XIIDRA- lifitegrast solution/ drops
Takeda Pharmaceuticals America, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use XIIDRA safely and effectively. See full prescribing information for XIIDRA.
XIIDRA® (lifitegrast ophthalmic solution), for topical ophthalmic use Initial U.S. Approval: 2016 INDICATIONS AND USAGEXiidra (lifitegrast ophthalmic solution) 5% is a lymphocyte function-associated antigen-1 (LFA-1) antagonist indicated for the treatment of the signs and symptoms of dry eye disease (DED). (1) DOSAGE AND ADMINISTRATIONOne drop twice daily in each eye (approximately 12 hours apart). (2) DOSAGE FORMS AND STRENGTHSOphthalmic solution containing lifitegrast 50 mg/mL (5%). (3) CONTRAINDICATIONSHypersensitivity. (4) ADVERSE REACTIONSThe most common adverse reactions (incidence 5%-25%) following the use of Xiidra were instillation site irritation, dysgeusia, and decreased visual acuity. (6)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 11/2019 |
FULL PRESCRIBING INFORMATION: CONTENTS*1 INDICATIONS AND USAGE2 DOSAGE AND ADMINISTRATION3 DOSAGE FORMS AND STRENGTHS4 CONTRAINDICATIONS6 ADVERSE REACTIONS6.1 Clinical Trials Experience6.2 Postmarketing Experience8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy8.2 Lactation8.4 Pediatric Use8.5 Geriatric Use11 DESCRIPTION12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action12.3 Pharmacokinetics13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility14 CLINICAL STUDIES16 HOW SUPPLIED/STORAGE AND HANDLING17 PATIENT COUNSELING INFORMATION
|
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Xiidra® (lifitegrast ophthalmic solution) 5% is indicated for the treatment of the signs and symptoms of dry eye disease (DED).
2 DOSAGE AND ADMINISTRATION
Instill one drop of Xiidra twice daily (approximately 12 hours apart) into each eye using a single-use container. Discard the single-use container immediately after using in each eye.
Contact lenses should be removed prior to the administration of Xiidra and may be reinserted 15 minutes following administration.
4 CONTRAINDICATIONS
Xiidra is contraindicated in patients with known hypersensitivity to lifitegrast or to any of the other ingredients in the formulation [see Adverse Reactions (6.2)].
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity [see Contraindications (4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In five clinical studies of DED conducted with lifitegrast ophthalmic solution, 1401 patients received at least one dose of lifitegrast (1287 of which received lifitegrast 5%). The majority of patients (84%) had ≤ 3 months of treatment exposure. One hundred-seventy patients were exposed to lifitegrast for approximately 12 months. The majority of the treated patients were female (77%). The most common adverse reactions reported in 5%-25% of patients were instillation-site irritation, dysgeusia, and reduced visual acuity.
Other adverse reactions reported in 1%-5% of the patients were blurred vision, conjunctival hyperemia, eye irritation, headache, increased lacrimation, eye discharge, eye discomfort, eye pruritus, and sinusitis.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Xiidra. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Rare cases of hypersensitivity, including anaphylactic reaction, bronchospasm, respiratory distress, pharyngeal edema, swollen tongue, and urticaria have been reported. Eye swelling and rash have been reported [see Contraindications (4)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on Xiidra use in pregnant women to inform any drug-associated risks. Intravenous (IV) administration of lifitegrast to pregnant rats, from pre-mating through gestation Day 17, did not produce teratogenicity at clinically relevant systemic exposures. Intravenous administration of lifitegrast to pregnant rabbits during organogenesis produced an increased incidence of omphalocele at the lowest dose tested, 3 mg/kg/day (400-fold the human plasma exposure at the recommended human ophthalmic dose [RHOD], based on the area under the curve [AUC] level). Since human systemic exposure to lifitegrast following ocular administration of Xiidra at the RHOD is low, the applicability of animal findings to the risk of Xiidra use in humans during pregnancy is unclear [see Clinical Pharmacology (12.3)].
Data
Animal Data
Lifitegrast administered daily by IV injection to rats, from pre-mating through gestation Day 17, caused an increase in mean pre-implantation loss and an increased incidence of several minor skeletal anomalies at 30 mg/kg/day, representing five, 400-fold the human plasma exposure at the RHOD of Xiidra, based on AUC. No teratogenicity was observed in the rat at 10 mg/kg/day (460-fold the human plasma exposure at the RHOD, based on AUC). In the rabbit, an increased incidence of omphalocele was observed at the lowest dose tested, 3 mg/kg/day (400-fold the human plasma exposure at the RHOD, based on AUC), when administered by IV injection daily from gestation Days 7 through 19. A fetal no observed adverse effect level (NOAEL) was not identified in the rabbit.
8.2 Lactation
Risk Summary
There are no data on the presence of lifitegrast in human milk, the effects on the breastfed infant, or the effects on milk production. However, systemic exposure to lifitegrast from ocular administration is low [see Clinical Pharmacology (12.3)]. The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for Xiidra and any potential adverse effects on the breastfed child from Xiidra.
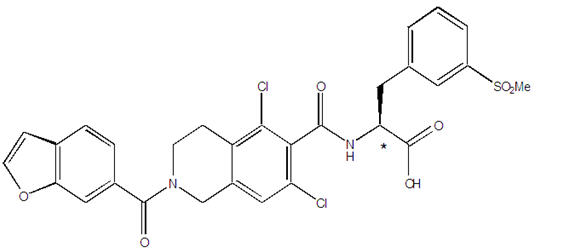
11 DESCRIPTION
The chemical name for lifitegrast is (S)-2-(2-(benzofuran-6-carbonyl)-5,7-dichloro-1,2,3,4-tetrahydroisoquinoline-6-carboxamido)-3-(3-(methylsulfonyl)phenyl)propanoic acid. The molecular formula of lifitegrast is C29H24Cl2N2O7S and its molecular weight is 615.5 g/mol. The structural formula of lifitegrast is:
|
|
| *Chiral center |
Lifitegrast is a white to off-white powder which is soluble in water.
Xiidra (lifitegrast ophthalmic solution) 5% is a lymphocyte function-associated antigen-1 (LFA-1) antagonist supplied as a sterile, clear, colorless to slightly brownish-yellow colored, isotonic solution of lifitegrast with a pH of 7.0-8.0, and an osmolality range of 200-330 mOsmol/kg.
Xiidra contains Active: lifitegrast 50 mg/mL; Inactives: sodium chloride, sodium phosphate dibasic anhydrous, sodium thiosulfate pentahydrate, and water for injection. Sodium hydroxide and/or hydrochloric acid (to adjust pH).
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Lifitegrast binds to the integrin LFA-1, a cell surface protein found on leukocytes and blocks the interaction of LFA-1 with its cognate ligand intercellular adhesion molecule-1 (ICAM-1). ICAM-1 may be overexpressed in corneal and conjunctival tissues in DED. LFA-1/ICAM-1 interaction can contribute to the formation of an immunological synapse resulting in T-cell activation and migration to target tissues. In vitro studies demonstrated that lifitegrast may inhibit T-cell adhesion to ICAM-1 in a human T-cell line and may inhibit secretion of inflammatory cytokines in human peripheral blood mononuclear cells. The exact mechanism of action of lifitegrast in DED is not known.
12.3 Pharmacokinetics
In a subset of DED patients (n = 47) enrolled in a Phase 3 trial, the pre-dose (trough) plasma concentrations of lifitegrast were measured after 180 and 360 days of topical ocular dosing (one drop twice daily) with Xiidra (lifitegrast ophthalmic solution) 5%. A total of nine of the 47 patients (19%) had plasma lifitegrast trough concentrations above 0.5 ng/mL (the lower limit of assay quantitation). Trough plasma concentrations that could be quantitated ranged from 0.55 ng/mL to 3.74 ng/mL.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal studies have not been conducted to determine the carcinogenic potential of lifitegrast.
Mutagenesis
Lifitegrast was not mutagenic in the in vitro Ames assay. Lifitegrast was not clastogenic in the in vivo mouse micronucleus assay. In an in vitro chromosomal aberration assay using mammalian cells (Chinese hamster ovary cells), lifitegrast was positive at the highest concentration tested, without metabolic activation.
Impairment of Fertility
Lifitegrast administered at IV doses of up to 30 mg/kg/day (5400-fold the human plasma exposure at the RHOD of lifitegrast ophthalmic solution, 5%) had no effect on fertility and reproductive performance in male and female treated rats.
14 CLINICAL STUDIES
The safety and efficacy of lifitegrast for the treatment of DED were assessed in a total of 1181 patients (1067 of which received lifitegrast 5%) in four 12-week, randomized, multi-center, double-masked, vehicle-controlled studies. Patients were randomized to Xiidra or vehicle (placebo) in a 1:1 ratio and dosed twice a day. Use of artificial tears was not allowed during the studies. The mean age was 59 years (range, 19-97 years). The majority of patients were female (76%). Enrollment criteria included, minimal signs (i.e., Corneal Fluorescein Staining and non-anesthetized Schirmer Tear Test) and symptoms (i.e., Eye Dryness Score (EDS) and Ocular Discomfort Score) severity scores at baseline.
Effects on Symptoms of Dry Eye Disease
Eye dryness score was rated by patients using a visual analogue scale (0 = no discomfort, 100 = maximal discomfort) at each study visit. The average baseline EDS was between 40 and 70. A larger reduction in EDS favoring Xiidra was observed in all studies at Day 42 and Day 84 (see Figure 1).
Figure 1: Mean Change (SD) From Baseline and Treatment Difference (Xiidra – Vehicle) in Eye Dryness Score in 12-Week Studies in Patients With Dry Eye Disease
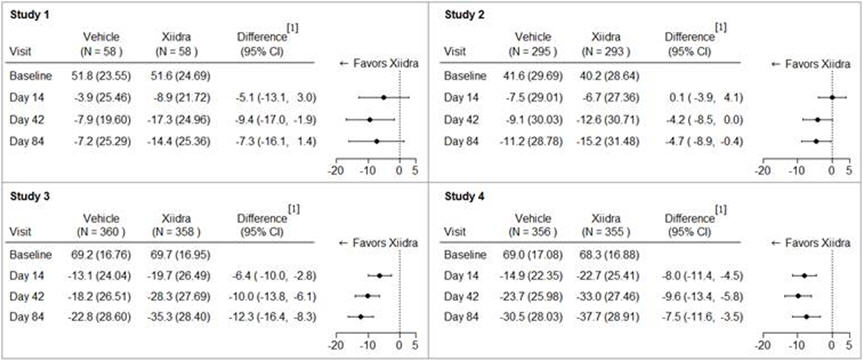
[1] Based on analysis of covariance (ANCOVA) model adjusted for baseline value in Study 1, and ANCOVA model adjusted for baseline value and randomization stratification factors in Studies 2-4. All randomized and treated patients were included in the analysis and missing data were imputed using last-available data. In Study 1, one Xiidra treated subject who did not have a baseline value was excluded from analysis.
Effects on Signs of Dry Eye Disease
Inferior fluorescein corneal staining score (ICSS) (0 = no staining, 1 = few/rare punctate lesions, 2 = discrete and countable lesions, 3 = lesions too numerous to count but not coalescent, 4 = coalescent) was recorded at each study visit. The average baseline ICSS was approximately 1.8 in Studies 1 and 2, and 2.4 in Studies 3 and 4. At Day 84, a larger reduction in ICSS favoring Xiidra was observed in three of the four studies (see Figure 2).
Figure 2: Mean Change (SD) From Baseline and Treatment Difference (Xiidra – Vehicle) in Inferior Corneal Staining Score in 12-Week Studies in Patients With Dry Eye Disease
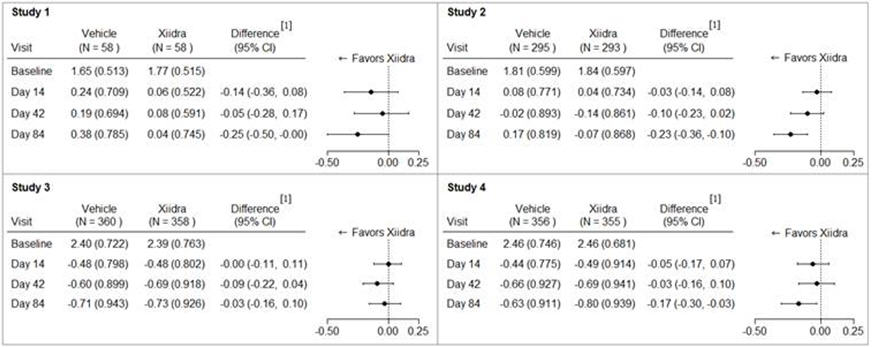
[1] Based on ANCOVA model adjusted for baseline value in Study 1, and ANCOVA model adjusted for baseline value and randomization stratification factors in Studies 2-4. All randomized and treated patients were included in the analysis and missing data were imputed using last-available data. In Study 2, one Vehicle treated subject who did not have a study eye designated was excluded from analysis.
16 HOW SUPPLIED/STORAGE AND HANDLING
Xiidra (lifitegrast ophthalmic solution) 5% (50 mg/mL) is supplied in a foil pouch containing 5 low density polyethylene 0.2 mL single-use containers.
Carton of 60 single-use containers NDC 54092-606-01
Storage:
Store at 20°C to 25°C (68°F to 77°F). Store single-use containers in the original foil pouch.
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Handling the Single-use Container
Advise patients not to touch the tip of the single-use container to their eye or to any surface, in order to avoid eye injury or contamination of the solution.
Use With Contact Lenses
Advise patients that contact lenses, should be removed prior to administration of Xiidra and can be reinserted 15 minutes after administration [see Dosage and Administration (2)].
Administration
Advise patients that the solution from one single-use container is to be used immediately after opening. It can be used to dose both eyes. The single-use container, including any remaining contents should be discarded immediately after administration [see Dosage and Administration (2)].
Storage Information
Instruct patients to store single-use containers in the original foil pouch until ready to use [see How Supplied/Storage and Handling (16)].
Manufactured for:
Novartis Pharmaceuticals Corporation
One Health Plaza
East Hanover, NJ 07936
T2019-110
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: November 2019 |
| PATIENT INFORMATION
Xiidra® (ZYE-druh) (lifitegrast ophthalmic solution) 5% for topical ophthalmic use |
|
| What is Xiidra?
Xiidra is a prescription eye drop solution used to treat the signs and symptoms of dry eye disease (DED). It is not known if Xiidra is safe and effective in children under 17 years of age. |
|
Do not use Xiidra:
|
|
Before you use Xiidra, tell your doctor if you:
|
|
| How should I use Xiidra?
See the complete Instructions for use at the end of this Patient Information leaflet for detailed instructions about the right way to use Xiidra.
|
|
| What are the possible side effects of Xiidra?
The most common side effects of Xiidra include eye irritation, discomfort, or blurred vision when the drops are applied to the eyes, and an unusual taste sensation (dysgeusia). Seek medical care immediately if you get any symptoms of wheezing, difficulty breathing, or swollen tongue. These are not all the possible side effects of Xiidra. Tell your doctor if you have any side effects that bother you. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store Xiidra?
|
|
| General information about the safe and effective use of Xiidra.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or doctor for information about Xiidra that is written for health professionals. Do not use Xiidra for a condition for which it was not prescribed. Do not give Xiidra to other people, even if they have the same symptoms you have. It may harm them. |
|
| What are the ingredients in Xiidra?
Active ingredient: lifitegrast Inactive ingredients: sodium chloride, sodium phosphate dibasic anhydrous, sodium thiosulfate pentahydrate, and water for injection. Sodium hydroxide and/or hydrochloric acid (to adjust pH). |
|
| Manufactured for: Novartis Pharmaceuticals Corporation, One Health Plaza, East Hanover, NJ 07936-1080 For more information, go to www.Xiidra.com or call 1-888-NOW-NOVA. |
|
T2019-111
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: November 2019 |
| INSTRUCTIONS FOR USE
Xiidra® [ZYE-druh] (lifitegrast ophthalmic solution) 5% for topical ophthalmic use |
|
| Read this Instructions for Use before you start using Xiidra and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your doctor about your medical condition or your treatment. | |
Important Information You Need to Know Before Using Xiidra:
|
|
|
Follow Steps 1 to 9 each time you use Xiidra: |
|
|
Step 1. Take a foil pouch out of the Xiidra box. Open the pouch and remove the strip of single-use containers. | Figure 1:
 |
|
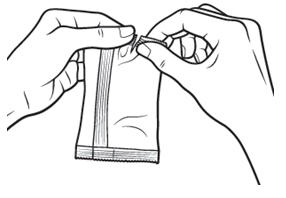
Pull off one single-use container from the strip. | Figure 2:
 |
|
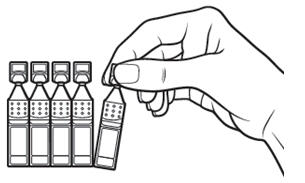
Step 2. Put the remaining strip of single-use containers back in the pouch. | Figure 3:
 |
|
Fold the edge to close the pouch. | Figure 4:
 |
|
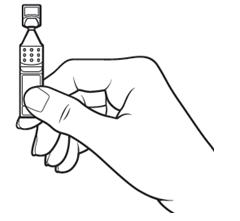
Step 3. Hold the Xiidra container upright. | Figure 5:
 |
|
Tap the top of the container until all of the solution is in the bottom part of the container. | Figure 6:
 |
|
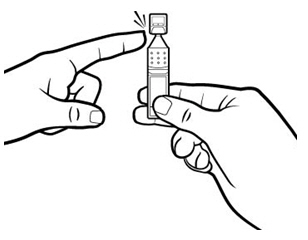
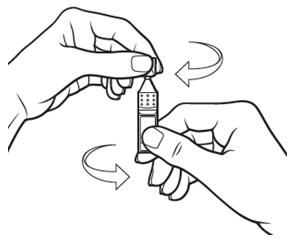
Step 4. Open the Xiidra single-use container by twisting off the tab. Make sure that the tip of the single-use container does not touch anything, to avoid contamination. | Figure 7:
 |
|
Step 5. Tilt your head backwards. If you are not able to tilt your head, lie down. |
|
|
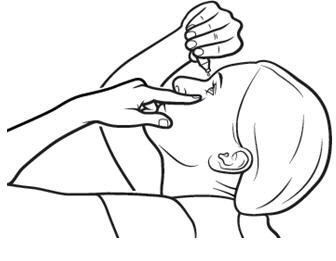
Step 6. Gently pull your lower eyelid downwards and look up. Step 7. Place the tip of the Xiidra single-use container close to your eye, but be careful not to touch your eye with it. Step 8. Gently squeeze the single-use container and let one drop of Xiidra fall into the space between your lower eyelid and your eye. If a drop misses your eye, try again. | Figure 8:
 |
|
Step 9. Repeat Steps 5-8 for your other eye. There is enough Xiidra in one single-use container for both eyes.
|
|
|
Manufactured for: Novartis Pharmaceuticals Corporation, One Health Plaza, East Hanover, NJ 07396-1080 |
|
T2019-112
| XIIDRA
lifitegrast solution/ drops |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Takeda Pharmaceuticals America, Inc. (830134016) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| The Ritedose Corporation | 837769546 | MANUFACTURE(54092-606) , ANALYSIS(54092-606) , PACK(54092-606) , LABEL(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Cambrex Charles City, Inc | 782974257 | API MANUFACTURE(54092-606) , ANALYSIS(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| AMRI Rensselaer, Inc. | 124193793 | API MANUFACTURE(54092-606) , ANALYSIS(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SGS Life Science Services | 062491980 | ANALYSIS(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Albany Molecular Research, Inc. | 080046430 | ANALYSIS(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ACM PHARMA | 384719527 | ANALYSIS(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| PPD Development, L.P. | 838082055 | ANALYSIS(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sharp Corporation | 143696495 | PACK(54092-606) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Laboratoire Unither | 574139809 | MANUFACTURE(54092-606) , ANALYSIS(54092-606) , PACK(54092-606) , LABEL(54092-606) | |