KAPVAY - clonidine hydrochloride tablet, extended release
Concordia Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use KAPVAY safely and effectively. See full prescribing information for KAPVAY.
KAPVAY® (clonidine hydrochloride) extended-release tablets, for oral use Initial U.S. Approval: 1974 INDICATIONS AND USAGEKAPVAY® is a centrally acting alpha2-adrenergic agonist indicated for the treatment of attention deficit hyperactivity disorder (ADHD) as monotherapy or as adjunctive therapy to stimulant medications. (1) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSExtended-release tablets: 0.1 mg, not scored. (3) CONTRAINDICATIONSHistory of a hypersensitivity reaction to clonidine. Reactions have included generalized rash, urticaria, angioedema. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (incidence at least 5% and twice the rate of placebo) as monotherapy in ADHD: somnolence, fatigue, irritability, nightmare, insomnia, constipation, dry mouth. (6.1) DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 2/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
KAPVAY® (clonidine hydrochloride) extended-release is indicated for the treatment of attention deficit hyperactivity disorder (ADHD) as monotherapy and as adjunctive therapy to stimulant medications [see Clinical Studies (14)].
2 DOSAGE AND ADMINISTRATION
2.1 General Dosing Information
KAPVAY is an extended-release tablet to be taken orally with or without food. Swallow tablets whole. Do not crush, chew, or break tablets because this will increase the rate of clonidine release.
Due to the lack of controlled clinical trial data and differing pharmacokinetic profiles, substitution of KAPVAY for other clonidine products on a mg-per-mg basis is not recommended [see Clinical Pharmacology (12.3)].
2.2 Dose Selection
The dose of KAPVAY, administered either as monotherapy or as adjunctive therapy to a psychostimulant, should be individualized according to the therapeutic needs and response of the patient. Dosing should be initiated with one 0.1 mg tablet at bedtime, and the daily dosage should be adjusted in increments of 0.1 mg/day at weekly intervals until the desired response is achieved. Doses should be taken twice a day, with either an equal or higher split dosage being given at bedtime (see Table 1).
| Total Daily Dose
| Morning Dose
| Bedtime Dose
|
| 0.1 mg/day | 0.1 mg |
|
| 0.2 mg/day | 0.1 mg | 0.1 mg |
| 0.3 mg/day | 0.1 mg | 0.2 mg |
| 0.4 mg/day | 0.2 mg | 0.2 mg |
Doses of KAPVAY higher than 0.4 mg/day (0.2 mg twice daily) were not evaluated in clinical trials for ADHD and are not recommended.
When KAPVAY is being added-on to a psychostimulant, the dose of the psychostimulant can be adjusted depending on the patient's response to KAPVAY.
2.3 Discontinuation
When discontinuing KAPVAY, the total daily dose should be tapered in decrements of no more than 0.1 mg every 3 to 7 days to avoid rebound hypertension [see Warnings and Precautions (5.3)].
3 DOSAGE FORMS AND STRENGTHS
KAPVAY is available as 0.1 mg strength extended-release tablets. The 0.1 mg tablets are round, white, non-scored, standard convex with debossing on one side. KAPVAY tablets must be swallowed whole and never crushed, cut or chewed.
4 CONTRAINDICATIONS
KAPVAY is contraindicated in patients with a history of a hypersensitivity reaction to clonidine. Reactions have included generalized rash, urticaria, and angioedema [see Adverse Reactions (6)].
5 WARNINGS AND PRECAUTIONS
5.1 Hypotension/Bradycardia
Treatment with KAPVAY can cause dose-related decreases in blood pressure and heart rate [see Adverse Reactions (6.1)]. Measure heart rate and blood pressure prior to initiation of therapy, following dose increases, and periodically while on therapy. Titrate KAPVAY slowly in patients with a history of hypotension, and those with underlying conditions that may be worsened by hypotension and bradycardia; e.g., heart block, bradycardia, cardiovascular disease, vascular disease, cerebrovascular disease, or chronic renal failure. In patients who have a history of syncope or may have a condition that predisposes them to syncope, such as hypotension, orthostatic hypotension, bradycardia, or dehydration, advise patients to avoid becoming dehydrated or overheated. Monitor blood pressure and heart rate, and adjust dosages accordingly in patients treated concomitantly with antihypertensives or other drugs that can reduce blood pressure or heart rate or increase the risk of syncope.
5.2 Sedation and Somnolence
Somnolence and sedation were commonly reported adverse reactions in clinical studies. In patients that completed 5 weeks of therapy in a controlled, fixed dose pediatric monotherapy study, 31% of patients treated with 0.4 mg/day and 38% treated with 0.2 mg/day versus 4% of placebo treated patients reported somnolence as an adverse event. In patients that completed 5 weeks of therapy in a controlled flexible dose pediatric adjunctive to stimulants study, 19% of patients treated with KAPVAY+stimulant versus 7% treated with placebo+stimulant reported somnolence. Before using KAPVAY with other centrally active depressants (such as phenothiazines, barbiturates, or benzodiazepines), consider the potential for additive sedative effects. Caution patients against operating heavy equipment or driving until they know how they respond to treatment with KAPVAY. Advise patients to avoid use with alcohol.
5.3 Rebound Hypertension
Abrupt discontinuation of KAPVAY can cause rebound hypertension. In adults with hypertension, sudden cessation of clonidine hydrochloride extended-release formulation treatment in the 0.2 to 0.6 mg/day range resulted in reports of headache, tachycardia, nausea, flushing, warm feeling, brief lightheadedness, tightness in chest, and anxiety. In adults with hypertension, sudden cessation of treatment with immediate-release clonidine has, in some cases, resulted in symptoms such as nervousness, agitation, headache, and tremor accompanied or followed by a rapid rise in blood pressure and elevated catecholamine concentrations in the plasma.
No studies evaluating abrupt discontinuation of KAPVAY in children with ADHD have been conducted; however, to minimize the risk of rebound hypertension, gradually reduce the dose of KAPVAY in decrements of no more than 0.1 mg every 3 to 7 days. Patients should be instructed not to discontinue KAPVAY therapy without consulting their physician due to the potential risk of withdrawal effects.
5.4 Allergic Reactions
In patients who have developed localized contact sensitization to clonidine transdermal system, continuation of clonidine transdermal system or substitution of oral KAPVAY therapy may be associated with the development of a generalized skin rash.
In patients who develop an allergic reaction from clonidine transdermal system, substitution of oral KAPVAY may also elicit an allergic reaction (including generalized rash, urticaria, or angioedema).
5.5 Cardiac Conduction Abnormalities
The sympatholytic action of clonidine may worsen sinus node dysfunction and atrioventricular (AV) block, especially in patients taking other sympatholytic drugs. There have been post-marketing reports of patients with conduction abnormalities and/or taking other sympatholytic drugs who developed severe bradycardia requiring IV atropine, IV isoproterenol, and temporary cardiac pacing while taking clonidine. Titrate KAPVAY slowly and monitor vital signs frequently in patients with cardiac conduction abnormalities or patients concomitantly treated with other sympatholytic drugs.
6 ADVERSE REACTIONS
The following serious adverse reactions are described in greater detail elsewhere in labeling:
- Hypotension/bradycardia [see Warnings and Precautions (5.1)]
- Sedation and somnolence [see Warnings and Precautions (5.2)]
- Rebound hypertension [see Warnings and Precautions (5.3)]
- Allergic reactions [see Warnings and Precautions (5.4)]
- Cardiac Conduction Abnormalities [see Warnings and Precautions (5.5)]
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Two KAPVAY ADHD clinical studies (Study 1, CLON-301 and Study 2, CLON-302) evaluated 256 patients in two 8-week placebo-controlled studies.
A third KAPVAY ADHD clinical study (Study 3, SHN-KAP-401) evaluated 135 children and adolescents in a 40-week placebo-controlled randomized-withdrawal study.
Study 1: Fixed-dose KAPVAY Monotherapy
Study 1 (CLON-301) was a short-term, multi-center, randomized, double-blind, placebo-controlled study of two fixed doses (0.2 mg/day or 0.4 mg/day) of KAPVAY in children and adolescents (6 to 17 years of age) who met DSM-IV criteria for ADHD hyperactive or combined inattentive/hyperactive subtypes.
Most Common Adverse Reactions (incidence of ≥5% and at least twice the rate of placebo): somnolence, fatigue, irritability, insomnia, nightmare, constipation, dry mouth.
Adverse Events Leading to Discontinuation of KAPVAY - Five patients (7%) in the low dose group (0.2 mg), 15 patients (20%) in the high dose group (0.4 mg), and 1 patient in the placebo group (1%) reported adverse reactions that led to discontinuation. The most common adverse reactions that led to discontinuation were somnolence and fatigue.
Commonly observed adverse reactions (incidence of ≥2% in either active treatment group and greater than the rate on placebo) during the treatment period are listed in Table 2.
|
Percentage of Patients Reporting Event |
|||
| Preferred Term
| KAPVAY 0.2 mg/day N=76 | KAPVAY 0.4 mg/day N=78 | Placebo (N=76) |
| PSYCHIATRIC DISORDERS Somnolence * Nightmare Emotional Disorder Aggression Tearfulness Enuresis Sleep Terror Poor Quality Sleep | 38% 4% 4% 3% 1% 0% 3% 0% | 31% 9% 4% 1% 3% 4% 0% 3% | 4% 0% 1% 0% 0% 0% 0% 1% |
| NERVOUS SYSTEM DISORDERS Headache Insomnia Tremor Abnormal Sleep-Related Event | 20% 5% 1% 3% | 13% 6% 4% 1% | 16% 1% 0% 0% |
| GASTROINTESTINAL DISORDERS Upper Abdominal Pain Nausea Constipation Dry Mouth | 15% 4% 1% 0% | 10% 5% 6% 5% | 12% 3% 0% 1% |
| GENERAL DISORDERS Fatigue† Irritability | 16% 9% | 13% 5% | 1% 4% |
| CARDIAC DISORDERS Dizziness Bradycardia | 7% 0% | 3% 4% | 5% 0% |
| INVESTIGATIONS Increased Heart Rate | 0% | 3% | 0% |
| METABOLISM AND NUTRITION DISORDERS Decreased Appetite |
3% |
4% |
4% |
Commonly observed adverse reactions (incidence of >2% in either active treatment group and greater than the rate on placebo) during the taper period are listed in Table 3.
|
|||
| Percentage of Patients Reporting Event
|
|||
| Preferred Term
| KAPVAY 0.2 mg/day N=76 | KAPVAY 0.4 mg/day N=78 | Placebo
(N=76) |
| Abdominal Pain Upper | 0% | 6% | 3% |
| Headache | 5% | 2% | 3% |
| Gastrointestinal Viral | 0% | 5% | 0% |
| Somnolence | 2% | 3% | 0% |
| Heart Rate Increased | 0% | 3% | 0% |
| Otitis Media Acute | 3% | 0% | 0% |
Study 2: Flexible-dose KAPVAY as Adjunctive Therapy to Psychostimulants
Study 2 (CLON-302) was a short-term, randomized, double-blind, placebo-controlled study of a flexible dose of KAPVAY as adjunctive therapy to a psychostimulant in children and adolescents (6 to 17 years) who met DSM-IV criteria for ADHD hyperactive or combined inattentive/hyperactive subtypes during which KAPVAY was initiated at 0.1 mg/day and titrated up to 0.4 mg/day over a 3-week period. Most KAPVAY treated patients (75.5%) were escalated to the maximum dose of 0.4 mg/day.
Most Common Adverse Reactions (incidence of ≥5% and at least twice the rate of placebo): somnolence, fatigue, decreased appetite, dizziness.
Adverse Events Leading to Discontinuation - There was one patient in the CLON+STM group (1%) who discontinued because of an adverse event (severe bradyphrenia, with severe fatigue).
Commonly observed adverse reactions (incidence of ≥2% in the treatment group and greater than the rate on placebo) during the treatment period are listed in Table 4.
| Percentage of Patients Reporting Event |
||
| Preferred Term |
KAPVAY+STM (N=102) |
PBO+STM (N=96) |
| PSYCHIATRIC DISORDERS Somnolence* Aggression Affect Lability Emotional Disorder |
19% 2% 2% 2% |
7% 1% 1% 0% |
| GENERAL DISORDERS Fatigue† Irritability |
14% 2% |
4% 7% |
| NERVOUS SYSTEM DISORDERS Headache Insomnia |
7% 4% |
12% 3% |
| GASTROINTESTINAL DISORDERS Upper Abdominal Pain |
7% |
4% |
| RESPIRATORY DISORDERS Nasal Congestion |
2% |
2% |
| METABOLISM AND NUTRITION DISORDERS Decreased Appetite |
6% |
3% |
| CARDIAC DISORDERS Dizziness |
5% |
1% |
Commonly observed adverse reactions (incidence of ≥2% in the treatment group and greater than the rate on placebo) during the taper period are listed in Table 5.
|
||
| Percentage of Patients Reporting Event
|
||
| Preferred Term
| KAPVAY+STM
(N=102) | PBO+STM
(N=96) |
| Nasal Congestion | 4% | 2% |
| Headache | 3% | 1% |
| Irritability | 3% | 2% |
| Throat Pain | 3% | 1% |
| Gastroenteritis Viral | 2% | 0% |
| Rash | 2% | 0% |
Adverse Reactions Leading to Discontinuation
Thirteen percent (13%) of patients receiving KAPVAY discontinued from the pediatric monotherapy study due to adverse events, compared to 1% in the placebo group. The most common adverse reactions leading to discontinuation of KAPVAY monotherapy treated patients were from somnolence/sedation (5%) and fatigue (4%).
Effect on Blood Pressure and Heart Rate
In patients that completed 5 weeks of treatment in a controlled, fixed-dose monotherapy study in pediatric patients, during the treatment period the maximum placebo-subtracted mean change in systolic blood pressure was -4.0 mmHg on KAPVAY 0.2 mg/day and -8.8 mmHg on KAPVAY 0.4 mg/day. The maximum placebo-subtracted mean change in diastolic blood pressure was -4.0 mmHg on KAPVAY 0.2 mg/day and -7.3 mmHg on KAPVAY 0.4 mg/day. The maximum placebo-subtracted mean change in heart rate was -4.0 beats per minute on KAPVAY 0.2 mg/day and -7.7 beats per minute on KAPVAY 0.4 mg/day.
During the taper period of the fixed-dose monotherapy study the maximum placebo-subtracted mean change in systolic blood pressure was +3.4 mmHg on KAPVAY 0.2 mg/day and -5.6 mmHg on KAPVAY 0.4 mg/day. The maximum placebo-subtracted mean change in diastolic blood pressure was +3.3 mmHg on KAPVAY 0.2 mg/day and -5.4 mmHg on KAPVAY 0.4 mg/day. The maximum placebo-subtracted mean change in heart rate was -0.6 beats per minute on KAPVAY 0.2 mg/day and -3.0 beats per minute on KAPVAY 0.4 mg/day.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of KAPVAY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events exclude those already mentioned in 6.1:
Psychiatric: hallucinations
Cardiovascular: Q-T prolongation
7 DRUG INTERACTIONS
The following have been reported with other oral immediate release formulations of clonidine:
| Concomitant Drug Name or Drug Class | Clinical Rationale | Clinical Recommendation |
| Tricyclic antidepressants | Increase blood pressure and may counteract clonidine’s hypotensive effects | Monitor blood pressure and adjust as needed |
| Antihypertensive drugs | Potentiate clonidine’s hypotensive effects | Monitor blood pressure and adjust as needed |
| CNS depressants | Potentiate sedating effects | Avoid use |
| Drugs that affect sinus node function or AV node conduction (e.g., digitalis, calcium channel blockers, beta blockers) | Potentiate bradycardia and risk of AV block | Avoid use |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ADHD medications, including KAPVAY, during pregnancy. Healthcare providers are encouraged to register patients by calling the National Pregnancy Registry for ADHD Medications at 1-866-961-2388 or visiting https://womensmentalhealth.org/adhd-medications/.
Risk Summary
Prolonged experience with clonidine in pregnant women over several decades, based on published literature, including controlled trials, a retrospective cohort study and case reports, have not identified a drug associated risk of major birth defects, miscarriage, and adverse maternal or fetal outcomes. In animal embryofetal studies, increased resorptions were seen in rats and mice administered oral clonidine hydrochloride from implantation through organogenesis at 10 and 5 times, respectively, the maximum recommended human dose (MRHD) given to adolescents on a mg/m2 basis. No developmental effects were seen in rabbits administered oral clonidine hydrochloride during organogenesis at doses up to 3 times the MRHD (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriages in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Oral administration of clonidine hydrochloride to pregnant rabbits during the period of embryo/fetal organogenesis at doses of up to 80 mcg/kg/day (approximately 3 times the oral maximum recommended daily dose [MRHD] of 0.4 mg/day given to adolescents on a mg/m2 basis) produced no developmental effects. In pregnant rats, however, doses as low as 15 mcg/kg/day (1/3 the MRHD given to adolescents on a mg/m2 basis) were associated with increased resorptions in a study in which dams were treated continuously from 2 months prior to mating and throughout gestation. Increased resorptions were not associated with treatment at the same or at higher dose levels (up to 3 times the MRHD) when treatment of the dams was restricted to gestation days 6-15. Increases in resorptions were observed in both rats and mice at 500 mcg/kg/day (10 and 5 times the MRHD in rats and mice, respectively) or higher when the animals were treated on gestation days 1-14; 500 mcg/kg/day was the lowest dose employed in this study.
8.2 Lactation
Risk Summary
Based on published lactation studies, clonidine hydrochloride is present in human milk at relative infant doses ranging from 4.1 to 8.4% of the maternal weight-adjusted dosage. Although in most cases, there were no reported adverse effects in breastfed infants exposed to clonidine, there is one case report of sedation, hypotonia, and apnea in an infant exposed to clonidine through breast milk. If an infant is exposed to clonidine hydrochloride through breastmilk, monitor for symptoms of hypotension and bradycardia, such as sedation, lethargy, tachypnea and poor feeding (see Clinical Considerations).The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for KAPVAY and any potential adverse effects on the breastfed child from KAPVAY or from the underlying maternal condition. Exercise caution when KAPVAY is administered to a nursing woman.
Clinical Considerations
Monitor breastfeeding infants exposed to KAPVAY through breast milk for symptoms of hypotension and/or bradycardia such as sedation, lethargy, tachypnea, and poor feeding.
8.3 Females and Males of Reproductive Potential
Infertility
Based on findings in Animal studies revealed that KAPVAY may impair fertility in females and males of reproductive potential [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and efficacy of KAPVAY in the treatment of ADHD have been established in pediatric patients 6 to 17 years of age. Use of KAPVAY in pediatric patients 6 to 17 years of age is supported by three adequate and well-controlled studies; a short-term, placebo-controlled monotherapy trial, a short-term adjunctive therapy trial and a longer-term randomized monotherapy trial [see Clinical Studies (14)]. Safety and efficacy in pediatric patients below the age of 6 years has not been established.
Juvenile Animal Data
In studies in juvenile rats, clonidine hydrochloride alone or in combination with methylphenidate had an effect on bone growth at clinically relevant doses and produced a slight delay in sexual maturation in males at 3 times the maximum recommended human dose (MRHD) for clonidine and methylphenidate.
In a study where juvenile rats were treated orally with clonidine hydrochloride from day 21 of age to adulthood, a slight delay in onset of preputial separation (delayed sexual maturation) was seen in males treated with 300 mcg/kg/day, which is approximately 3 times the MRHD of 0.4 mg/day on a mg/m2 basis. The no-effect dose was 100 mcg/kg/day, which is approximately equal to the MRHD. There was no drug effects on fertility or on other measures of sexual or neurobehavioral development.
In a study where juvenile rats were treated with clonidine alone (300 mcg/kg/day) or in combination with methylphenidate (10 mg/kg/day in females and 50/30 mg/kg/day in males; the dose was lowered from 50 to 30 mg/kg/day in males due to self-injurious behavior during the first week of treatment) from day 21 of age to adulthood, decreases in bone mineral density and mineral content were observed in males treated with 300 mcg/kg/day clonidine alone and in combination with 50/30 mg/kg/day methylphenidate and a decrease in femur length was observed in males treated with the combination at the end of the treatment period. These doses are approximately 3 times the MRHD of 0.4 mg/day clonidine and 54 mg/day methylphenidate on a mg/m2 basis. All these effects in male were not reversed at the end of a 4-week recovery period. In addition, similar findings were seen in males treated with a lower dose of clonidine (30 mcg/kg/day) in combination with 50 mg/kg/day of methylphenidate and a decrease in femur length was observed in females treated with clonidine alone at the end of the recovery period. These effects were accompanied by a decrease in body weight gain in treated animals during the treatment period but the effect was reversed at the end of the recovery period. A delay in preputial separation (sexual maturation) was observed in males treated with the combination treatment of 300 mcg/kg/day clonidine and 50/30 mg/kg/day methylphenidate. There was no effect on reproduction or sperm analysis in these males.
8.6 Renal Impairment
The impact of renal impairment on the pharmacokinetics of clonidine in children has not been assessed. The initial dosage of KAPVAY should be based on degree of impairment. Monitor patients carefully for hypotension and bradycardia, and titrate to higher doses cautiously. Since only a minimal amount of clonidine is removed during routine hemodialysis, there is no need to give supplemental KAPVAY following dialysis.
10 OVERDOSAGE
Symptoms
Clonidine overdose: hypertension may develop early and may be followed by hypotension, bradycardia, respiratory depression, hypothermia, drowsiness, decreased or absent reflexes, weakness, irritability and miosis. The frequency of CNS depression may be higher in children than adults. Large overdoses may result in reversible cardiac conduction defects or dysrhythmias, apnea, coma and seizures. Signs and symptoms of overdose generally occur within 30 minutes to two hours after exposure.
Treatment
Consult with a Certified Poison Control Center (1-800-222-1222) for up-to-date guidance and advice.
11 DESCRIPTION
KAPVAY (clonidine hydrochloride) extended-release is a centrally acting alpha2-adrenergic agonist available as 0.1 mg extended-release tablets for oral administration. Each 0.1 mg tablet is equivalent to 0.087 mg of the free base.
The inactive ingredients are sodium lauryl sulfate, lactose monohydrate, hypromellose type 2208, partially pregelatinized starch, colloidal silicon dioxide, and magnesium stearate. The formulation is designed to delay the absorption of active drug in order to decrease peak to trough plasma concentration differences. Clonidine hydrochloride is an imidazoline derivative and exists as a mesomeric compound. The chemical name is 2-(2,6-dichlorophenylamino)-2-imidazoline hydrochloride. The following is the structural formula:
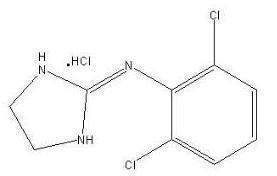
C9H9Cl2N3•HCl Mol. Wt. 266.56
Clonidine hydrochloride is an odorless, bitter, white, crystalline substance soluble in water and alcohol.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Clonidine stimulates alpha2-adrenergic receptors in the brain. Clonidine is not a central nervous system stimulant. The mechanism of action of clonidine in ADHD is not known.
12.2 Pharmacodynamics
Clonidine is a known antihypertensive agent. By stimulating alpha2-adrenergic receptors in the brain stem, clonidine reduces sympathetic outflow from the central nervous system and decreases peripheral resistance, renal vascular resistance, heart rate, and blood pressure.
12.3 Pharmacokinetics
Single-dose Pharmacokinetics in Adults
Immediate-release clonidine hydrochloride and KAPVAY have different pharmacokinetic characteristics; dose substitution on a milligram for milligram basis will result in differences in exposure. A comparison across studies suggests that the Cmax is 50% lower for KAPVAY compared to immediate-release clonidine hydrochloride.
Following oral administration of an immediate release formulation, plasma clonidine concentration peaks in approximately 3 to 5 hours and the plasma half-life ranges from 12 to 16 hours. The half-life increases up to 41 hours in patients with severe impairment of renal function. Following oral administration about 40-60% of the absorbed dose is recovered in the urine as unchanged drug in 24 hours.
About 50% of the absorbed dose is metabolized in the liver. Although studies of the effect of renal impairment and studies of clonidine excretion have not been performed with KAPVAY, results are likely to be similar to those of the immediate release formulation.
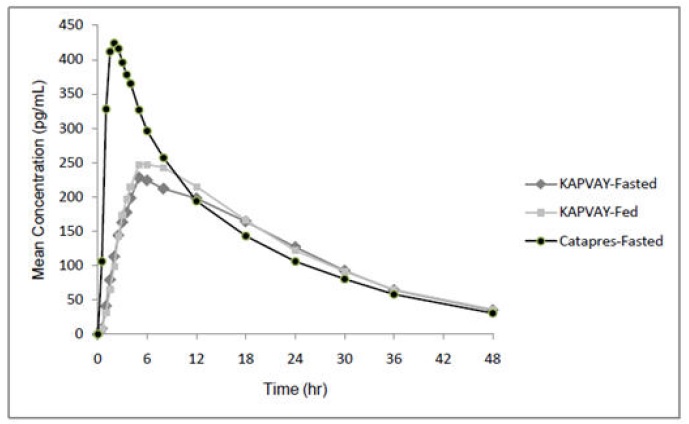
The pharmacokinetic profile of KAPVAY administration was evaluated in an open-label, three-period, randomized, crossover study of 15 healthy adult subjects who received three single-dose regimens of clonidine: 0.1 mg of KAPVAY under fasted conditions, 0.1 mg of KAPVAY following a high fat meal, and 0.1 mg of clonidine immediate-release (Catapres®) under fasted conditions. Treatments were separated by one-week washout periods.
Mean concentration-time data from the 3 treatments are shown in Table 7 and Figure 1. After administration of KAPVAY, maximum clonidine concentrations were approximately 50% of the Catapres maximum concentrations and occurred approximately 5 hours later relative to Catapres. Similar elimination half-lives were observed and total systemic bioavailability following KAPVAY was approximately 89% of that following Catapres.
Food had no effect on plasma concentrations, bioavailability, or elimination half-life.
| CATAPRES-Fasted
n=15 | KAPVAY-Fed
n=15 | KAPVAY-Fasted
n=14 |
||||
| Parameter
| Mean
| SD
| Mean
| SD
| MEAN
| SD
|
| Cmax (pg/mL) | 443 | 59.6 | 235 | 34.7 | 258 | 33.3 |
| AUCinf
(hr*pg/mL) | 7313 | 1812 | 6505 | 1728 | 6729 | 1650 |
| hTmax (hr) | 2.07 | 0.5 | 6.80 | 3.61 | 6.50 | 1.23 |
| T1/2 (hr) | 12.57 | 3.11 | 12.67 | 3.76 | 12.65 | 3.56 |
Figure 1 Mean Clonidine Concentration-Time Profiles after Single Dose Administration
Multiple-dose Pharmacokinetics in Children and Adolescents
Plasma clonidine concentrations in children and adolescents (0.1 mg bid and 0.2 mg bid) with ADHD are greater than those of adults with hypertension with children and adolescents receiving higher doses on a mg/kg basis. Body weight normalized clearance (CL/F) in children and adolescents was higher than CL/F observed in adults with hypertension. Clonidine concentrations in plasma increased with increases in dose over the dose range of 0.2 to 0.4 mg/day. Clonidine CL/F was independent of dose administered over the 0.2 to 0.4 mg/day dose range. Clonidine CL/F appeared to decrease slightly with increases in age over the range of 6 to 17 years, and females had a 23% lower CL/F than males. The incidence of "sedation-like" AEs (somnolence and fatigue) appeared to be independent of clonidine dose or concentration within the studied dose range in the titration study. Results from the add-on study showed that clonidine CL/F was 11% higher in patients who were receiving methylphenidate and 44% lower in those receiving amphetamine compared to subjects not on adjunctive therapy.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis and Impairment of Fertility
Carcinogenesis
Clonidine HCl was not carcinogenic when administered in the diet of rats (for up to 132 weeks) or mice (for up to 78 weeks) at doses of up to 1620 (male rats), 2040 (female rats), or 2500 (mice) mcg/kg/day. These doses are approximately 20, 25, and 15 times, respectively, the maximum recommended human dose (MRHD) of 0.4 mg/day on a mg/m2 basis.
Mutagenesis
There was no evidence of genotoxicity in the Ames test for mutagenicity or mouse micronucleus test for clastogenicity.
Impairment of Fertility
In a reproduction study fertility of female rats appeared to be adversely affected at dose levels of 500 and 2000 mcg/kg/day (10 and 40 times the MRHD on a mg/m2 basis). Lower doses have not been adequately evaluated and a no adverse effect level could not be established.
14 CLINICAL STUDIES
Efficacy of KAPVAY in the treatment of ADHD was established in children and adolescents (6 to 17 years) in:
- One short-term, placebo-controlled monotherapy trial (Study 1)
- One short-term adjunctive therapy to psychostimulants trial (Study 2)
- One randomized withdrawal trial as monotherapy (Study 3)
Short-term Monotherapy and Adjunctive Therapy to Psychostimulant Studies for ADHD
The efficacy of KAPVAY in the treatment of ADHD was established in 2 (one monotherapy and one adjunctive therapy) placebo-controlled trials in pediatric patients aged 6 to 17, who met DSM-IV criteria of ADHD hyperactive or combined hyperactive/inattentive subtypes. Signs and symptoms of ADHD were evaluated using the investigator administered and scored ADHD Rating Scale-IV-Parent Version (ADHDRS-IV) total score including hyperactive/impulsivity and inattentive subscales.
Study 1 (CLON-301), was an 8-week randomized, double-blind, placebo-controlled, fixed dose study of children and adolescents aged 6 to 17 (N=236) with a 5-week primary efficacy endpoint. Patients were randomly assigned to one of the following three treatment groups: KAPVAY (CLON) 0.2 mg/day (N=78), KAPVAY 0.4 mg/day (N=80), or placebo (N=78). Dosing for the KAPVAY groups started at 0.1 mg/day and was titrated in increments of 0.1 mg/week to their respective dose (as divided doses). Patients were maintained at their dose for a minimum of 2 weeks before being gradually tapered down to 0.1 mg/day at the last week of treatment. At both doses, improvements in ADHD symptoms were statistically significantly superior in KAPVAY-treated patients compared with placebo-treated patients at the end of 5 weeks as measured by the ADHDRS-IV total score (Table 8).
Study 2 (CLON-302) was an 8-week randomized, double-blind, placebo-controlled, flexible dose study in children and adolescents aged 6 to 17 (N=198) with a 5-week primary efficacy end point. Patients had been treated with a psychostimulant (methylphenidate or amphetamine) for four weeks with inadequate response. Patients were randomly assigned to one of two treatment groups: KAPVAY adjunct to a psychostimulant (N=102) or psychostimulant alone (N=96). The KAPVAY dose was initiated at 0.1 mg/day and doses were titrated in increments of 0.1 mg/week up to 0.4 mg/day, as divided doses, over a 3-week period based on tolerability and clinical response. The dose was maintained for a minimum of 2 weeks before being gradually tapered to 0.1 mg/day at the last week of treatment. ADHD symptoms were statistically significantly improved in KAPVAY plus stimulant group compared with the stimulant alone group at the end of 5 weeks as measured by the ADHDRS-IV total score (Table 8).
| Study Number
| Treatment Group
| Primary Efficacy Measure: ADHDRS-IV Total Score |
||
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo-subtracted Differencea (95% CI) |
||
| Study 1 | KAPVAY (0.2 mg/day) | 43.8 (7.47) | -15.0 (1.38) | -8.5 (-12.2, - 4.8) |
| KAPVAY (0.4 mg/day) | 44.6 (7.73) | -15.6 (1.33) | -9.1 (-12.8, - 5.5) |
|
| Placebo | 45.0 (8.53) | -6.5 (1.35) | -- |
|
| Study 2 | KAPVAY (0.4 mg/day) + Psychostimulant | 38.9 (6.95) | -15.8 (1.18) | -4.5 (-7.8, -1.1) |
| Psychostimulant alone | 39.0 (7.68) | -11.3 (1.24) | -- |
|
SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: unadjusted confidence interval.
a Difference (drug minus placebo) in least-squares mean change from baseline.
Maintenance Monotherapy for ADHD
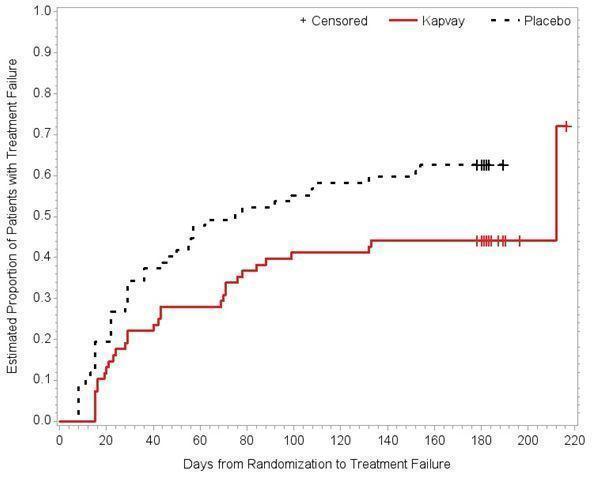
Study 3 (SHN-KAP-401), was a double-blind, placebo-controlled, randomized-withdrawal study in children and adolescents aged 6 to 17 years (n=253) with DSM-IV-TR diagnosis of ADHD. The study consisted of a 10-week, open-label phase (4 weeks of dose optimization and 6 weeks of dose maintenance), a 26-week double-blind phase, and a 4-week taper-down and follow-up phase. All patients were initiated at 0.1 mg/day and increased at weekly intervals in increments of 0.1 mg/day until reaching personalized optimal dose (0.1, 0.2, 0.3 or 0.4 mg/day, as divided doses). Eligible patients had to demonstrate treatment response as defined by ≥ 30% reduction in ADHD-RS-IV total score and a Clinical Global Impression-Improvement score of 1 or 2 during the open label phase. Patients who sustained treatment response (n=135) until the end of the open label phase were randomly assigned to one of the two treatment groups, KAPVAY (N=68) and Placebo (N=67), to evaluate the long-term efficacy of maintenance dose of KAPVAY in the double-blind phase. The primary efficacy endpoint was the percentage of patients with treatment failure defined as a ≥ 30% increase (worsening) in ADHD-RS-IV total score and ≥ 2 points increase (worsening) in Clinical Global Impression – Severity Scale in 2 consecutive visits or early termination for any reason. A total of 73 patients experienced treatment failure in the double-blind phase: 31 patients (45.6%) in the KAPVAY group and 42 patients (62.7%) in the placebo group, with a statistically significant difference in the primary endpoint favoring KAPVAY (Table 9). The cumulative proportion of patients with treatment failure over time during the double-blind phase is displayed in Figure 2.
| Study 3 | Double-Blind Full Analysis Set |
|
| Kapvay® | Placebo |
|
| Number of subjects | 68 | 67 |
| Number of treatment failures | 31 (45.6%) | 42 (62.7%) |
| Basis of Treatment Failure |
||
| Clinical criteriaa,b
| 11 (16.2%) | 9 (13.4%) |
| Lack of efficacyc
| 1 (1.5%) | 3 (4.5%) |
| Withdrawal of informed assent/consent | 4 (5.9%) | 20 (29.9%) |
| Other early terminations | 15 (22.1%) | 10 (14.9%) |
ADHD-RS-IV = Attention Deficit Hyperactivity Disorder-Rating Scale-4th edition; CGI-S = Clinical Global Impression-Severity
a At the same 2 consecutive visits a (1) 30% or greater reduction in ADHD-RS-IV, and (2) 2-point or more increase in CGI-S.
b Two subjects (1 placebo and 1 KAPVAY) withdrew consent, but met the clinical criteria for treatment failure.
c Three subjects (all placebo) discontinued the study due to treatment failure, but met only the criterion for ADHD-RS-IV.
Figure 2: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Treatment Failure (Study 3)
16 HOW SUPPLIED/STORAGE AND HANDLING
KAPVAY extended-release tablets are round, white, non-scored, standard convex with debossing "651" on one side.
NDC 59212-658-60 and NDC 59212-658-13 - 0.1 mg tablets supplied in a carton containing one bottle of 60 tablets.
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
Dispense in a tight container.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved Patient Labeling (Patient Information)
Dosage and Administration
Advise patients that KAPVAY must be swallowed whole, never crushed, cut, or chewed, and may be taken with or without food. When initiating treatment, provide dosage escalation instructions [see Dosage and Administration (2.1)].
Missed Dose
If patients miss a dose of KAPVAY, advise them to skip the dose and take the next dose as scheduled and not to take more than the prescribed total daily amount of KAPVAY in any 24-hour period [see Dosage and Administration (2.4)].
Hypotension/Bradycardia
Advise patients who have a history of syncope or may have a condition that predisposes them to syncope, such as hypotension, orthostatic hypotension, bradycardia, or dehydration, to avoid becoming dehydrated or overheated [see Warnings and Precautions (5.1)].
Sedation and Somnolence
Instruct patients to use caution when driving a car or operating hazardous machinery until they know how they will respond to treatment with KAPVAY. Also advise patients to avoid the use of KAPVAY with other centrally active depressants and with alcohol [see Warnings and Precautions (5.2)].
Rebound Hypertension
Advise patients not to discontinue KAPVAY abruptly [see Warnings and Precautions (5.3)].
Allergic Reactions
Advise patients to discontinue KAPVAY and seek immediate medical attention if any signs or symptoms of a hypersensitivity reaction occur, such as generalized rash, urticaria, or angioedema [see Warnings and Precautions (5.4)].
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in patients exposed to KAPVAY during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Advise breastfeeding women using KAPVAY to monitor infants for excess sedation, decreased muscle tone, and respiratory depression and to seek medical care if they notice these signs [see Use in Specific Populations (8.2)].
Fertility
Advise females and males of reproductive potential that KAPVAY may impair fertility [see Use in Specific Populations (8.3) and Nonclinical Toxicology (13.1)].
Patient Information
KAPVAY® (KAP-vay)
(clonidine hydrochloride) Extended-Release Tablets
Read the Patient Information that comes with KAPVAY before you start taking it and each time you get a refill. There may be new information. This Patient Information leaflet does not take the place of talking to your doctor about your medical condition or treatment.
What is KAPVAY?
KAPVAY is a prescription medicine used for the treatment of Attention-Deficit Hyperactivity Disorder (ADHD). Your doctor may prescribe KAPVAY alone or together with certain other ADHD medicines.
- KAPVAY is not a central nervous system (CNS) stimulant.
- KAPVAY should be used as part of a total treatment program for ADHD that may include counseling or other therapies.
Who should not take KAPVAY?
- Do not take KAPVAY if you are allergic to clonidine in KAPVAY. See the end of this leaflet for a complete list of ingredients in KAPVAY.
What should I tell my doctor before taking KAPVAY?
Before you take KAPVAY, tell your doctor if you:
- have kidney problems
- have low or high blood pressure
- have a history of passing out (syncope)
- have heart problems, including history of heart attack
- have had a stroke or have stroke symptoms
- had a skin reaction (such as a rash) after taking clonidine in a transdermal form (skin patch)
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if KAPVAY will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
- There is a pregnancy registry for females who are exposed to ADHD medications, including KAPVAY, during pregnancy. The purpose of the registry is to collect information about the health of females exposed to KAPVAY and their baby. If you or your child becomes pregnant during treatment with KAPVAY, talk to your healthcare provider about registering with the National Pregnancy Registry of ADHD Medications at 1-866-961-2388 or visit online at https://womensmentalhealth.org/adhdmedications/.
- are breastfeeding or plan to breastfeed. KAPVAY passes into your breast milk. Talk to your doctor about the best way to feed your baby if you take KAPVAY.
Tell your doctor about all of the medicines that you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
KAPVAY and certain other medicines may affect each other causing serious side effects. Sometimes the doses of other medicines may need to be changed while taking KAPVAY.
Especially tell your doctor if you take:
- anti-depression medicines
- heart or blood pressure medicine
- other medicines that contain clonidine
- a medicine that makes you sleepy (sedation)
Ask your doctor or pharmacist for a list of these medicines, if you are not sure if your medicine is listed above.
Know the medicines that you take. Keep a list of your medicines with you to show your doctor and pharmacist when you get a new medicine.
How should I take KAPVAY?
- Take KAPVAY exactly as your doctor tells you to take it.
- Your doctor will tell you how many KAPVAY tablets to take and when to take them. Your doctor may change your dose of KAPVAY. Do not change your dose of KAPVAY without talking to your doctor.
- Do not stop taking KAPVAY without talking to your doctor.
- KAPVAY can be taken with or without food.
- KAPVAY should be taken 2 times a day (in the morning and at bedtime).
- If you miss a dose of KAPVAY, skip the missed dose. Just take the next dose at your regular time. Do not take two doses at the same time.
- Take KAPVAY tablets whole. Do not chew, crush or break KAPVAY tablets. Tell your doctor if you cannot swallow KAPVAY tablets whole. You may need a different medicine.
- If you take too much KAPVAY, call your Poison Control Center or go to the nearest hospital emergency room right away.
What should I avoid while taking KAPVAY?
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking KAPVAY until you talk with your doctor. KAPVAY taken with alcohol or medicines that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- Do not drive, operate heavy machinery or do other dangerous activities until you know how KAPVAY will affect you.
- Avoid becoming dehydrated or overheated.
What are possible side effects of KAPVAY?
KAPVAY may cause serious side effects, including:
- Low blood pressure and low heart rate. Your doctor should check your heart rate and blood pressure before starting treatment and regularly during treatment with KAPVAY.
- Sleepiness.
- Withdrawal symptoms. Suddenly stopping KAPVAY may cause withdrawal symptoms including: increased blood pressure, headache, increased heart rate, lightheadedness, tightness in your chest and nervousness.
The most common side effects of KAPVAY include:
- sleepiness
- tiredness
- irritability
- trouble sleeping (insomnia)
- nightmare
- constipation
- dry mouth
- decreased appetite
- dizziness
Tell your doctor if you have any side effects that bother you or that does not go away.
These are not all of the possible side effects of KAPVAY. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store KAPVAY?
- Store KAPVAY between 68°-77°F (20°-25°C).
- Keep KAPVAY in a tightly closed container.
Keep KAPVAY and all medicines out of the reach of children.
General information about the safe and effective use of KAPVAY
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use KAPVAY for a condition for which it was not prescribed.
Do not give KAPVAY to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about KAPVAY. If you would like more information, talk with your doctor. You can also ask your doctor or pharmacist for information about KAPVAY that is written for healthcare professionals.
For more information about KAPVAY call 1-877-370-1142.
What are the ingredients in KAPVAY?
- Active Ingredient: clonidine hydrochloride
- Inactive Ingredients: sodium lauryl sulfate, lactose monohydrate, hypromellose type 2208, partially pregelatinized starch, colloidal silicon dioxide, and magnesium stearate
Revised: 02/2020
Mfd. for:
Concordia Pharmaceuticals
Distributed by:
Amdipharm Limited
17 Northwood House
Dublin 9, Ireland
Kapvay® is a registered trademark under exclusive license to Amdipharm Limited.
| KAPVAY
clonidine hydrochloride tablet, extended release |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Concordia Pharmaceuticals Inc. (815240092) |
