Label: ARTHRITIS PAIN RELIEVER- acetaminophen tablet, film coated, extended release
- NDC Code(s): 68084-777-25, 68084-777-95
- Packager: American Health Packaging
- This is a repackaged label.
- Source NDC Code(s): 51660-333
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 7, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 6 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product.
- Directions
- Other information
- Inactive ingredients
-
Questions?
- about the drug product, call Sun Pharmaceutical Industries, Inc. at 1-800-406-7984
- about the packaging, call American Health Packaging at 1-800-707-4621
Contains No Aspirin
PACKAGING INFORMATION
American Health Packaging unit dose blisters contain drug product from Ohm Laboratories Inc. as follows:
(650 mg / 30 UD) NDC 68084-777-25 packaged from NDC 51660-333Distributed by:
American Health Packaging
Columbus, OH 432178277725/0121
-
Principal Display Panel – Carton – 650 mg

NDC 68084- 777-25
Acetaminophen
Extended-release Tablets, USP
Pain Reliever/Fever Reducer650 mg
30 Tablets (5 x 6)
DO NOT USE WITH OTHER MEDICINES
CONTAINING ACETAMINOPHEN.Drug Facts
Active Ingredient Purpose
(in each caplet)
Acetaminophen USP, 650 mg ................. Pain reliever/fever reducerUses
See package insert for complete Drug Facts information.Warnings
Liver warning: This product contains acetaminophen.
Severe liver damage may occur if you take • more than 6 caplets
in 24 hours, which is the maximum daily amount • with other
drugs containing acetaminophen • 3 or more alcoholic drinks
every day while using this productAllergy alert: acetaminophen may cause severe skin reactions.
Symptoms may include: • skin reddening • blisters • rash
If a skin reaction occurs, stop use and seek medical help right
away.Keep out of reach of children.
See package insert for additional Drug Facts warnings.
Directions
• do not take more than directed, see package insert for
overdose warning and complete Drug Facts information.Other Information • store at 20° to 25°C (68° to 77°F).
Avoid excessive heat 40°C (104°F). • FOR YOUR
PROTECTION: Do not use product if blister is torn or broken.Contains No Aspirin
The drug product contained in this package is
from NDC # 51660-333, Ohm Laboratories Inc.Distributed by: American Health Packaging
2550 John Glenn Avenue, Suite A
Columbus, OH 43217077725
0277725/0222 - Principal Display Panel – Blister – 650 mg
-
INGREDIENTS AND APPEARANCE
ARTHRITIS PAIN RELIEVER
acetaminophen tablet, film coated, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68084-777(NDC:51660-333) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 650 mg Inactive Ingredients Ingredient Name Strength CROSPOVIDONE (UNII: 2S7830E561) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score no score Shape OVAL (Capsule Shaped) Size 19mm Flavor Imprint Code cor116 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68084-777-25 30 in 1 BOX, UNIT-DOSE 08/18/2014 1 NDC:68084-777-95 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 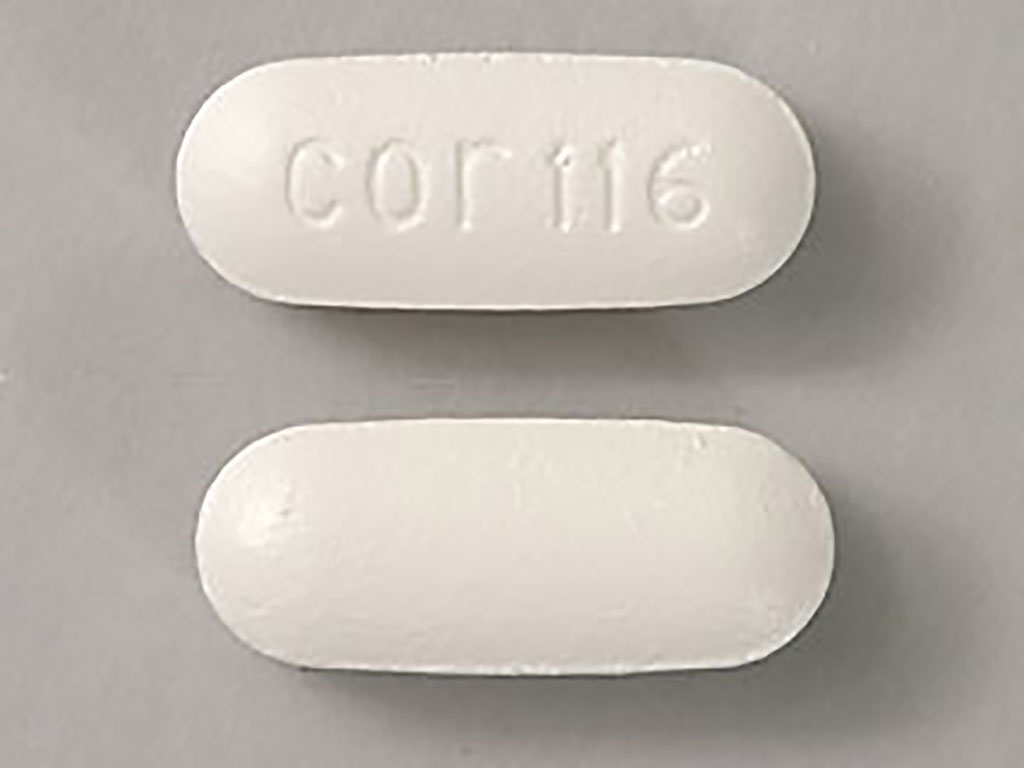
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076200 08/18/2014 Labeler - American Health Packaging (929561009) Establishment Name Address ID/FEI Business Operations American Health Packaging 929561009 repack(68084-777)



