REPAGLINIDE- repaglinide tablet
Sandoz Inc
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use REPAGLINIDE TABLETS safely and effectively. See full prescribing information for REPAGLINIDE TABLETS.
REPAGLINIDE tablets, for oral use Initial U.S. Approval: 1997 RECENT MAJOR CHANGES
Dosage and Administration (2.3) 2/2017 INDICATIONS AND USAGEDOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSTablets: 0.5 mg, 1 mg, 2 mg (3) CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions (5% or greater incidence) among patients treated with repaglinide were: hypoglycemia, upper respiratory infection, headache, sinusitis, arthralgia, nausea, diarrhea, and back pain. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION. Revised: 2/2017 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Repaglinide tablets, USP are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Limitation of Use
Repaglinide should not be used in patients with type 1 diabetes mellitus or for the treatment of diabetic ketoacidosis.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Administration
The recommended starting dose for patients whose HbA1C is less than 8% is 0.5 mg orally before each meal. For patients whose HbA1C is 8% or greater the starting dose is 1 or 2 mg orally before each meal.
The recommended dose range is 0.5 mg to 4 mg before meals, with a maximum daily dose of 16 mg. The patient’s dose should be doubled up to 4 mg with each meal until satisfactory glycemic control is achieved. At least one week should elapse to assess response after each dose adjustment.
Instruct patients to take repaglinide within 30 minutes before meals. Repaglinide may be dosed 2, 3, or 4 times a day in response to changes in the patient’s meal pattern.
In patients who skip meals, instruct patients to skip the scheduled dose of repaglinide to reduce the risk of hypoglycemia. In patients who experience hypoglycemia, the dose of repaglinide should be reduced [see Warnings and Precautions (5.1)].
2.2 Patients with Severe Renal Impairment
In patients with severe renal impairment (CrCl=20–40 mL/min) initiate repaglinide 0.5 mg orally before each meal. Gradually titrate the dose, if needed to achieve glycemic control.
2.3 Dose Modifications for Drug Interactions
Dosage adjustments are recommended in patients taking concomitant strong CYP3A4 or CYP2C8 inhibitors or strong CYP3A4 or CYP2C8 inducers [see Drug Interactions (7), Clinical Pharmacology (12.3)].
Concomitant use with gemfibrozil is contraindicated [see Contraindications (4)].
Avoid concomitant use of repaglinide with clopidogrel. If concomitant use cannot be avoided, initiate repaglinide at 0.5 mg before each meal and do not exceed a total daily dose of 4 mg [see Drug Interactions (7), Clinical Pharmacology (12.3)].
Do not exceed a total daily dose of 6 mg of repaglinide in patients receiving cyclosporine [see Drug Interactions (7), Clinical Pharmacology (12.3)].
3 DOSAGE FORMS AND STRENGTHS
- •
- 0.5 mg tablets (white to off-white colored, round tablet debossed with ‘SZ’ on one side and ‘361’on the other side).
- •
- 1 mg tablets (yellow to pale yellow colored, slightly mottled, round tablet debossed with ‘SZ’ on one side and ‘362’ on the other side).
- •
- 2 mg tablets (yellow to pale yellow colored, slightly mottled, capsule shaped tablet debossed with ‘SZ’ on one side and ‘363’ on the other side).
4 CONTRAINDICATIONS
Repaglinide is contraindicated in patients with:
- •
- Concomitant use of gemfibrozil [see Drug Interactions (7)].
- •
- Known hypersensitivity to repaglinide or any inactive ingredients.
5 WARNINGS AND PRECAUTIONS
5.1 Hypoglycemia
All glinides, including Repaglinide, can cause hypoglycemia [see Adverse Reactions (6.1)]. Severe hypoglycemia can cause seizures, may be life-threatening, or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place an individual and others at risk in situations where these abilities are important (e.g., driving or operating other machinery).
Hypoglycemia can happen suddenly and symptoms may differ in each individual and change over time in the same individual. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic nerve disease, in patients using medications that block the sympathetic nervous system (e.g., beta-blockers) [see Drug Interactions (7)], or in patients who experience recurrent hypoglycemia.
Factors which may increase the risk of hypoglycemia include changes in meal pattern (e.g., macronutrient content), changes in level of physical activity, changes to co-administered medication [see Drug Interactions (7)], and concomitant use with other antidiabetic agents. Patients with renal or hepatic impairment may be at higher risk of hypoglycemia [see Use in Specific Populations (8.6, 8.7)].
Patients should administer repaglinide before meals and be instructed to skip the dose of repaglinide if a meal is skipped. In patients who experience hypoglycemia, the dose of repaglinide should be reduced [see Dosage and Administration (2.1)]. Patients and caregivers must be educated to recognize and manage hypoglycemia. Self-monitoring of blood glucose plays an essential role in the prevention and management of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended.
5.2 Serious Cardiovascular Adverse Reactions with Concomitant Use with NPH-insulin
Across seven controlled trials, there were six serious adverse events of myocardial ischemia in patients treated with repaglinide plus NPH-insulin from two studies, and one event in patients using insulin formulations alone from another study [See Adverse Reactions (6.1)]. Repaglinide is not indicated for use in combination with NPH-insulin.
6 ADVERSE REACTIONS
The following serious adverse reaction is also described elsewhere in the labeling:
- Hypoglycemia [see Warnings and Precautions (5.1)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying designs, the adverse reaction rates reported in one clinical trial may not be easily compared to those rates reported in another clinical trial, and may not reflect the rates actually observed in clinical practice.
Repaglinide has been administered to 2931 individuals during clinical trials. Approximately 1500 of these individuals with type 2 diabetes have been treated for at least 3 months, 1000 for at least 6 months, and 800 for at least 1 year. The majority of these individuals (1228) received repaglinide in one of five 1-year, active-controlled trials. Over one year, 13% of repaglinide patients were discontinued due to adverse reactions. The most common adverse reactions leading to withdrawal were hyperglycemia, hypoglycemia, and related symptoms.
Table 1 lists the common adverse reactions for repaglinide patients compared to placebo in trials 12 to 24 weeks duration.
- Table 1 Adverse Reactions (%) occurring ≥2% in Repaglinide Treated Patients from Pool of 12 to 24 Week Placebo Controlled Trials*
|
Respiratory |
Repaglinide N=352 |
Placebo N=108 |
|
Upper Respiratory Infection |
16 |
8 |
|
Headache |
11 |
10 |
|
Sinusits |
6 |
2 |
|
Arthralgia |
6 |
3 |
|
Nausea |
5 |
5 |
|
Diarrhea |
5 |
2 |
|
Back Pain |
5 |
4 |
|
Rhinitis |
3 |
3 |
|
Constipation |
3 |
2 |
|
Vomiting |
3 |
3 |
|
Paresthesia |
3 |
3 |
|
Chest pain |
3 |
1 |
|
Bronchitis |
2 |
1 |
|
Dyspepsia |
2 |
2 |
|
Urinary tract infection |
2 |
1 |
|
Tooth disorder |
2 |
0 |
|
Allergy |
2 |
0 |
* See trial descriptions in Clinical Trials (14)
Hypoglycemia
In clinical trials with repaglinide, hypoglycemia is the most commonly observed adverse reaction. Mild or moderate hypoglycemia occurred in 31% of repaglinide treated patients and 7% of placebo treated patients [see Warnings and Precautions (5.1]).
Hypoglycemia was reported in 16% of 1228 repaglinide patients, 20% of 417 glyburide patients, and 19% of 81 glipizide patients in 1‑year controlled trials. Of repaglinide-treated patients with symptomatic hypoglycemia, none developed coma or required hospitalization.
In a 24-week placebo controlled trial, patients who were naïve to oral hypoglycemic agent therapy and patients with a HbA1C below 8% at baseline had a higher frequency of hypoglycemia.
Weight Gain
There was no average gain in body weight when patients previously treated with oral hypoglycemic agents were switched to repaglinide. The average weight gain in patients treated with repaglinide and not previously treated with sulfonylurea drugs was 3.3%.
Cardiovascular Events
The incidence of total serious cardiovascular adverse events, including ischemia, was higher for repaglinide (51/1228 or 4%) than for sulfonylurea drugs (13/498 or 3%) in controlled comparator clinical trials.
- Table 2 Summary of Serious Cardiovascular Events in Trials Comparing Repaglinide to Sulfonylureas (% of total patients with events)
|
||
|
Repaglinide |
SU* |
|
|
Total Exposed |
1228 |
498 |
|
Serious CV Events |
4% |
3% |
|
Cardiac Ischemic Events |
2% |
2% |
|
Deaths due to CV Events |
0.5% |
0.4% |
Seven controlled clinical trials included repaglinide combination therapy with NPH-insulin (n=431), insulin formulations alone (n=388) or other combinations (sulfonylurea plus NPH-insulin or repaglinide plus metformin) (n=120). There were six serious adverse events of myocardial ischemia in patients treated with repaglinide plus NPH-insulin from two studies, and one event in patients using insulin formulations alone from another study [see Warnings and Precautions (5.3)].
Combination Therapy with Thiazolidinediones
Hypoglycemia
During 24-week treatment clinical trials of repaglinide-rosiglitazone or repaglinide-pioglitazone combination therapy (a total of 250 patients in combination therapy), hypoglycemia (blood glucose < 50 mg/dL) occurred in 7% of patients in combination therapy compared to 7% for repaglinide monotherapy, and 2% for thiazolidinedione monotherapy.
Peripheral Edema and Heart Failure
Peripheral edema was reported in 12 out of 250 (4.8%) repaglinide-thiazolidinedione combination therapy patients and 3 out of 124 (2.4%) thiazolidinedione monotherapy patients, with no cases reported in these trials for repaglinide monotherapy. There were reports in 2 of 250 patients (0.8%) treated with repaglinide-thiazolidinedione therapy of episodes of edema with congestive heart failure. Both patients had a prior history of coronary artery disease and recovered after treatment with diuretic agents. No comparable cases in the monotherapy treatment groups were reported.
Weight Gain
Mean weight increases associated with combination, repaglinide and pioglitazone therapy were 5.5 kg, 0.3 kg, and 2.0 kg respectively. Mean weight increases associated with combination, repaglinide and rosiglitazone therapy were 4.5 kg, 1.3 kg, and 3.3 kg respectively.
Infrequent Adverse Events (<1% of Patients)
Less common adverse clinical or laboratory events observed in clinical trials included elevated liver enzymes, thrombocytopenia, leukopenia, and anaphylactoid reactions.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post approval use of repaglinide. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or a causal relationship to drug exposure.
- •
- Alopecia
- •
- Hemolytic anemia
- •
- Pancreatitis
- •
- Stevens-Johnson Syndrome
- •
- Severe hepatic dysfunction including jaundice and hepatitis
7 DRUG INTERACTIONS
Clinically Important Drug Interactions with Repaglinide
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with repaglinide and instructions for preventing or managing them.
Table 3 Clinically Important Drug Interactions with Repaglinide
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C.
There are no adequate and well-controlled studies in pregnant women. It is unknown whether repaglinide can cause fetal harm when administered to a pregnant woman. Repaglinide should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Repaglinide was not teratogenic in rats or rabbits at doses 40 times (rats) and approximately 0.8 times (rabbit) clinical exposure (on a mg/m2 basis) throughout pregnancy. Offspring of rat dams exposed to repaglinide at 15 times clinical exposure on a mg/m2 basis during days 17 to 22 of gestation and during lactation developed nonteratogenic skeletal deformities consisting of shortening, thickening, and bending of the humerus during the postnatal period. This effect was not seen at doses up to 2.5 times clinical exposure (on a mg/m2 basis) on days 1 to 22 of pregnancy or at higher doses given during days 1 to 16 of pregnancy. Relevant human exposure has not occurred to date and therefore the safety of repaglinide administration throughout pregnancy or lactation cannot be established.
8.3 Nursing Mothers
Although it is not known whether repaglinide is excreted in human milk some oral agents are known to be excreted by this route.
Because the potential for hypoglycemia in nursing infants may exist, and because of the effects on nursing animals, a decision should be made as to whether repaglinide should be discontinued in nursing mothers, or if mothers should discontinue nursing. If repaglinide is discontinued and if diet alone is inadequate for controlling blood glucose, insulin therapy should be considered. In rat reproduction studies, measurable levels of repaglinide were detected in the breast milk of the dams and lowered blood glucose levels were observed in the pups. Cross fostering studies indicated that skeletal changes [see Use in Specific Populations (8.1)] could be induced in control pups nursed by treated dams, although this occurred to a lesser degree than those pups treated in utero.
8.5 Geriatric Use
In clinical studies of 24 weeks or greater duration, 415 patients were over 65 years of age and no patients were greater than 75 years of age. In one-year, active-controlled trials, no differences were seen in effectiveness or adverse events between these subjects and those less than 65. There was no increase in frequency or severity of hypoglycemia in older subjects, but greater sensitivity of some older individuals to repaglinide therapy cannot be ruled out.
8.6 Renal Impairment
Pharmacokinetic studies of repaglinide were conducted in patients with mild to moderate renal function impairment (CrCl=40–80 mL/min), and severe renal function impairment (CrCl=20–40 mL/min). Initial dose adjustment is not required in patients with mild to moderate renal dysfunction. However, patients with severe renal function impairment should initiate repaglinide therapy with the 0.5 mg dose and be carefully titrated [see Dosage and Administration (2.2)].
Studies were not conducted in patients with creatinine clearances below 20 mL/min or patients with renal failure requiring hemodialysis.
8.7 Hepatic Impairment
A single-dose study was conducted 12 patients with chronic liver disease. Patients with moderate to severe impairment of liver function had higher and more prolonged serum concentrations. Therefore, repaglinide should be used cautiously in patients with impaired liver function. Longer intervals between dose adjustments may be needed to allow full assessment of response.
10 OVERDOSAGE
Severe hypoglycemic reactions with coma, seizure, or other neurological impairment may occur and constitute medical emergencies requiring immediate hospitalization. Hypoglycemic symptoms without loss of consciousness or neurologic findings should be treated aggressively with oral glucose and adjustments in drug dosage and/or meal patterns. Close monitoring may continue until the physician is assured that the patient is out of danger. Patients should be closely monitored for a minimum of 24 to 48 hours, since hypoglycemia may recur after apparent clinical recovery. There is no evidence that repaglinide is dialyzable using hemodialysis.
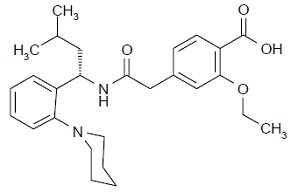
11 DESCRIPTION
Repaglinide is an oral blood glucose-lowering drug of the glinide class used in the management of type 2 diabetes mellitus (also known as non-insulin dependent diabetes mellitus or NIDDM). Repaglinide, S(+)2-ethoxy-4(2((3-methyl-1-(2-(1-piperidinyl) phenyl)-butyl) amino)-2-oxoethyl) benzoic acid, is chemically unrelated to the oral sulfonylurea insulin secretagogues.
The structural formula is as shown below:
Repaglinide is a white to off-white powder with molecular formula C27 H36 N2 O4 and a molecular weight of 452.6.
Repaglinide tablets, USP contain 0.5 mg, 1 mg, or 2 mg of repaglinide. In addition each tablet contains the following inactive ingredients: corn starch, dibasic calcium phosphate, glycerin, magnesium stearate, meglumine, microcrystalline cellulose, poloxamer 188, povidone, polacrilin potassium and talc. The 1 mg and 2 mg tablets contain ferric oxide yellow as coloring agent.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Repaglinide lowers blood glucose levels by stimulating the release of insulin from the pancreas. This action is dependent upon functioning beta (ß) cells in the pancreatic islets. Insulin release is glucose-dependent and diminishes at low glucose concentrations.
Repaglinide closes ATP-dependent potassium channels in the ß-cell membrane by binding at characterizable sites. This potassium channel blockade depolarizes the ß-cell, which leads to an opening of calcium channels. The resulting increased calcium influx induces insulin secretion. The ion channel mechanism is highly tissue selective with low affinity for heart and skeletal muscle.
12.2 Pharmacodynamics
A four-week, double-blind, placebo-controlled dose-response trial was conducted in 138 patients with type 2 diabetes using doses ranging from 0.25 (not an approved dose) to 4 mg taken with each of three meals. Repaglinide therapy resulted in dose-proportional glucose lowering over the full dose range. Plasma insulin levels increased after meals and reverted toward baseline before the next meal. Most of the fasting blood glucose-lowering effect was demonstrated within 1-2 weeks.
In a double-blind, placebo-controlled, 3-month dose titration study, repaglinide or placebo doses for each patient were increased weekly from 0.25 mg (not an approved dose) through 0.5, 1, and 2 mg, to a maximum of 4 mg, until a fasting plasma glucose (FPG) level <160 mg/dL was achieved or the maximum dose reached. The dose that achieved the targeted control or the maximum dose was continued to end of study. FPG and 2-hour post-prandial glucose (PPG) increased in patients receiving placebo and decreased in patients treated with repaglinide. Differences between the repaglinide- and placebo-treated groups were -61 mg/dL (FPG) and -104 mg/dL (PPG) (Table 4).
Table 4 Repaglinide vs. Placebo
- Mean Change from Baseline after 3 Months of Treatment
|
||
|
Repaglinide |
Placebo |
|
|
N |
66 |
33 |
|
Fasting Plasma Glucose (mg/dL) | ||
|
220.2 |
215.3 |
|
-31* |
30.3 |
|
Post Prandial Glucose (mg/dL) | ||
|
261.7 |
245.2 |
|
-47.6* |
56.5 |
The dosing of repaglinide relative to meal-related insulin release was studied in three trials including 58 patients. Glycemic control was maintained during a period in which the meal and dosing pattern was varied (2, 3 or 4 meals per day; before meals x 2, 3, or 4) compared with a period of 3 regular meals and 3 doses per day (before meals x 3). Blood glucose-lowering effect did not differ when repaglinide was administered at the start of a meal, 15 minutes before, or 30 minutes before the meal.
12.3 Pharmacokinetics
The pharmacokinetic parameters of repaglinide obtained from a single-dose, crossover study in healthy subjects and from a multiple-dose, parallel, dose-proportionality (0.5, 1, 2 and 4 mg) study in patients with type 2 diabetes are summarized in Tables 5 and 6. These data indicate that repaglinide did not accumulate in serum. Clearance of oral repaglinide did not change over the 0.5 mg to 4 mg dose range, indicating a linear relationship between dose and plasma drug levels.
Table 5: Pharmacokinetic Parameters for Repaglinide in Healthy Subjects
| |
|
|
|
|
|
|
- CL =total body clearance
- Vss= volume of distribution at steady state
- AbsBio = absolute bioavailability
Table 6: Pharmacokinetic Parameters for Repaglinide in Patients with Type 2 Diabetes*
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- *dosed preprandially with three meals
Absorption
After oral administration, repaglinide is completely absorbed from the gastrointestinal tract. After single and multiple oral doses in healthy subjects or in patients, peak plasma drug levels (Cmax) occur within 1 hour (T max). Repaglinide is eliminated from the blood stream with a half-life of approximately 1 hour. The mean absolute bioavailability is 56%. When repaglinide was given with food, the mean Tmax was not changed, but the mean Cmax and AUC (area under the time/plasma concentration curve) were decreased 20% and 12.4%, respectively.
Distribution
After intravenous (IV) dosing in healthy subjects, the volume of distribution at steady state (Vss) was 31 L, and the total body clearance (CL) was 38 L/h. Protein binding and binding to human serum albumin was greater than 98%.
Metabolism and Elimination
Repaglinide is completely metabolized by oxidative biotransformation and direct conjugation with glucuronic acid after either an IV or oral dose. The major metabolites are an oxidized dicarboxylic acid (M2), the aromatic amine (M1), and the acyl glucuronide (M7). The cytochrome P-450 enzyme system, specifically 2C8 and 3A4, have been shown to be involved in the N-dealkylation of repaglinide to M2 and the further oxidation to M1. Metabolites do not contribute to the glucose-lowering effect of repaglinide.
Within 96 hours after dosing with 14C-repaglinide as a single, oral dose, approximately 90% of the radiolabel was recovered in the feces and approximately 8% in the urine. Only 0.1% of the dose is cleared in the urine as parent compound. The major metabolite (M2) accounted for 60% of the administered dose. Less than 2% of parent drug was recovered in feces. Repaglinide appears to be a substrate for active hepatic uptake transporter (organic anion transporting protein OATP1B1).
Variability of Exposure
Repaglinide AUC after multiple doses of 0.25 to 4 mg with each meal varies over a wide range. The intra-individual and inter-individual coefficients of variation were 36% and 69%, respectively. AUC over the therapeutic dose range included 69 to 1005 ng/mL*hr, but AUC exposure up to 5417 ng/mL*hr was reached in dose escalation studies without apparent adverse consequences.
Specific Populations
Geriatric
Healthy volunteers were treated with a regimen of 2 mg repaglinide taken before each of 3 meals. There were no significant differences in repaglinide pharmacokinetics between the group of patients <65 years of age and a comparably sized group of patients ≥65 years of age [see Use in Specific Populations (8.5)].
Gender
A comparison of pharmacokinetics in males and females showed the AUC over the 0.5 mg to 4 mg dose range to be 15% to 70% higher in females with type 2 diabetes. This difference was not reflected in the frequency of hypoglycemic episodes (male: 16%; female: 17%) or other adverse events.
Race
No pharmacokinetic studies to assess the effects of race have been performed, but in a U.S. 1-year study in patients with type 2 diabetes, the blood glucose-lowering effect was comparable between Caucasians (n=297) and African-Americans (n=33). In a U.S. dose-response study, there was no apparent difference in exposure (AUC) between Caucasians (n=74) and Hispanics (n=33).
Renal Impairment
Single-dose and steady-state pharmacokinetics of repaglinide were compared between patients with type 2 diabetes and normal renal function (CrCl >80 mL/min), mild to moderate renal function impairment (CrCl=40 to 80 mL/min), and severe renal function impairment (CrCl=20 to 40 mL/min). Both AUC and Cmax of repaglinide were similar in patients with normal and mild to moderately impaired renal function (mean values 56.7 ng/mL*hr vs 57.2 ng/mL*hr and 37.5 ng/mL vs 37.7 ng/mL, respectively). Patients with severely reduced renal function had elevated mean AUC and Cmax values (98.0 ng/mL*hr and 50.7 ng/mL, respectively), but this study showed only a weak correlation between repaglinide levels and creatinine clearance.
Hepatic Impairment
A single-dose, open-label study was conducted in 12 healthy subjects and 12 patients with chronic liver disease (CLD) classified by Child-Pugh scale and caffeine clearance. Patients with moderate to severe impairment of liver function had higher and more prolonged serum concentrations of both total and unbound repaglinide than healthy subjects (AUChealthy: 91.6 ng/mL*hr; AUCCLDpatients: 368.9 ng/mL*hr; Cmax , healthy: 46.7 ng/mL; Cmax, CLD patients: 105.4 ng/mL). AUC was statistically correlated with caffeine clearance. No difference in glucose profiles was observed across patient groups.
Drug-Drug Interactions
Drug interaction studies performed in healthy volunteers show that repaglinide had no clinically relevant effect on the pharmacokinetic properties of digoxin, theophylline, or warfarin. Co-administration of cimetidine with repaglinide did not significantly alter the absorption and disposition of repaglinide.
Additionally, the following drugs were studied in healthy volunteers with co-administration of repaglinide.
Table 7 Effect of Other Drugs on AUC and Cmax of Repaglinide
|
Study Drug |
Dosing |
Repaglinide Dosing* |
Repaglinide |
|
|
AUC |
Cmax |
|||
|
Clarithromycin† |
250 mg BID for 4 days |
40%↑ |
67%↑ |
|
|
Clopidogrel† |
300 mg (Day 1) 75 mg QD (Day 2-3) |
0.25 mg (Day 1 and 3) |
(day 1) 5.1 fold↑ (3.9-6.6) (day 3) 3.9 fold↑ (2.9-5.3) |
2.5 fold↑ (1.8-3.5) 2.0 fold↑ (1.3-3.1) |
|
Cyclosporine |
100 mg (2 doses 12 hours apart) |
2.5 fold↑ |
1.8 fold↑ |
|
|
Deferasirox† |
30 mg/kg QD for 4 days |
0.5 mg |
2.3 fold↑ |
62% ↑ |
|
Fenofibrate |
200 mg QD for 5 days |
0% |
0% |
|
|
Gemfibrozil† |
600 mg BID for 3 days |
8.1 fold↑ |
2.4 fold↑ |
|
|
Itraconazole† |
100 mg BID for 3 days |
1.4 fold↑ |
1.5 fold↑ |
|
|
Gemfibrozil+Itraconazole† Co-administration |
Gem: 600 mg BID for 3 days Itra: 100 mg BID for 3 days |
19 fold↑ |
2.8 fold↑ |
|
|
Ketoconazole |
200 mg QD for 4 days |
2 mg |
15% ↑ |
16% ↑ |
|
Levonorgestrel/ethinyl Estradiol |
(0.15 mg/0.03 mg) Combination tablet QD for 21 days |
2 mg |
0%↑ |
20% ↑ |
|
Nifedipine† |
10 mg TID for 4 days |
2 mg |
0%↑ |
0% |
|
Rifampin† |
600 mg QD for 6-7 days |
4 mg |
32 – 80% ↓ |
17 – 79% ↓ |
|
Simvastatin |
20 mg QD for 4 days |
2 mg |
0%↓ |
26% ↓ |
|
Trimethoprim† |
160 mg BID for 2 days 160 mg QD for 1 day |
61% ↑ |
41% ↓ |
|
↑ Indicates increase
↓ Indicates decrease
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week carcinogenicity study in rats at doses up to 120 mg/kg/day, which is approximately 60 times clinical exposure on a mg/m2 basis, the incidences of benign adenomas of the thyroid and liver were increased in male rats. No evidence of carcinogenicity was found in female rats. The higher incidences of thyroid and liver tumors in male rats were not seen at lower dose of 30 mg/kg/day and 60 mg/kg/day respectively (which are over 15 and 30 times, respectively, clinical exposures on a mg/m2 basis). In a 104-week carcinogenicity study in mice at doses up to 500 mg/kg/day, no evidence of carcinogenicity was found in mice (which is approximately 125 times clinical exposure on a mg/m2 basis).
Repaglinide was non-genotoxic in a battery of in vivo and in vitro studies: Bacterial mutagenesis (Ames test), in vitro forward cell mutation assay in V79 cells (HGPRT), in vitro chromosomal aberration assay in human lymphocytes, unscheduled and replicating DNA synthesis in rat liver, and in vivo mouse and rat micronucleus tests.
In a rat fertility study, repaglinide was administered to male and female rats at doses up to 300 and 80 mg/kg/day, respectively. No adverse effects on fertility were observed (which are over 40 times clinical exposure on a mg/m2 basis).
14 CLINICAL STUDIES
14.1 Monotherapy Trials
A double-blind, placebo-controlled trial was carried out in 362 patients treated for 24 weeks. HbA1C for the repaglinide-treated groups (1 and 4 mg groups combined) at the end of the study was decreased compared to the placebo-treated group in treatment naïve patients and in patients previously treated with oral hypoglycemic agents by 2.1% and 1.7%, respectively. In this fixed-dose trial, patients who were treatment naïve to oral hypoglycemic agent therapy and patients with a HbA1C below 8% at baseline showed greater blood glucose-lowering.
14.2 Combination Trials
Repaglinide in Combination With Metformin
Repaglinide was studied in combination with metformin in 83 patients not satisfactorily controlled on exercise, diet, and metformin alone. Repaglinide dosage was titrated for 4 to 8 weeks, followed by a 3-month maintenance period. Combination therapy with repaglinide and metformin resulted in statistically significant improvement in HbA1C and fasting plasma glucose (FPG) compared to repaglinide or metformin monotherapy (Table 8). In this study where metformin dosage was kept constant, the combination therapy of Repaglinide and metformin showed dose-sparing effects with respect to repaglinide. The improvement in HbA1C and FPG of the combination group was achieved at a lower daily repaglinide dosage than in the repaglinide monotherapy group (Table 8).
Table 8 Repaglinide in Combination with Metformin:
- Mean Change from Baseline after 4 to 5 Months of Treatment**
|
Repaglinide Monotherapy |
Repaglinide Combination Therapy with Metformin |
Metformin Monotherapy |
|
|
N |
28 |
27 |
27 |
|
Median Final Dose (mg/day) |
12 |
6 (repaglinide) 1500 (metformin) |
1500 |
|
HbA1C (%) |
|||
|
8.6 |
8.3 |
8.6 |
|
-0.38* |
-1.41* |
-0.33 |
|
Fasting Plasma Glucose (mg/dL) |
|||
|
174 |
184 |
194 |
|
8.8 |
-39.2* |
-4.5 |
|
Weight (kg) |
|||
|
87 |
93 |
91 |
|
3.0 |
2.4† |
-0.90 |
** Based on intent-to-treat analysis.
Repaglinide in Combination With Pioglitazone
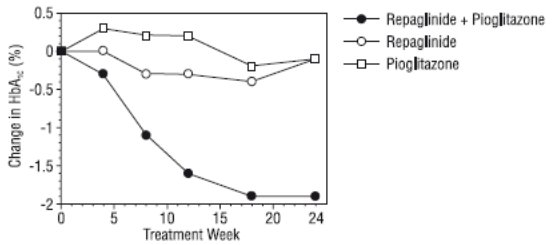
A combination therapy regimen of repaglinide and pioglitazone (N=123) was compared to repaglinide alone (N=61) and pioglitazone alone (N=62) in a 24-week trial that enrolled 246 patients previously treated with sulfonylurea or metformin monotherapy (HbA1C >7.0%). Repaglinide dosage was titrated during the first 12 weeks, followed by a 12-week maintenance period. Combination therapy resulted in statistically significant improvement in HbA1C and FPG compared to monotherapy (Figure 1). The changes from baseline for completers in FPG (mg/dL) and HbA1C (%), respectively were: -39.8 mg/dL and -0.1% for repaglinide, -35.3 mg/dL and -0.1% for pioglitazone and -92.4 mg/dL and -1.9% for the combination. In this study where pioglitazone dosage was kept constant, the combination therapy group showed dose-sparing effects with respect to repaglinide (see Figure 1 Legend). The improvement in HbA1C and FPG of the combination group was achieved at a lower daily repaglinide dosage than in the repaglinide monotherapy group.
Figure 1: Repaglinide in Combination with Pioglitazone: HbA1C Values
- LEGEND: HbA1C values by study week for patients who completed study (combination, N = 101; repaglinide, N = 35, pioglitazone, N = 26). Subjects with FPG above 270 mg/dL were withdrawn from the study.
- Pioglitazone dose: fixed at 30 mg/day; repaglinide median final dose: 6 mg/day for combination and 10 mg/day for monotherapy.
Repaglinide in Combination With Rosiglitazone
A combination therapy regimen of repaglinide and rosiglitazone was compared to monotherapy with either agent alone in a 24-week trial that enrolled 252 patients previously treated with sulfonylurea or metformin (HbA1C>7.0%). Combination therapy resulted in statistically significant improvement in HbA1C and FPG compared to monotherapy (Table 9 below). The glycemic effects of the combination therapy were dose-sparing with respect to both total daily repaglinide dosage and total daily rosiglitazone dosage (see Table 9 Legend). The improvement in HbA1C and FPG of the combination therapy group was achieved with lower daily dose of repaglinide and rosiglitazone, as compared to the respective monotherapy groups.
- Table 9 Repaglinide in Combination with Rosiglitazone: Mean Change from Baseline in a 24-Week Study**
|
Repaglinide Monotherapy |
Repaglinide Combination Therapy with Rosiglitazone |
Rosiglitazone Monotherapy |
|
|
N |
63 |
127 |
62 |
|
Median Final Dose (mg/day) |
12 |
6 (repaglinide) 4 (rosiglitazone) |
8 |
|
HbA1C (%) |
|||
|
9.3 |
9.1 |
9.0 |
|
-0.17 |
-1.43* |
-0.56 |
|
Fasting Plasma Glucose (mg/dL) |
|||
|
269 |
257 |
252 |
|
-54 |
-94* |
-67 |
|
Change in Weight (kg) |
+1.3 |
+4.5† |
+3.3 |
**Based on intent-to-treat analysis.
16 HOW SUPPLIED/STORAGE AND HANDLING
Repaglinide tablets, USP are available as follows:
0.5 mg, white to off-white colored, round tablet debossed with ‘SZ’ on one side and ‘361’on the other side.
NDC 0781-5148-01, bottle of 100 tablets
NDC 0781-5148-05, bottle of 500 tablets
NDC 0781-5148-10, bottle of 1000 tablets
NDC 0781-5148-13, in unit dose pack of 100 tablets (10 blister cards, each containing 10 tablets)
1 mg, yellow to pale yellow colored, slightly mottled, round tablet debossed with ‘SZ’ on one side and ‘362’ on the other side.
NDC 0781-5149-01, bottle of 100 tablets
NDC 0781-5149-05, bottle of 500 tablets
NDC 0781-5149-10, bottle of 1000 tablets
NDC 0781-5149-13, in unit dose pack of 100 tablets (10 blister cards, each containing 10 tablets)
2 mg, yellow to pale yellow colored, slightly mottled, capsule shaped tablet debossed with ‘SZ’ on one side and ‘363’ on the other side.
NDC 0781-5150-01, bottle of 100 tablets
NDC 0781-5150-05, bottle of 500 tablets
NDC 0781-5150-10, bottle of 1000 tablets
NDC 0781-5150-13, in unit dose pack of 100 tablets (10 blister cards, each containing 10 tablets)
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature]. Protect from moisture. Keep bottle tightly closed. Dispense in a tight, light-resistant container.
17 PATIENT COUNSELING INFORMATION
Hypoglycemia
Inform patients that repaglinide tablets can cause hypoglycemia and instruct patients and their caregivers on self-management procedures including glucose monitoring and management of hypoglycemia. Inform patients that their ability to concentrate and react may be impaired as a result of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended [see Warnings and Precautions (5.1)].
Administration
Instruct patients to take repaglinide tablets within 30 minutes before meals. Instruct patients to skip their dose of repaglinide when a meal is skipped [see Dosage and Administration (2)].
Drug Interactions
Discuss potential drug interactions with patients and inform them of potential drug-drug interactions with repaglinide tablets [see Drug Interactions (7)].
Manufactured in India by
Sandoz Private Ltd., for
Sandoz Inc., Princeton, NJ 08540
Rev. February 2017
| REPAGLINIDE
repaglinide tablet |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| REPAGLINIDE
repaglinide tablet |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| REPAGLINIDE
repaglinide tablet |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Sandoz Inc (110342024) |