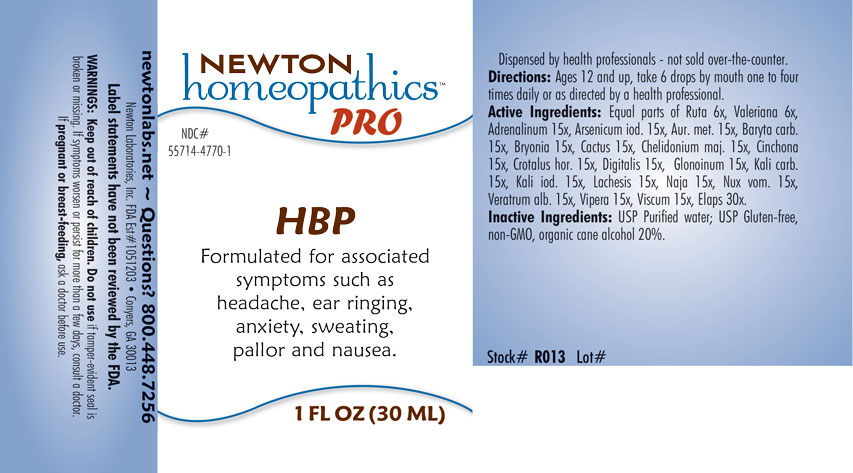
HBP- ruta graveolens, valeriana officinalis, adrenalinum, arsenicum iodatum, aurum metallicum, baryta carbonica, bryonia, cactus grandiflorus, chelidonium majus, cinchona officinalis, crotalus horridus, digitalis purpurea, glonoinum, kali carbonicum, kali iodatum, lachesis mutus, naja tripudians, nux vomica, veratrum album, vipera berus, viscum album, elaps corallinus. liquid
Newton Laboratories, Inc.
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
HBP 4841L-Dis
INDICATIONS & USAGE SECTION
Formulated for associated symptoms such as headache, ear ringing, anxiety, sweating, pallor and nausea.
DOSAGE & ADMINISTRATION SECTION
Directions: Ages 12 and up, take 6 drops by mouth one to four times daily or as directed by a health professional.
OTC - ACTIVE INGREDIENT SECTION
Equal parts of Ruta graveolens 6x, Valeriana officinalis 6x, Adrenalinum 15x, Arsenicum iodatum 15x, Aurum metallicum 15x, Baryta carbonica 15x, Bryonia 15x, Cactus grandiflorus 15x, Chelidonium majus 15x, Cinchona officinalis 15x, Crotalus horridus 15x, Digitalis purpurea 15x, Glonoinum 15x, Kali carbonicum 15x, Kali iodatum 15x, Lachesis mutus 15x, Naja tripudians 15x, Nux vomica 15x, Veratrum album 15x, Vipera berus 15x, Viscum album 15x, Elaps corallinus 30x.
OTC - PURPOSE SECTION
Formulated for associated symptoms such as headache, ear ringing, anxiety, sweating, pallor and nausea.
INACTIVE INGREDIENT SECTION
Inactive Ingredients: USP Purified Water; USP Gluten-free, non-GMO, organic cane alcohol 20%.
QUESTIONS SECTION
newtonlabs.net - Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 Conyers, GA 30013
| HBP
ruta graveolens, valeriana officinalis, adrenalinum, arsenicum iodatum, aurum metallicum, baryta carbonica, bryonia, cactus grandiflorus, chelidonium majus, cinchona officinalis, crotalus horridus, digitalis purpurea, glonoinum, kali carbonicum, kali iodatum, lachesis mutus, naja tripudians, nux vomica, veratrum album, vipera berus, viscum album, elaps corallinus. liquid |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Newton Laboratories, Inc. (788793610) |
| Registrant - Newton Laboratories, Inc. (788793610) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Newton Laboratories, Inc. | 788793610 | manufacture(55714-4841) | |