Label: CLEARSKIN PORE PENETRATING COOLING TONER- salicylic acid liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 10096-0289-1 - Packager: Avon Products, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 6, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
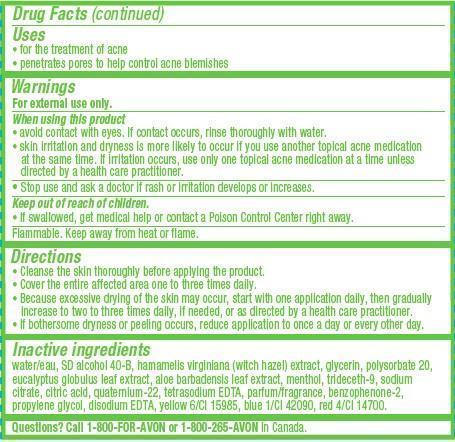
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only.When using this product
• avoid contact with eyes. If contact occurs, rinse thoroughly with water.
• skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, use only one topical acne medication at a time unless directed by a health care practitioner. -
DOSAGE & ADMINISTRATION
Directions
• Cleanse the skin thoroughly before applying the product.
• Cover the entire affected area one to three times daily.
• Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two to three times daily, if needed, or as directed by a health care practitioner.
• If bothersome dryness or peeling occurs, reduce application to once a day or every other day. -
INACTIVE INGREDIENT
Inactive ingredients
water/eau, SD alcohol 40-B, hamamelis virginiana (witch hazel) extract, glycerin, polysorbate 20, eucalyptus globulus leaf extract, aloe barbadensis leaf extract, menthol, trideceth-9, sodium citrate, citric acid, quaternium-22, tetrasodium EDTA, parfum/fragrance, benzophenone-2, propylene glycol, disodium EDTA, yellow 6/CI 15985, blue 1/CI 42090, red 4/CI 14700. - QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLEARSKIN PORE PENETRATING COOLING TONER
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10096-0289 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 5 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10096-0289-1 250 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 11/06/2012 Labeler - Avon Products, Inc. (001468693) Establishment Name Address ID/FEI Business Operations Avon Products, Inc. 005149471 manufacture(10096-0289)