LONG ACTING COUGH SOFTGELS- dextromethorphan hbr capsule, liquid filled
CHAIN DRUG MARKETING ASSOCIATION INC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Quality Choice 44-732-Delisted
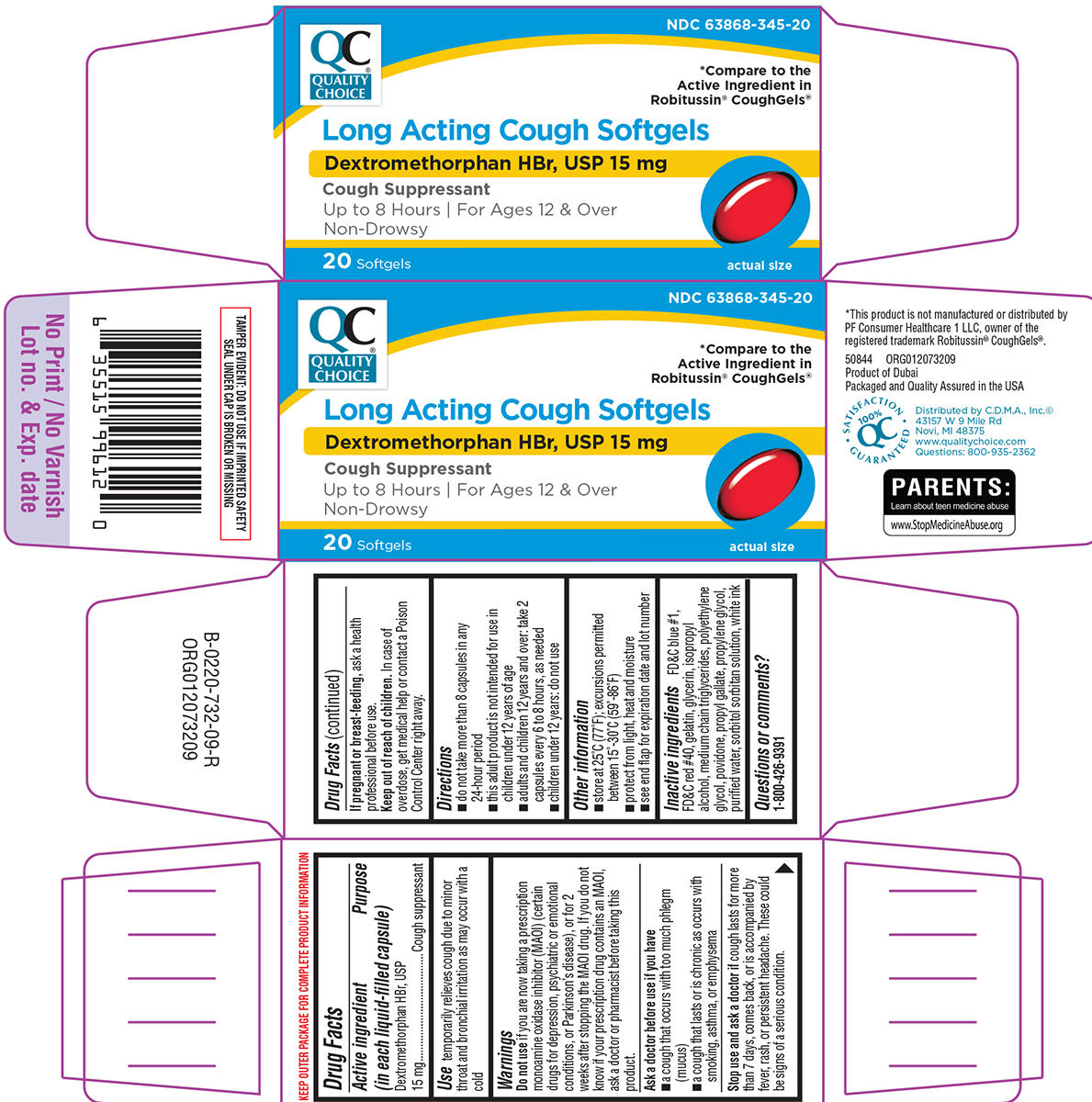
Use
temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- a cough that occurs with too much phlegm (mucus)
- a cough that lasts or is chronic as occurs with smoking, asthma, or emphysema
Directions
- do not take more than 8 capsules in any 24-hour period
- this adult product is not intended for use in children under 12 years of age
- adults and children 12 years and over: take 2 capsules every 6 to 8 hours, as needed
- children under 12 years: do not use
Other information
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- protect from light, heat and moisture
- see end flap for expiration date and lot number
Inactive ingredients
FD&C blue #1, FD&C red #40, gelatin, glycerin, isopropyl alcohol, medium chain triglycerides, polyethylene glycol, povidone, propyl gallate, propylene glycol, purified water, sorbitol sorbitan solution, white ink
Principal display panel
QC®
QUALITY
CHOICE
NDC 63868-345-20
*Compare to the active ingredient
in Robitussin® CoughGels®
Long Acting Cough Softgels
Dextromethorphan HBr, USP 15 mg
Cough Suppressant
Up to 8 Hours | For Ages 12 & Over
Non-Drowsy
actual size
20 Softgels
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL
UNDER CAP IS BROKEN OR MISSING
*This product is not manufactured or distributed by PF Consumer Healthcare 1 LLC, owner of the registered trademark Robitussin® CoughGels®.
50844 ORG012073209
Product of Dubai
Packaged and Quality Assured in the USA
SATISFACTION
100% QC
GUARANTEED
Distributed by C.D.M.A., Inc.©
43157 W 9 Mile Rd
Novi, MI 48375
www.qualitychoice.com
Questions: 800-935-2362
PARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
44-732
| LONG ACTING COUGH SOFTGELS
dextromethorphan hbr capsule, liquid filled |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - CHAIN DRUG MARKETING ASSOCIATION INC (011920774) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | pack(63868-345) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | pack(63868-345) | |