PAIN RELIEF 8 HR- acetaminophen tablet, film coated, extended release
Kroger Company
----------
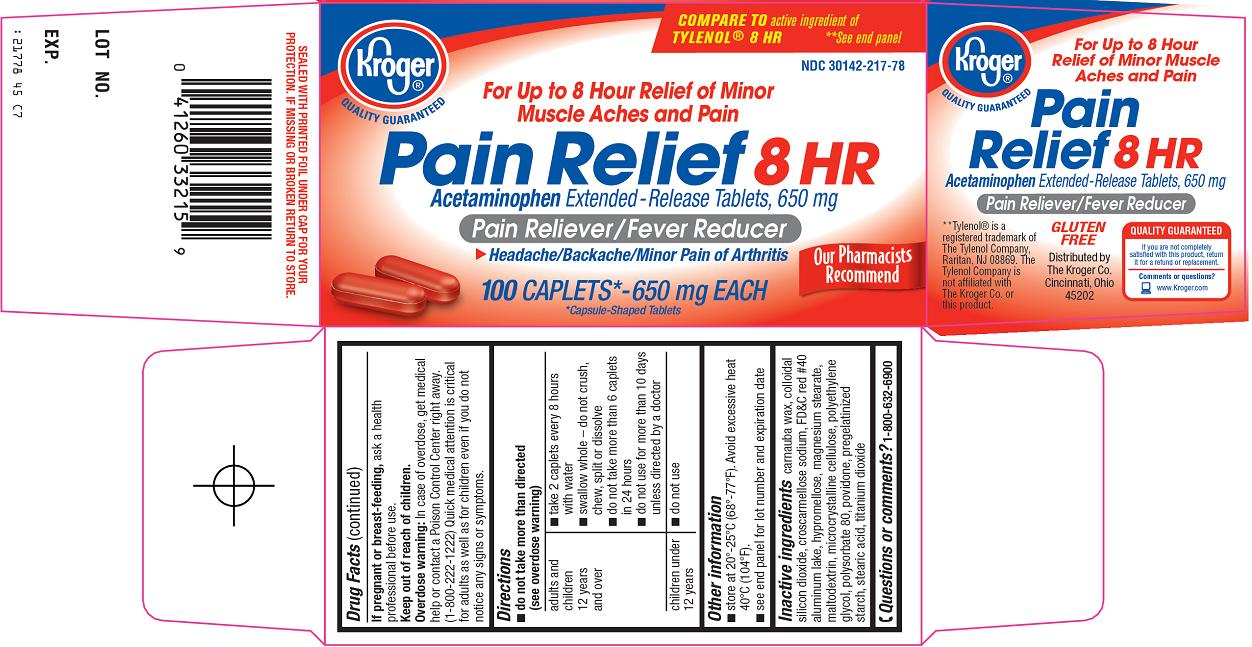
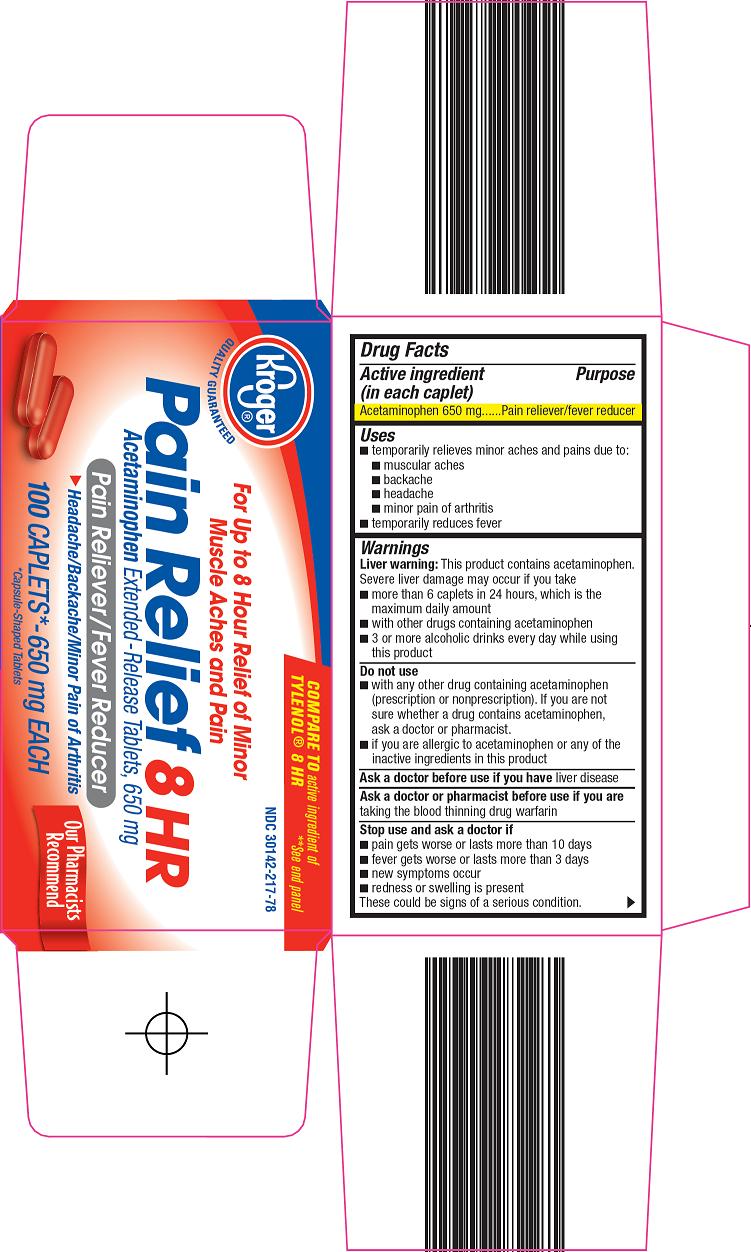
Kroger Co. Pain Relief 8 HR Drug Facts
Uses
- •
- temporarily relieves minor aches and pains due to:
- •
- muscular aches
- •
- backache
- •
- headache
- •
- minor pain of arthritis
- •
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- •
- more than 6 caplets in 24 hours, which is the maximum daily amount
- •
- with other drugs containing acetaminophen
- •
- 3 or more alcoholic drinks every day while using this product
Do not use
- •
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
Directions
- •
- do not take more than directed (see overdose warning)
|
adults and children 12 years and over |
|
|
children under 12 years |
|
Other information
- •
- store at 20°-25°C (68°-77°F). Avoid excessive heat 40°C (104°F).
- •
- see end panel for lot number and expiration date
Inactive ingredients
carnauba wax, colloidal silicon dioxide, croscarmellose sodium, FD&C red #40 aluminum lake, hypromellose, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, polysorbate 80, povidone, pregelatinized starch, stearic acid, titanium dioxide
Principal Display Panel
COMPARE TO active ingredient of TYLENOL® 8 HR
See end panel
For up to 8 Hour Relief of Minor Muscle Aches and Pain
Pain Relief 8 HR
Acetaminophen Extended-Release Tablets, 650 mg
Pain Reliever/Fever Reducer
Headache/Backache/Minor Pain of Arthritis
650 mg EACH
*Capsule-Shaped Tablets
| PAIN RELIEF 8 HR
acetaminophen tablet, film coated, extended release |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Kroger Company (006999528) |