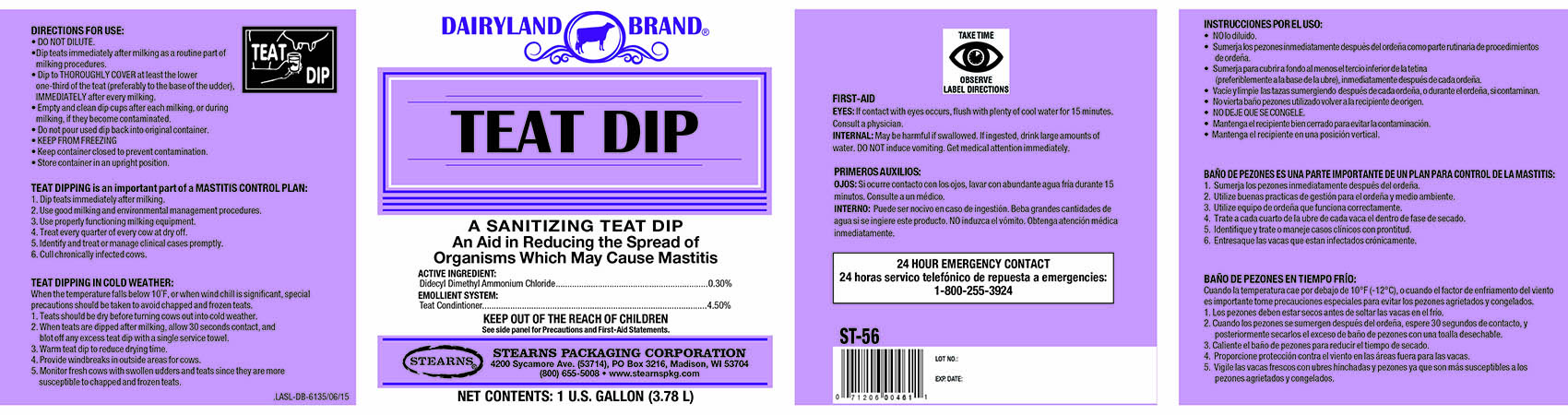
DAIRYLAND BRAND TEAT DIP- didecyldimethylammonium chloride liquid
Stearns Packaging Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Teat Dip
TEAT DIP
TAKE TIME
OBSERVE
LABEL DIRECTIONS
TEAT DIPPING is an important part of a MASTITIS CONTROL PLAN:
1. Dip teats immediately after milking.
2. Use good milking and environmental management procedures.
3. Use properly functioning milking equipment.
4. Treat every quarter of every cow at dry off.
5. Identify and treat or manage clinical cases promptly.
6. Cull chronically infected cows.
TEAT DIPPING IN COLD WEATHER:
When the temperature falls below 10 degrees F, or when wind chill is significant, special
precautions should be taken to avoid chapped and frozen teats.
1. Teats should be dry before turning cows out into cold weather.
2. When teats are dipped after milking, allow 30 seconds contact, and
blot off any excess teat dip with a single service towel.
3. Warm the teat dip to reduce drying time.
4. Provide windbreaks in outside areas for cows.
5. Monitor fresh cows with swollen udders and teats since they are more
susceptible to chapped and frozen teats.
DIRECTIONS FOR USE:
• DO NOT DILUTE.
• Dip teats immediately after milking as a routine part of
milking procedures.
• Dip to THOROUGHLY COVER at least the lower
one-third of the teat(preferably to the base of the udder),
IMMEDIATELY after every milking.
• Empty and clean dip cups after each milking, or during
milking, if they become contaminated.
• Do not pour used dip back into original container.
• KEEP FROM FREEZING
• Keep container closed to prevent contamination.
• Store container in an upright position.
FIRST-AID
EYES: If contact with eyes occurs, flush with plenty of cool water for 15 minutes.
Consult a physician.
INTERNAL: May be harmful if swallowed. If ingested, drink large amounts ofwater. DO NOT induce vomiting. Get medical attention immediately.
24 HOUR EMERGENCY CONTACT
1-800-255-3924
DAIRYLAND BRAND
TEAT DIP
A SANITIZING TEAT DIP
An Aid in Reducing the Spread of
Organisms Which May Cause Mastitis
ACTIVE INGREDIENT:
Didecyl Dimethyl Ammonium Chloride................................ 0.30%
EMOLLIENT SYSTEM:
Teat Conditioner............................................................... 4.50%
KEEP OUT OF REACH OF CHILDREN
See side panel for Precautions and First-Aid Statements
STEARNS PACKAGING CORPORATION
4200 Sycamore Ave. (53714), PO Box 3216, Madison, WI 53704
(800) 655-5008 • www.stearnspkg.com
NET CONTENTS: 1 U.S. FL. GALLON (3.78 L)
| DAIRYLAND BRAND TEAT DIP
didecyldimethylammonium chloride liquid |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Stearns Packaging Corporation (006069256) |
| Registrant - Stearns Packaging Corporation (006069256) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Stearns Packaging Corporation | 006069256 | manufacture, api manufacture | |