan Gum, Cellulose Gum, C20-40 Pareth-10, Hydroxyethyl Behenamidopropyl Dimonium Chloride, Polyquatemium-67, Disodium EDTA, Ethylhexylglycerin, Phenoxyethanol.
- Company Information
- Product Packaging
-
INGREDIENTS AND APPEARANCE
ALUMIER MD POST PROCEDURE KIT
titanium dioxide, zinc oxide kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69473-012 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69473-012-01 1 in 1 CARTON; Type 0: Not a Combination Product 11/30/2020 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 15 mL Part 2 1 TUBE 15 mL Part 3 1 TUBE 15 mL Part 1 of 3 ALUMIER MD SHEER HYDRATION
titanium dioxide, zinc oxide lotionProduct Information Item Code (Source) NDC:69473-006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 62.8 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 70 mg in 1 mL Inactive Ingredients Ingredient Name Strength TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) WATER (UNII: 059QF0KO0R) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ACACIA (UNII: 5C5403N26O) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) HYDROXYETHYL BEHENAMIDOPROPYL DIMONIUM CHLORIDE (UNII: 9CO8OUL4TH) MAGNESIUM ASCORBYL PHOSPHATE (UNII: 0R822556M5) C20-40 PARETH-10 (UNII: TE3MZI4V3F) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) GLYCERIN (UNII: PDC6A3C0OX) VITIS VINIFERA SEED (UNII: C34U15ICXA) ALUMINUM MONOSTEARATE (UNII: P9BC99461E) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) XANTHAN GUM (UNII: TTV12P4NEE) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) PROPANEDIOL (UNII: 5965N8W85T) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) EDETATE DISODIUM (UNII: 7FLD91C86K) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PEG-100 STEARATE (UNII: YD01N1999R) ALUMINUM OXIDE (UNII: LMI26O6933) GLUTATHIONE (UNII: GAN16C9B8O) PHENOXYETHANOL (UNII: HIE492ZZ3T) RESVERATROL (UNII: Q369O8926L) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 15 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 11/30/2020 Part 2 of 3 ALUMIER MD SENSICALM
cleansing (cold creams, cleansing lotions, liquids, and pads) creamProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) INGR CETYL ALCOHOL (UNII: 936JST6JCN) INGR NIACINAMIDE (UNII: 25X51I8RD4) INGR ALLANTOIN (UNII: 344S277G0Z) INGR .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) INGR P-ANISIC ACID (UNII: 4SB6Y7DMM3) INGR XANTHAN GUM (UNII: TTV12P4NEE) INGR SODIUM HYDROXIDE (UNII: 55X04QC32I) INGR CAPRYLYL/CAPRYL OLIGOGLUCOSIDE (UNII: E00JL9G9K0) INGR XYLITOL (UNII: VCQ006KQ1E) INGR CAPRYLIC ACID (UNII: OBL58JN025) INGR 4-TERPINEOL, (+/-)- (UNII: L65MV77ZG6) INGR EDETATE SODIUM (UNII: MP1J8420LU) INGR EDETATE DISODIUM (UNII: 7FLD91C86K) INGR COCAMIDOPROPYL HYDROXYSULTAINE (UNII: 62V75NI93W) INGR WATER (UNII: 059QF0KO0R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR PROPANEDIOL (UNII: 5965N8W85T) INGR SODIUM ACRYLATE/SODIUM ACRYLOYLDIMETHYLTAURATE COPOLYMER (4000000 MW) (UNII: 1DXE3F3OZX) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR YUCCA SCHIDIGERA ROOT (UNII: E2H9ET15AT) INGR SUNFLOWER OIL (UNII: 3W1JG795YI) INGR LINALYL ACETATE (UNII: 5K47SSQ51G) INGR .BETA.-OCIMENE, (3Z)- (UNII: 472UVP4R7T) INGR SODIUM CHLORIDE (UNII: 451W47IQ8X) INGR DISODIUM COCOYL GLUTAMATE (UNII: MBK0CP8F5A) INGR CAPRYLYL GLYCOL (UNII: 00YIU5438U) INGR LINALOOL, (+/-)- (UNII: D81QY6I88E) INGR SORBITAN MONOOLEATE (UNII: 06XEA2VD56) INGR CERAMIDE NG (UNII: C04977SRJ5) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 15 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 07/01/2019 Part 3 of 3 ALUMIER MD RECOVERY BALM
face and neck (excluding shaving preparations) creamProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR DIMETHICONE (UNII: 92RU3N3Y1O) INGR DOCOSANOL (UNII: 9G1OE216XY) INGR COCOA BUTTER (UNII: 512OYT1CRR) INGR NIACINAMIDE (UNII: 25X51I8RD4) INGR PREZATIDE COPPER (UNII: 6BJQ43T1I9) INGR INDIAN FRANKINCENSE (UNII: 4PW41QCO2M) INGR HONEY (UNII: Y9H1V576FH) INGR CHAMAEMELUM NOBILE FLOWER (UNII: O2T154T6OG) INGR PATCHOULI OIL (UNII: F3IN55X5PO) INGR ROSEMARY (UNII: IJ67X351P9) INGR EDETATE DISODIUM (UNII: 7FLD91C86K) INGR TRICAPRYLIN (UNII: 6P92858988) INGR XYLITOL (UNII: VCQ006KQ1E) INGR SODIUM METHYL STEAROYL TAURATE (UNII: JFM219LJ55) INGR XANTHAN GUM (UNII: TTV12P4NEE) INGR PENTYLENE GLYCOL (UNII: 50C1307PZG) INGR ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) INGR CRANBERRY (UNII: 0MVO31Q3QS) INGR MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) INGR PROPANEDIOL (UNII: 5965N8W85T) INGR SHEA BUTTER (UNII: K49155WL9Y) INGR JOJOBA OIL (UNII: 724GKU717M) INGR .BETA.-THUJAPLICIN (UNII: U5335D6EBI) INGR RESVERATROL (UNII: Q369O8926L) INGR LAVANDULA ANGUSTIFOLIA FLOWERING TOP (UNII: 9YT4B71U8P) INGR ASPALATHUS LINEARIS LEAF (UNII: H7UGK1GJCU) INGR EUCALYPTUS GLOBULUS LEAF (UNII: S546YLW6E6) INGR MENTHA SPICATA (UNII: O2H83I4PUN) INGR SQUALANE (UNII: GW89575KF9) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR GREEN TEA LEAF (UNII: W2ZU1RY8B0) INGR APPLE (UNII: B423VGH5S9) INGR PSEUDOPTEROGORGIA ELISABETHAE (UNII: UDY3H1OUX5) INGR POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) INGR SODIUM HYDROXIDE (UNII: 55X04QC32I) INGR CAPRYLYL GLYCOL (UNII: 00YIU5438U) INGR CAPRYLIC ACID (UNII: OBL58JN025) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 15 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 07/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 11/30/2020 Labeler - Alumier Labs (079603173)