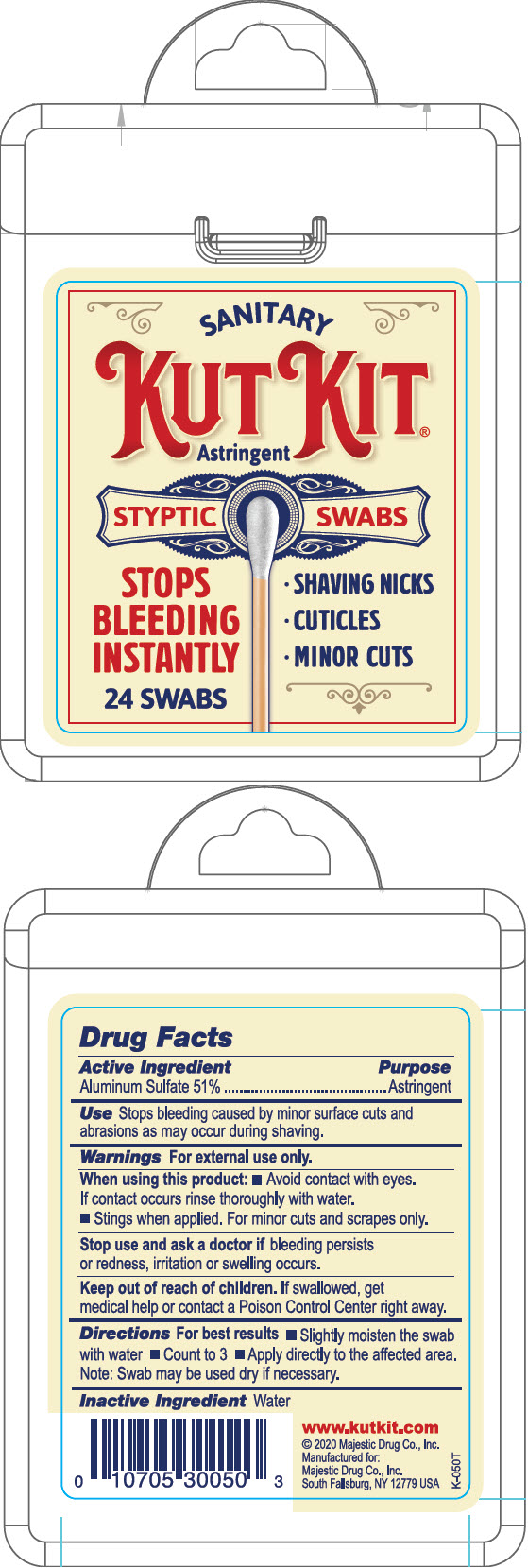
KUTKIT- aluminum sulfate swab
Majestic Drug Co., Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
KutKit®
Warnings
For external use only.
| KUTKIT
aluminum sulfate swab |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Majestic Drug Co., Inc. (001496777) |
Revised: 1/2022
Document Id: 635fb007-7807-4757-a275-7ac37e67d72c
Set id: a29460b5-7dd2-479d-9b4b-5ab8dc135ad4
Version: 3
Effective Time: 20220124
Majestic Drug Co., Inc.