SPF 30 MOISTURIZING FACIAL SUNSCREEN CVS HEALTH- homosalate, meradimate, octinoxate, octocrylene, zinc oxide lotion
CVS PHARMACY
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
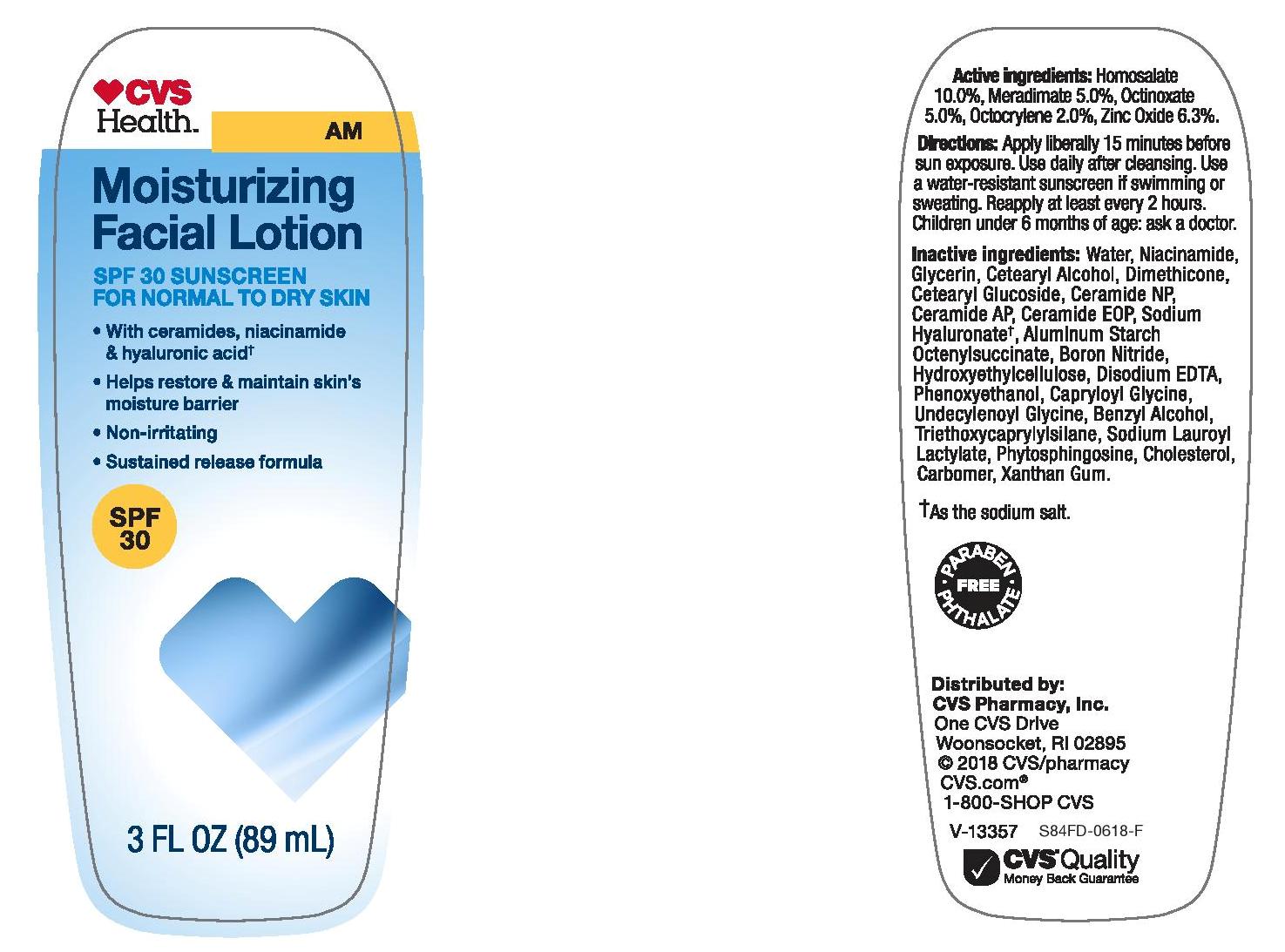
CVS HEALTH SPF 30 MOISTURIZING FACIAL LOTION
Active ingredients
Homosalate 10.0%, Meradimate 5.0%, Octinoxate 5.0%, Octocrylene 2.0%, Zinc Oxide 6.3%
Warnings
Skin Cancer/ Skin Aging Alert: Spending time in the sun increases your risk of skin cancer and early skin aging. This product has been shown only to help prevent sunburn, not skin cancer or early skin aging.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months of age: ask a doctor
Other information
- protect this product from excessive heat and direct sun
- may stain or damage some fabrics/ materials or surfaces
Inactive ingredients
water, niacinamide, glycerin, cetearyl alcohol, dimethicone, cetearyl glucoside, ceramide NP, ceramide AP, ceramide EOP, sodium hyaluronate, aluminum starch octenylsuccinate, boron nitride, hydroxyethylcellulose, disodium EDTA, phenoxyethanol, capryloyl glycine, undecylenoyl glycine, benzyl alcohol, triethoxycaprylylsilane, sodium lauroyl lactylate, phytosphingosine, cholesterol, carbomer, xanthan gum.
| SPF 30 MOISTURIZING FACIAL SUNSCREEN
CVS HEALTH
homosalate, meradimate, octinoxate, octocrylene, zinc oxide lotion |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - CVS PHARMACY (062312574) |
| Registrant - FRUIT OF THE EARTH, INC. (079559467) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| FRUIT OF THE EARTH, INC. | 080086802 | manufacture(69842-287) | |