CAPEX- fluocinolone acetonide
Galderma Laboratories, L.P.
----------
Capex® Shampoo
(fluocinolone acetonide)
Topical Shampoo, 0.01%
For Topical Use Only
Rx only
Not for Ophthalmic Use
NDC 0299-5500-04
DESCRIPTION:
Capex® Shampoo 0.01% is supplied as a shampoo formulation with a 12 mg fluocinolone acetonide capsule which is emptied into the shampoo base by the pharmacist at the time of dispensing. After mixing, Capex® Shampoo contains fluocinolone acetonide(6α,9-Difluoro-11β,16α, 17,21-tetrahydroxypregna-1,4-diene-3, 20-dione cyclic 16,17-acetal with acetone), a synthetic fluorinated corticosteroid for topical dermatologic use. The corticosteroids constitute a class of primarily synthetic steroids used topically as an anti-inflammatory and antipruritic agents.
Chemically, Capex® Shampoo mixture is C24H30F2O6. It has the following structural formula:
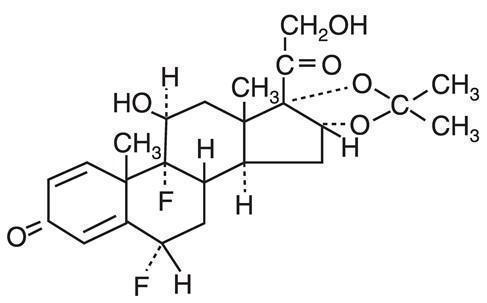
Fluocinolone acetonide in Capex® Shampoo has the molecular weight of 452.50. It is a white crystalline powder that is odorless, stable in light and melts at 270°F with decomposition; soluble in alcohol, acetone and methanol; slightly soluble in chloroform; insoluble in water.
Each fluocinolone capsule contains 12 mg of fluocinolone acetonide, 548 mg of dibasic calcium phosphate dihydrate USP, and 240 mg of talc USP. The shampoo base contains aluminum acetate dibasic, benzalkonium chloride solution, boric acid, citric acid anhydrous, cocamido-ether-sulfate complex, cocoamine oxide, lauramide DEA, magnesium aluminum silicate, methylparaben, oat flour, propylene glycol, propylparaben, purified water, and fragrances, with D&C Yellow #10 and FD&C Blue #1 as coloring.
CLINICAL PHARMACOLOGY:
Like other topical corticosteroids, fluocinolone acetonide has anti-inflammatory, antipruritic and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
Pharmacokinetics: The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle and the integrity of the epidermal barrier. Occlusive dressings with hydrocortisone for up to 24 hours have not been demonstrated to increase penetration; however, occlusion of hydrocortisone for 96 hours markedly enhances penetration. Topical corticosteroids can be absorbed from normal intact skin while inflammation and/or other disease processes in the skin increase percutaneous absorption.
Capex® Shampoo is in the low to medium range of potency as compared with other topical corticosteroids.
CLINICAL STUDIES:
In vehicle-controlled studies for the treatment of seborrheic dermatitis of the scalp, after 14 days of treatment, 84% of patients on active treatment and 29% of patients on the drug vehicle had cleared or markedly improved.
INDICATIONS AND USAGE:
Capex® Shampoo is a low to medium potency corticosteroid indicated for the treatment of seborrheic dermatitis of the scalp. This product has not been proven to be effective in other corticosteroid-responsive dermatoses.
CONTRAINDICATIONS:
Capex® Shampoo is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
PRECAUTIONS:
General: Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticoid insufficiency after withdrawal of treatment. Manifestations of Cushing’s syndrome, hyperglycemia and glucosuria can also be produced in some patients by systemic absorption of topical corticosteroids while on treatment.
Patients applying a topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA axis suppression. This may be done by using the ACTH stimulation, A.M. plasma cortisol, and urinary free cortisol tests. Patients receiving superpotent corticosteroids should not be treated for more than 2 weeks at a time and only small areas should be treated at any one time due to the increased risk of HPA suppression.
If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent corticosteroid. Infrequently, signs and symptoms of glucocorticoid insufficiency may occur requiring supplemental systemic corticosteroids. For information on systemic supplementation, see prescribing information for those products.
Pediatric patients may be more susceptible to systemic toxicity from equivalent doses due to their larger skin surface to body mass ratios. (See PRECAUTIONS-Pediatric Use).
If irritation develops, Capex® Shampoo should be discontinued and appropriate therapy instituted. Allergic contact dermatitis with corticosteroids is usually diagnosed by a failure to heal rather than noting a clinical exacerbation as with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic patch testing.
If concomitant skin infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of Capex® Shampoo should be discontinued until the infection has been adequately controlled.
Information for Patients:
Patients using topical corticosteroids should receive the following information and instructions:
-
This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes. In case of contact, wash eyes liberally with water.
-
This medication should not be used for any disorder other than that for which it was prescribed.
-
The treated scalp area should not be bandaged or otherwise covered or wrapped so as to be occlusive unless directed by the physician.
-
Patients should report to their physician any signs of local adverse reactions.
-
Discard contents after two (2) months.
Laboratory Tests:
The following tests may be helpful in evaluating patients for HPA axis suppression:
ACTH stimulation test
A.M. plasma cortisol test
Urinary free cortisol test
Carcinogenesis, mutagenesis, and impairment of fertility:
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of Capex® Shampoo.
Pregnancy:
Teratogenic effects: Pregnancy category C: Corticosteroids have been shown to be teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Some corticosteroids have been shown to be teratogenic after dermal application in laboratory animals.
There are no adequate and well-controlled studies in pregnant women or teratogenic effects from Capex® Shampoo. Therefore, Capex® Shampoo should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers:
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Because many drugs are secreted in human milk, caution should be exercised when Capex® Shampoo is administered to a nursing woman.
Pediatric Use:
Safety and effectiveness in children and infants have not been established. Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of HPA axis suppression when they are treated with topical corticosteroids. They are therefore also at a greater risk of glucocorticoid insufficiency after withdrawal of treatment and of Cushing’s syndrome while on treatment. Adverse effects including striae have been reported with inappropriate use of topical corticosteroids in infants and children.
HPA axis suppression, Cushing’s syndrome and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include linear growth retardation, delayed weight gain, low plasma cortisol levels and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches and bilateral papilledema.
ADVERSE REACTIONS:
The following local adverse reactions have been reported infrequently with topical corticosteroids. They may occur more frequently with the use of occlusive dressings, especially with higher potency corticosteroids. These reactions are listed in an approximate decreasing order of occurrence: dryness, folliculitis, acneiform eruptions, perioral dermatitis, allergic contact dermatitis, secondary infection, skin atrophy, striae, miliaria, burning, itching, irritation and hypopigmentation.
OVERDOSAGE:
Topically applied Capex® Shampoo can be absorbed in sufficient amounts to produce systemic effects (See PRECAUTIONS).
DOSAGE AND ADMINISTRATION:
The pharmacist must empty the contents of the enclosed capsule into the shampoo base prior to dispensing to the patient. This product should be shaken well prior to use. No more than approximately one (1) ounce of the medicated shampoo should be applied to the scalp area once daily, worked into a lather and allowed to remain on the scalp for approximately 5 minutes. The hair and scalp should then be rinsed thoroughly with water.
HOW SUPPLIED:
Capex® Shampoo is supplied as a two component package: a capsule which contains the active component fluocinolone acetonide, and a separate package of liquid shampoo. The pharmacist must mix the content of the capsule into the base at the time of dispensing. Capex® Shampoo is dispensed to the patient in a 6 ounce bottle.
Shake well before using.
Store at controlled room temperature 68° - 77°F (20° - 25°C) with excursions permitted between 59° - 86°F (15° - 30°C) in tightly closed containers.
Marketed by:
GALDERMA LABORATORIES, L.P.
Fort Worth, Texas 76177 USA
Made in Canada.
P52306-3
GALDERMA is a registered trademark.
Revised: December 2015
PATIENT INFORMATION
Capex® (kap-eks) Shampoo
(fluocinolone acetonide)
Topical Shampoo, 0.01%
What is Capex Shampoo?
Capex Shampoo is a prescription medicine used on the scalp to treat a skin condition called seborrheic dermatitis. It is not known if Capex Shampoo is safe and effective in children and infants.
Who should not use Capex Shampoo?
Do not use Capex Shampoo if you are allergic to any of its ingredients. See the end of this leaflet for a complete list of ingredients in Capex Shampoo.
Before using Capex Shampoo, tell your doctor about all of your medical conditions, including if you:
• have a scalp infection. You may need medicine to treat the scalp infection before you use Capex Shampoo.
• are pregnant or plan to become pregnant. It is not known if Capex shampoo can harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
• are breastfeeding or plan to breastfeed. It is not known if Capex Shampoo passes into your breast milk and if it can harm your baby. Talk to your doctor about the best way to feed you baby if you use Capex Shampoo.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your doctor if you take other corticosteroid medicines by mouth or use other products on your skin that contain corticosteroids. Do not use other products containing a corticosteroid medicine with Capex Shampoo without talking to your doctor first.
How should I use Capex Shampoo?
• When you receive your Capex Shampoo, it will already be mixed in a bottle by your pharmacist.
• Use Capex Shampoo exactly as your doctor tells you to use it.
• Your doctor should tell you how much Capex Shampoo to use and where to apply it.
• Capex Shampoo is for use on your scalp only (topical).
• Shake the Capex Shampoo bottle well before using.
• Do not apply more than 1 ounce of Capex Shampoo to your scalp, 1 time each day.
• Work Capex Shampoo into a lather.
• Leave Capex Shampoo on your scalp for at least 5 minutes.
• Rinse your hair and scalp well with water.
• Avoid getting Capex Shampoo in your eyes. If you get Capex Shampoo in your eyes, rinse your eyes well with water.
• Do not bandage or cover your head after using Capex Shampoo unless your doctor tells you to.
• Tell your doctor if you develop irritation where you apply Capex Shampoo, or if your seborrheic dermatitis is not getting better with Capex Shampoo.
What are the possible side effects of Capex Shampoo?
Capex Shampoo can pass through your skin.
The side effects of corticosteroids medicines used on the skin (topical) include:
• dry, itchy, and reddened skin
• burning or stinging at application site
• inflamed hair follicle
• acne
• thinning of skin
• loss of skin color
These are not all the possible side effects of Capex Shampoo. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Capex Shampoo?
• Store Capex Shampoo at room temperature between 68°F to 77°F (20°C to 25°C).
• Keep the Capex Shampoo bottle tightly closed.
• Throw away (discard) your Capex Shampoo bottle after 2 months, even if there is still medicine left in the bottle.
Keep Capex Shampoo and all medicines out of the reach of children.
General information about the safe and effective use of Capex Shampoo.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use Capex Shampoo for a condition for which it was not prescribed. Do not give Capex Shampoo to other people, even if they have the same symptoms you have. It may harm them. You can also ask your doctor or pharmacist for information about Capex Shampoo that is written for health professionals.
What are the ingredients in Capex Shampoo?
Active ingredient: fluocinolone acetonide
Inactive ingredients: aluminum acetate dibasic, benzalkonium chloride solution, boric acid, citric acid anhydrous, cocamido-ether-sulfate complex, cocoamine oxide, dibasic calcium phosphate dihydrate, lauramide DEA, magnesium aluminum silicate, methylparaben, oat flour, propylene glycol, propylparaben, purified water, talc, fragrances, D&C Yellow 10, and FD&C Blue 1 as coloring.
PACKAGE LABEL

ATTN PHARMACIST: Empty contents of enclosed capsule into shampoo bottle.
Rx only
NDC 0299-5500-04
Capex®
(fluocinolone acetonide)
topical shampoo
SHAMPOO (as dispensed)
0.01%
For Topical Use Only
Shake Well Before Use
Discard Contents After Two Months
4 fl oz
GALDERMA
ATTN PHARMACIST: Empty contents of enclosed capsule into shampoo bottle.
Usual Dose: Apply no more than approximately one ounce of shampoo on affected area, let stand for 5 minutes, then rinse thoroughly with water.
See package insert for full prescribing information.
Marketed by:
GALDERMA LABORATORIES, L.P.
Fort Worth, Texas 76177 USA
Made in Canada.
All trademarks are the property of their respective owners.
P52307-3
Contains: (As dispensed) Fluocinolone Acetonide (0.01%), dibasic calcium phosphate dihydrate USP, talc USP, in a shampoo base containing aluminum acetate dibasic, benzalkonium chloride solution, boric acid, citric acid anhydrous, cocamido-ether sulfate complex, cocoamine oxide, lauramide DEA, magnesium aluminum silicate, methylparaben, oat flour, propylene glycol, propylparaben, purified water and fragrance with D & C Yellow No. 10 and FD&C Blue No. 1 as coloring.
Storage: Keep tightly closed. Store at controlled room temperature 68° - 77°F (20° - 25°C) with excursions permitted between 59° - 86°F (15° - 30°C)
Contents: 4 fl oz in 6 oz bottle for convenience of Pharmacist.
For External Use Only.
Keep out of reach of children.
Warning: In case of eye contact rinse eye thoroughly with water.
| CAPEX
fluocinolone acetonide kit |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| Labeler - Galderma Laboratories, L.P. (047350186) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Confab Laboratories | 241754217 | MANUFACTURE(0299-5500) | |