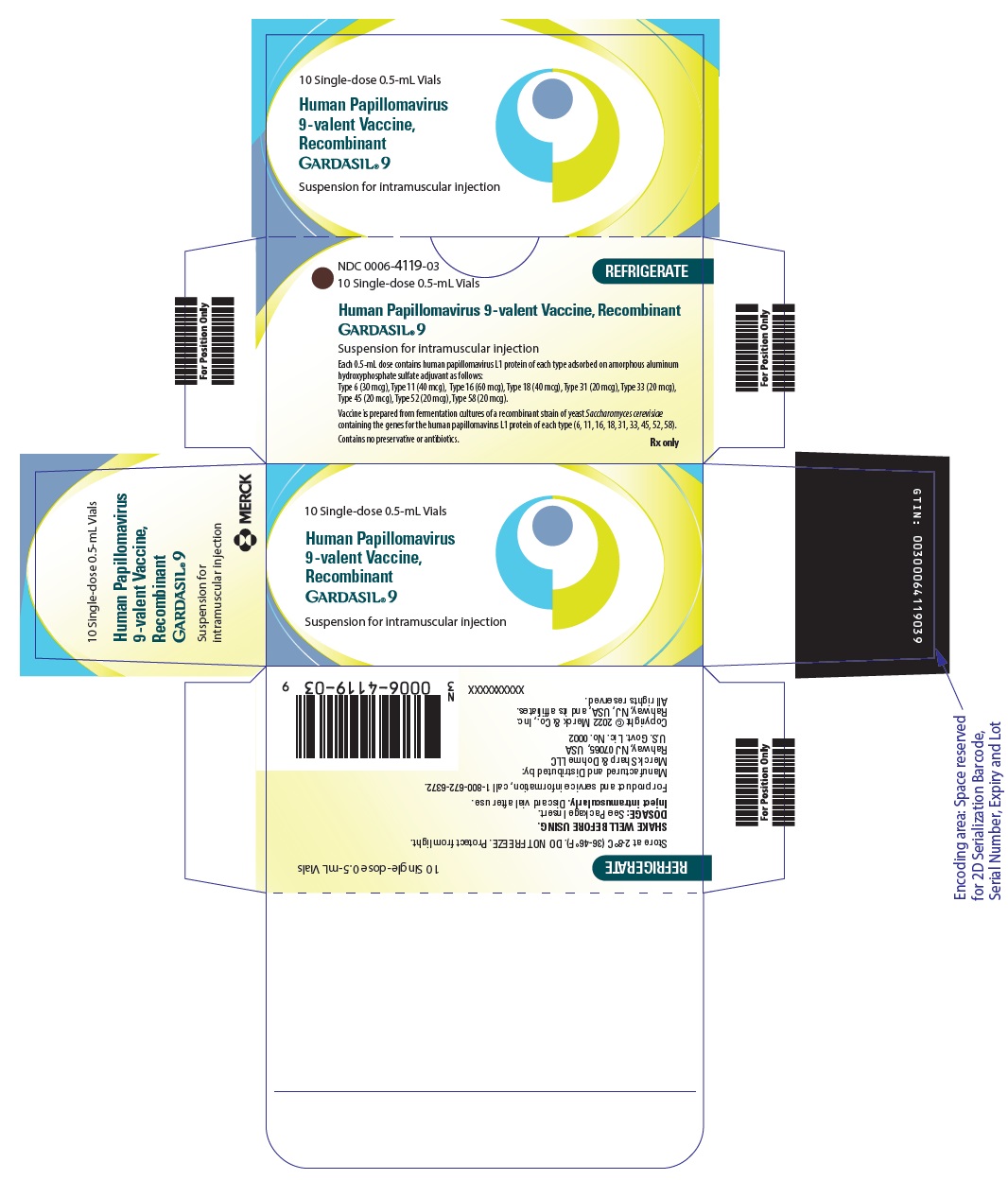
Label: GARDASIL 9- human papillomavirus 9-valent vaccine, recombinant injection, suspension
- NDC Code(s): 0006-4119-01, 0006-4119-03, 0006-4121-01, 0006-4121-02
- Packager: Merck Sharp & Dohme LLC
- Category: VACCINE LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated March 14, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use GARDASIL 9 safely and effectively. See full prescribing information for GARDASIL 9.
GARDASIL®9
(Human Papillomavirus 9-valent Vaccine, Recombinant)
Suspension for intramuscular injection
Initial U.S. Approval: 2014INDICATIONS AND USAGE
GARDASIL 9 is a vaccine indicated in girls and women 9 through 45 years of age for the prevention of the following diseases:
- Cervical, vulvar, vaginal, anal, oropharyngeal and other head and neck cancers caused by Human Papillomavirus (HPV) types 16, 18, 31, 33, 45, 52, and 58. (1.1)
- Genital warts (condyloma acuminata) caused by HPV types 6 and 11. (1.1)
And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58:
- Cervical intraepithelial neoplasia (CIN) grade 2/3 and cervical adenocarcinoma in situ (AIS). (1.1)
- Cervical intraepithelial neoplasia (CIN) grade 1. (1.1)
- Vulvar intraepithelial neoplasia (VIN) grade 2 and grade 3. (1.1)
- Vaginal intraepithelial neoplasia (VaIN) grade 2 and grade 3. (1.1)
- Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3. (1.1)
GARDASIL 9 is indicated in boys and men 9 through 45 years of age for the prevention of the following diseases:
- Anal, oropharyngeal and other head and neck cancers caused by HPV types 16, 18, 31, 33, 45, 52, and 58. (1.2)
- Genital warts (condyloma acuminata) caused by HPV types 6 and 11. (1.2)
And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58:
- Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3. (1.2)
The oropharyngeal and head and neck cancer indication is approved under accelerated approval based on effectiveness in preventing HPV- related anogenital disease. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial (1).
Limitations of Use and Effectiveness:- Vaccination with GARDASIL 9 does not eliminate the necessity for vaccine recipients to undergo screening for cervical, vulvar, vaginal, anal, oropharyngeal and other head and neck cancers as recommended by a health care provider. (1.3, 17)
- GARDASIL 9 has not been demonstrated to provide protection against disease caused by:
- HPV types not covered by the vaccine
- HPV types to which a person has previously been exposed through sexual activity. (1.3)
- Not all vulvar, vaginal, anal, oropharyngeal and other head and neck cancers are caused by HPV, and GARDASIL 9 protects only against those vulvar, vaginal, anal, oropharyngeal and other head and neck cancers caused by HPV 16, 18, 31, 33, 45, 52, and 58. (1.3)
- GARDASIL 9 is not a treatment for external genital lesions; cervical, vulvar, vaginal, anal, oropharyngeal and other head and neck cancers; CIN; VIN; VaIN; or AIN. (1.3)
- Vaccination with GARDASIL 9 may not result in protection in all vaccine recipients. (1.3)
DOSAGE AND ADMINISTRATION
For intramuscular administration only. (2)
Each dose of GARDASIL 9 is 0.5-mL
Administer GARDASIL 9 as follows: (2.1)
Age Regimen Schedule 9 through 14 years 2-dose 0, 6 to 12 months* 3-dose 0, 2, 6 months 15 through 45 years 3-dose 0, 2, 6 months DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
Because vaccinees may develop syncope, sometimes resulting in falling with injury, observation for 15 minutes after administration is recommended. Syncope, sometimes associated with tonic-clonic movements and other seizure-like activity, has been reported following HPV vaccination. When syncope is associated with tonic-clonic movements, the activity is usually transient and typically responds to restoring cerebral perfusion by maintaining a supine or Trendelenburg position. (5.1)
ADVERSE REACTIONS
The most common (≥10%) local and systemic adverse reactions reported:
- In girls and women 16 through 26 years of age: injection-site pain (89.9%), injection-site swelling (40.0%), injection-site erythema (34.0%) and headache (14.6%). (6.1)
- In girls 9 through 15 years of age: injection-site pain (89.3%), injection-site swelling (47.8%), injection-site erythema (34.1%) and headache (11.4%). (6.1)
- In women 27 through 45 years of age: injection-site pain (82.8%), injection-site swelling (23.3%), injection-site erythema (16.9%), and headache (13.6%) (6.1)
- In boys and men 16 through 26 years of age: injection-site pain (63.4%), injection-site swelling (20.2%) and injection-site erythema (20.7%). (6.1)
- In boys 9 through 15 years of age: injection-site pain (71.5%), injection-site swelling (26.9%), and injection-site erythema (24.9%). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or VAERS at 1-800-822-7967 or www.vaers.hhs.gov .
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Girls and Women
1.2 Boys and Men
1.3 Limitations of Use and Effectiveness
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
2.2 Method of Administration
2.3 Administration of GARDASIL 9 in Individuals Who Have Been Previously Vaccinated with GARDASIL®
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Syncope
5.2 Managing Allergic Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Use with Systemic Immunosuppressive Medications
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Immunocompromised Individuals
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Efficacy and Effectiveness Data for GARDASIL
14.2 Clinical Trials for GARDASIL 9
14.3 Efficacy – HPV Types 31, 33, 45, 52 and 58 in Girls and Women 16 through 26 Years of Age
14.4 Long term Follow-up of Individuals Vaccinated with GARDASIL 9
14.5 Effectiveness in Prevention of HPV-Related Oropharyngeal and Other Head and Neck Cancers
14.6 Immunogenicity of a 3-Dose Regimen
14.7 Immune Responses to GARDASIL 9 Using a 2-Dose Regimen in Individuals 9 through 14 Years of Age
14.8 Studies with Menactra and Adacel
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Girls and Women
GARDASIL®9 is a vaccine indicated in girls and women 9 through 45 years of age for the prevention of the following diseases:
- Cervical, vulvar, vaginal, anal, oropharyngeal and other head and neck cancers caused by Human Papillomavirus (HPV) types 16, 18, 31, 33, 45, 52, and 58
- Genital warts (condyloma acuminata) caused by HPV types 6 and 11
And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58:
- Cervical intraepithelial neoplasia (CIN) grade 2/3 and cervical adenocarcinoma in situ (AIS)
- Cervical intraepithelial neoplasia (CIN) grade 1
- Vulvar intraepithelial neoplasia (VIN) grade 2 and grade 3
- Vaginal intraepithelial neoplasia (VaIN) grade 2 and grade 3
- Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3
1.2 Boys and Men
GARDASIL 9 is indicated in boys and men 9 through 45 years of age for the prevention of the following diseases:
- Anal, oropharyngeal and other head and neck cancers caused by HPV types 16, 18, 31, 33, 45, 52, and 58
- Genital warts (condyloma acuminata) caused by HPV types 6 and 11
And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58:
- Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3
The oropharyngeal and head and neck cancer indication is approved under accelerated approval based on effectiveness in preventing HPV-related anogenital disease [see Clinical Studies (14.5)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
1.3 Limitations of Use and Effectiveness
- Vaccination with GARDASIL 9 does not eliminate the necessity for vaccine recipients to undergo screening for cervical, vulvar, vaginal, anal, oropharyngeal and other head and neck cancers as recommended by a health care provider.
- GARDASIL 9 has not been demonstrated to provide protection against disease caused by:
- HPV types not covered by the vaccine [see Description (11)],
- HPV types to which a person has previously been exposed through sexual activity.
- Not all vulvar, vaginal, anal, oropharyngeal and other head and neck cancers are caused by HPV, and GARDASIL 9 protects only against those vulvar, vaginal, anal, oropharyngeal and other head and neck cancers caused by HPV 16, 18, 31, 33, 45, 52, and 58.
- GARDASIL 9 is not a treatment for external genital lesions; cervical, vulvar, vaginal, anal, oropharyngeal and other head and neck cancers; CIN; VIN; VaIN; or AIN.
- Vaccination with GARDASIL 9 may not result in protection in all vaccine recipients.
-
2 DOSAGE AND ADMINISTRATION
For intramuscular use only
2.1 Dosage
Each dose of GARDASIL 9 is 0.5-mL.
Administer GARDASIL 9 as follows:
Age Regimen Schedule - *
- If the second dose is administered earlier than 5 months after the first dose, administer a third dose at least 4 months after the second dose. [See Clinical Studies (14.2 and 14.7).]
9 through 14 years 2-dose 0, 6 to 12 months* 3-dose 0, 2, 6 months 15 through 45 years 3-dose 0, 2, 6 months 2.2 Method of Administration
- Do not dilute or mix GARDASIL 9 with other vaccines.
- Shake well immediately before use to maintain suspension of the vaccine.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use the product if particulates are present or if it appears discolored. After thorough agitation, GARDASIL 9 is a white cloudy liquid.
- Administer intramuscularly in the deltoid or anterolateral area of the thigh.
- Observe patients for 15 minutes after administration [see Warnings and Precautions (5)].
2.3 Administration of GARDASIL 9 in Individuals Who Have Been Previously Vaccinated with GARDASIL®
Safety and immunogenicity were assessed in individuals who completed a three-dose vaccination series with GARDASIL 9 and had previously completed a three-dose vaccination series with GARDASIL [see Adverse Reactions (6.1) and Clinical Studies (14.6)]. Studies using a mixed regimen of HPV vaccines to assess interchangeability were not performed for GARDASIL 9.
-
3 DOSAGE FORMS AND STRENGTHS
GARDASIL 9 is a suspension for intramuscular administration available in 0.5-mL single-dose vials and prefilled syringes. [See Description (11)] for the complete listing of ingredients.
-
4 CONTRAINDICATIONS
Hypersensitivity, including severe allergic reactions to yeast (a vaccine component), or after a previous dose of GARDASIL 9 or GARDASIL [see Description (11)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Syncope
Because vaccinees may develop syncope, sometimes resulting in falling with injury, observation for 15 minutes after administration is recommended. Syncope, sometimes associated with tonic-clonic movements and other seizure-like activity, has been reported following HPV vaccination. When syncope is associated with tonic-clonic movements, the activity is usually transient and typically responds to restoring cerebral perfusion by maintaining a supine or Trendelenburg position.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice.
The safety of GARDASIL 9 was evaluated in seven clinical studies that included 15,703 individuals who received at least one dose of GARDASIL 9 and had safety follow-up. Study 1 and Study 3 also included 7,378 individuals who received at least one dose of GARDASIL as a control and had safety follow-up. The vaccines were administered on the day of enrollment and the subsequent doses administered approximately two and six months thereafter. Safety was evaluated using vaccination report card (VRC)-aided surveillance for 14 days after each injection of GARDASIL 9 or GARDASIL.
The individuals who were monitored using VRC-aided surveillance included 9,097 girls and women 16 through 26 years of age, 1,394 boys and men 16 through 26 years of age, and 5,212 girls and boys 9 through 15 years of age (3,436 girls and 1,776 boys) at enrollment who received GARDASIL 9; and 7,078 girls and women 16 through 26 years of age and 300 girls 9 through 15 years of age at enrollment who received GARDASIL. The race distribution of the integrated safety population for GARDASIL 9 was similar between girls and women 16 through 26 years of age (56.8% White; 25.2% Other Races or Multiracial; 14.1% Asian; 3.9% Black), girls and boys 9 through 15 years of age (62.0% White; 19.2% Other Races or Multiracial; 13.5% Asian; 5.4% Black), and boys and men 16 through 26 years of age (62.1% White; 22.6% Other Races or Multiracial; 9.8% Asian; 5.5% Black). The safety of GARDASIL 9 was compared directly to the safety of GARDASIL in two studies (Study 1 and Study 3) for which the overall race distribution of the GARDASIL cohorts (57.0% White; 26.3% Other Races or Multiracial; 13.6% Asian; 3.2% Black) was similar to that of the GARDASIL 9 cohorts.
Safety of GARDASIL 9 in women 27 through 45 years of age was evaluated in a clinical trial comparing 640 women 27 through 45 years of age and 570 girls and women 16 through 26 years of age. The race distribution was similar between women 27 through 45 years of age (97.7% White, 1.6% Asian, 0.3% Other or Multiracial, 0.5% Black) and girls and women 16 through 26 years of age (94.6% White, 3.0% Asian, 1.6% Other or Multiracial, 0.9% Black).
Safety of GARDASIL 9 in men 27 through 45 years of age is inferred from the safety data of GARDASIL 9 in boys and men 9 through 26 years of age and girls and women 9 through 45 years of age and GARDASIL in individuals 9 through 45 years of age.
Injection-Site and Systemic Adverse Reactions
Injection-site reactions (pain, swelling, and erythema) and oral temperature were solicited using VRC-aided surveillance for five days after each injection of GARDASIL 9 during the clinical studies. The rates and severity of these solicited adverse reactions that occurred within five days following each dose of GARDASIL 9 compared with GARDASIL in Study 1 (girls and women 16 through 26 years of age) and Study 3 (girls 9 through 15 years of age) are presented in Table 1. Among subjects who received GARDASIL 9, the rates of injection-site pain were approximately equal across the three reporting time periods. Rates of injection-site swelling and injection-site erythema increased following each successive dose of GARDASIL 9. Recipients of GARDASIL 9 had numerically higher rates of injection-site reactions compared with recipients of GARDASIL.
Table 1: Rates (%) and Severity of Solicited Injection-Site and Systemic Adverse Reactions Occurring within Five Days of Each Vaccination with GARDASIL 9 Compared with GARDASIL (Studies 1 and 3) GARDASIL 9 GARDASIL Post-dose 1 Post-dose 2 Post-dose 3 Post any dose Post-dose 1 Post-dose 2 Post-dose 3 Post any dose The data for girls and women 16 through 26 years of age are from Study 1 (NCT00543543), and the data for girls 9 through 15 years of age are from Study 3 (NCT01304498). N=number of subjects vaccinated with safety follow-up n=number of subjects with temperature data Pain, Any=mild, moderate, severe or unknown intensity Pain, Severe=incapacitating with inability to work or do usual activity Swelling, Any=any size or size unknown Swelling, Severe=maximum size greater than 2 inches Erythema, Any=any size or size unknown Erythema, Severe=maximum size greater than 2 inches Girls and Women 16 through 26 Years of Age Injection-Site Adverse Reactions N=7069 N=6997 N=6909 N=7071 N=7076 N=6992 N=6909 N=7078 Pain, Any 70.7 73.5 71.6 89.9 58.2 62.2 62.6 83.5 Pain, Severe 0.7 1.7 2.6 4.3 0.4 1.0 1.7 2.6 Swelling, Any 12.5 23.3 28.3 40.0 9.3 14.6 18.7 28.8 Swelling, Severe 0.6 1.5 2.5 3.8 0.3 0.5 1.0 1.5 Erythema, Any 10.6 18.0 22.6 34.0 8.1 12.9 15.6 25.6 Erythema, Severe 0.2 0.5 1.1 1.6 0.2 0.2 0.4 0.8 Systemic Adverse Reactions n=6995 n=6913 n=6743 n=7022 n=7003 n=6914 n=6725 n=7024 Temperature ≥100°F 1.7 2.6 2.7 6.0 1.7 2.4 2.5 5.9 Temperature ≥102°F 0.3 0.3 0.4 1.0 0.2 0.3 0.3 0.8 Girls 9 through 15 Years of Age Injection-Site Adverse Reactions N=300 N=297 N=296 N=299 N=299 N=299 N=294 N=300 Pain, Any 71.7 71.0 74.3 89.3 66.2 66.2 69.4 88.3 Pain, Severe 0.7 2.0 3.0 5.7 0.7 1.3 1.7 3.3 Swelling, Any 14.0 23.9 36.1 47.8 10.4 17.7 25.2 36.0 Swelling, Severe 0.3 2.4 3.7 6.0 0.7 2.7 4.1 6.3 Erythema, Any 7.0 15.5 21.3 34.1 9.7 14.4 18.4 29.3 Erythema, Severe 0 0.3 1.4 1.7 0 0.3 1.7 2.0 Systemic Adverse Reactions n=300 n=294 n=295 n=299 n=299 n=297 n=291 n=300 Temperature ≥100°F 2.3 1.7 3.0 6.7 1.7 1.7 0 3.3 Temperature ≥102°F 0 0.3 1.0 1.3 0.3 0.3 0 0.7 Unsolicited injection-site and systemic adverse reactions (assessed as vaccine-related by the investigator) observed among recipients of either GARDASIL 9 or GARDASIL in Studies 1 and 3 at a frequency of at least 1% are shown in Table 2. Few individuals discontinued study participation due to adverse experiences after receiving either vaccine (GARDASIL 9 = 0.1% vs. GARDASIL <0.1%).
Table 2: Rates (%) of Unsolicited Injection-Site and Systemic Adverse Reactions Occurring among ≥1.0% of Individuals after Any Vaccination with GARDASIL 9 Compared with GARDASIL (Studies 1 and 3) Girls and Women 16 through 26 Years of Age Girls 9 through 15 Years of Age GARDASIL 9
N=7071GARDASIL
N=7078GARDASIL 9
N=299GARDASIL
N=300The data for girls and women 16 through 26 years of age are from Study 1 (NCT00543543), and the data for girls 9 through 15 years of age are from Study 3 (NCT01304498). N=number of subjects vaccinated with safety follow-up Injection-Site Adverse Reactions (1 to 5 Days Post-Vaccination, Any Dose) Pruritus 5.5 4.0 4.0 2.7 Bruising 1.9 1.9 0 0 Hematoma 0.9 0.6 3.7 4.7 Mass 1.3 0.6 0 0 Hemorrhage 1.0 0.7 1.0 2.0 Induration 0.8 0.2 2.0 1.0 Warmth 0.8 0.5 0.7 1.7 Reaction 0.6 0.6 0.3 1.0 Systemic Adverse Reactions (1 to 15 Days Post-Vaccination, Any Dose) Headache 14.6 13.7 11.4 11.3 Pyrexia 5.0 4.3 5.0 2.7 Nausea 4.4 3.7 3.0 3.7 Dizziness 3.0 2.8 0.7 0.7 Fatigue 2.3 2.1 0 2.7 Diarrhea 1.2 1.0 0.3 0 Oropharyngeal pain 1.0 0.6 2.7 0.7 Myalgia 1.0 0.7 0.7 0.7 Abdominal pain, upper 0.7 0.8 1.7 1.3 Upper respiratory tract infection 0.1 0.1 0.3 1.0 In an uncontrolled clinical trial with 639 boys and 1,878 girls 9 through 15 years of age (Study 2), the rates and severity of solicited adverse reactions following each dose of GARDASIL 9 were similar between boys and girls. Rates of solicited and unsolicited injection-site and systemic adverse reactions in boys 9 through 15 years of age were similar to those among girls 9 through 15 years of age. Solicited and unsolicited adverse reactions reported by boys in this study are shown in Table 3.
In another uncontrolled clinical trial with 1,394 boys and men and 1,075 girls and women 16 through 26 years of age (Study 7), the rates of solicited and unsolicited adverse reactions following each dose of GARDASIL 9 among girls and women 16 through 26 years of age were similar to those reported in Study 1. Rates of solicited and unsolicited adverse reactions reported by boys and men 16 through 26 years of age in this study are shown in Table 3.
In an uncontrolled clinical trial with 640 women 27 through 45 years of age and 570 girls and women 16 through 26 years of age (Study 9), the rates of solicited and unsolicited adverse reactions following each dose of GARDASIL 9 among girls and women 16 through 26 years of age were similar to those reported in Study 1. Rates of solicited and unsolicited adverse reactions reported by women 27 through 45 years of age in this study are shown in Table 3.
Table 3: Rates (%) of Solicited and Unsolicited* Injection-Site and Systemic Adverse Reactions among Boys 9 through 15 Years of Age, among Boys and Men 16 through 26 Years of Age and Women 27 through 45 Years of Age Who Received GARDASIL 9 (Studies 2, 7, and 9) GARDASIL 9 The data for GARDASIL 9 boys 9 through 15 years of age are from Study 2 (NCT00943722). The data for boys and men 16 through 26 years of age for GARDASIL 9 are from Study 7 (NCT01651949). The data for women 27 through 45 years of age are from Study 9 (NCT03158220). N=number of subjects vaccinated with safety follow-up Pain, Any=mild, moderate, severe or unknown intensity Pain, Severe=incapacitating with inability to work or do usual activity Swelling, Any=any size or size unknown Swelling, Severe=maximum size greater than 2 inches Erythema, Any=any size or size unknown Erythema, Severe=maximum size greater than 2 inches Boys and Men 16 through 26 Years of Age N=1394 Solicited Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Pain, Any 63.4 Injection-Site Pain, Severe 0.6 Injection-Site Erythema, Any 20.7 Injection-Site Erythema, Severe 0.4 Injection-Site Swelling, Any 20.2 Injection-Site Swelling, Severe 1.1 Oral Temperature ≥100.0°F† 4.4 Oral Temperature ≥102°F 0.6 Unsolicited Injection-Site Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Hypersensitivity 1.0 Injection-Site Pruritus 1.0 Unsolicited Systemic Adverse Reactions (1-15 Days Post-Vaccination, Any Dose) Headache 7.3 Pyrexia 2.4 Fatigue 1.4 Dizziness 1.1 Nausea 1.0 Boys 9 through 15 Years of Age N=639 Solicited Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Pain, Any 71.5 Injection-Site Pain, Severe 0.5 Injection-Site Erythema, Any 24.9 Injection-Site Erythema, Severe 1.9 Injection-Site Swelling, Any 26.9 Injection-Site Swelling, Severe 5.2 Oral Temperature ≥100.0°F† 10.4 Oral Temperature ≥102°F 1.4 Unsolicited Injection-Site Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Hematoma 1.3 Injection-Site Induration 1.1 Unsolicited Systemic Adverse Reactions (1-15 Days Post-Vaccination, Any Dose) Headache 9.4 Pyrexia 8.9 Nausea 1.3 Women 27 through 45 Years of Age N=640 Solicited Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Pain, Any 82.8 Injection-Site Pain, Severe 1.9 Injection-Site Erythema, Any 16.9 Injection-Site Erythema, Severe 0.5 Injection-Site Swelling, Any 23.3 Injection-Site Swelling, Severe 1.9 Oral Temperature ≥100.0°F† 2.5 Oral Temperature ≥102°F 0.3 Unsolicited Injection-Site Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Pruritus 1.6 Injection-Site Hematoma 1.3 Unsolicited Systemic Adverse Reactions (1-15 Days Post-Vaccination, Any Dose) Headache 13.6 Fatigue 3.4 Pyrexia 1.7 Nausea 1.7 Oropharyngeal pain 1.1 Serious Adverse Events in Clinical Studies
Serious adverse events were collected throughout the entire study period (range one month to 48 months post-last dose) for the seven clinical studies for GARDASIL 9. Out of the 15,705 individuals who were administered GARDASIL 9 and had safety follow-up, 354 reported a serious adverse event; representing 2.3% of the population. As a comparison, of the 7,378 individuals who were administered GARDASIL and had safety follow-up, 185 reported a serious adverse event; representing 2.5% of the population. Four GARDASIL 9 recipients each reported at least one serious adverse event that was determined to be vaccine-related. The vaccine-related serious adverse reactions were pyrexia, allergy to vaccine, asthmatic crisis, and headache.
Deaths in the Entire Study Population
Across the clinical studies, ten deaths occurred (five each in the GARDASIL 9 and GARDASIL groups); none were assessed as vaccine-related. Causes of death in the GARDASIL 9 group included one automobile accident, one suicide, one case of acute lymphocytic leukemia, one case of hypovolemic septic shock, and one unexplained sudden death 678 days following the last dose of GARDASIL 9. Causes of death in the GARDASIL control group included one automobile accident, one airplane crash, one cerebral hemorrhage, one gunshot wound, and one stomach adenocarcinoma.
Systemic Autoimmune Disorders
In all of the clinical trials with GARDASIL 9 subjects were evaluated for new medical conditions potentially indicative of a systemic autoimmune disorder. In total, 2.2% (351/15,703) of GARDASIL 9 recipients and 3.3% (240/7,378) of GARDASIL recipients reported new medical conditions potentially indicative of systemic autoimmune disorders, which were similar to rates reported following GARDASIL, AAHS control, or saline placebo in historical clinical trials.
Clinical Trials Experience for GARDASIL 9 in Individuals Who Have Been Previously Vaccinated with GARDASIL
A clinical study (Study 4) evaluated the safety of GARDASIL 9 in 12- through 26-year-old girls and women who had previously been vaccinated with three doses of GARDASIL. The time interval between the last injection of GARDASIL and the first injection of GARDASIL 9 ranged from approximately 12 to 36 months. Individuals were administered GARDASIL 9 or saline placebo and safety was evaluated using VRC-aided surveillance for 14 days after each injection of GARDASIL 9 or saline placebo in these individuals. The individuals who were monitored included 608 individuals who received GARDASIL 9 and 305 individuals who received saline placebo. Few (0.5%) individuals who received GARDASIL 9 discontinued due to adverse reactions. The vaccine-related adverse experiences that were observed among recipients of GARDASIL 9 at a frequency of at least 1.0% and also at a greater frequency than that observed among saline placebo recipients are shown in Table 4. Overall, the safety profile was similar between individuals vaccinated with GARDASIL 9 who were previously vaccinated with GARDASIL and those who were naïve to HPV vaccination with the exception of numerically higher rates of injection-site swelling and erythema among individuals who were previously vaccinated with GARDASIL (Tables 1 and 4).
Table 4: Rates (%) of Solicited and Unsolicited* Injection-Site and Systemic Adverse Reactions among Individuals Previously Vaccinated with GARDASIL Who Received GARDASIL 9 or Saline Placebo (Girls and Women 12 through 26 Years of Age) (Study 4) GARDASIL 9
N=608Saline Placebo
N=305The data for GARDASIL 9 and saline placebo are from Study 4 (NCT01047345). N=number of subjects vaccinated with safety follow-up Solicited Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Pain 90.3 38.0 Injection-Site Erythema 42.3 8.5 Injection-Site Swelling 49.0 5.9 Oral Temperature ≥100.0°F† 6.5 3.0 Unsolicited Injection-Site Adverse Reactions (1-5 Days Post-Vaccination, Any Dose) Injection-Site Pruritus 7.7 1.3 Injection-Site Hematoma 4.8 2.3 Injection-Site Reaction 1.3 0.3 Injection-Site Mass 1.2 0.7 Unsolicited Systemic Adverse Reactions (1-15 Days Post-Vaccination, Any Dose) Headache 19.6 18.0 Pyrexia 5.1 1.6 Nausea 3.9 2.0 Dizziness 3.0 1.6 Abdominal pain, upper 1.5 0.7 Influenza 1.2 1.0 Safety in Concomitant Use with Menactra and Adacel
In Study 5, the safety of GARDASIL 9 when administered concomitantly with Menactra [Meningococcal (Groups A, C, Y and W-135) Polysaccharide Diphtheria Toxoid Conjugate Vaccine] and Adacel [Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine Adsorbed (Tdap)] was evaluated in a randomized study of 1,241 boys (n = 620) and girls (n = 621) with a mean age of 12.2 years [see Clinical Studies (14.8)].
Of the 1,237 boys and girls vaccinated, 1,220 had safety follow-up for injection-site adverse reactions. The rates of injection-site adverse reactions were similar between the concomitant group and non-concomitant group (vaccination with GARDASIL 9 separated from vaccination with Menactra and Adacel by 1 month) with the exception of an increased rate of swelling reported at the injection site for GARDASIL 9 in the concomitant group (14.4%) compared to the non-concomitant group (9.4%). The majority of injection-site swelling adverse reactions were reported as being mild to moderate in intensity.
6.2 Postmarketing Experience
The postmarketing adverse experiences were reported voluntarily from a population of uncertain size, therefore, it is not possible to reliably estimate their frequency or to establish a causal relationship to vaccine exposure.
The safety profile of GARDASIL 9 and GARDASIL are similar. The postmarketing safety experience with GARDASIL is relevant to GARDASIL 9 since the vaccines are manufactured similarly and contain the same L1 HPV proteins of four of the same HPV types.
GARDASIL 9
In addition to the adverse reactions reported in the clinical studies, the following adverse experiences have been spontaneously reported during post-approval use of GARDASIL 9:
Gastrointestinal disorders: Vomiting
Skin and subcutaneous tissue disorders: Urticaria
GARDASIL
Additionally, the following postmarketing adverse experiences have been spontaneously reported for GARDASIL:
Blood and lymphatic system disorders: Autoimmune hemolytic anemia, idiopathic thrombocytopenic purpura, lymphadenopathy.
Respiratory, thoracic and mediastinal disorders: Pulmonary embolus.
Gastrointestinal disorders: Pancreatitis.
General disorders and administration site conditions: Asthenia, chills, death, malaise.
Immune system disorders: Autoimmune diseases, hypersensitivity reactions including anaphylactic/anaphylactoid reactions, bronchospasm.
Musculoskeletal and connective tissue disorders: Arthralgia, myalgia.
Nervous system disorders: Acute disseminated encephalomyelitis, Guillain-Barré syndrome, motor neuron disease, paralysis, seizures, transverse myelitis.
Infections and infestations: Cellulitis.
Vascular disorders: Deep venous thrombosis.
-
7 DRUG INTERACTIONS
7.1 Use with Systemic Immunosuppressive Medications
Immunosuppressive therapies, including irradiation, antimetabolites, alkylating agents, cytotoxic drugs, and corticosteroids (used in greater than physiologic doses), may reduce the immune responses to vaccines [see Use in Specific Populations (8.6)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively. There are no adequate and well-controlled studies of GARDASIL 9 in pregnant women. Data from GARDASIL are relevant to GARDASIL 9 because both vaccines are manufactured using the same process and have overlapping compositions. Data were evaluated from pre-licensure studies in which 55 and 62 pregnancies with known outcomes were associated with women inadvertently exposed to GARDASIL and GARDASIL 9, respectively. Pregnancy Exposure Registry data are available from 1640 and 69 prospectively enrolled women with known pregnancy outcomes who were vaccinated with GARDASIL and GARDASIL 9, respectively. Available data do not suggest an increased risk of major birth defects and miscarriage in women who received GARDASIL or GARDASIL 9.
In one developmental toxicity study, 0.5 mL of a vaccine formulation containing between 1 and 1.5 –fold of each of the 9 HPV antigen types was administered to female rats prior to mating and during gestation. In a second study, animals were administered a single human dose (0.5 mL) of GARDASIL 9 prior to mating, during gestation and during lactation. These animal studies revealed no evidence of harm to the fetus due to GARDASIL 9 [see Data].
Data
Human Data
In pre-licensure clinical studies of GARDASIL 9, women underwent pregnancy testing immediately prior to administration of each dose of GARDASIL 9 or control vaccine (GARDASIL). Subjects who were determined to be pregnant were instructed to defer vaccination until the end of their pregnancy. Despite this pregnancy screening regimen, some subjects were vaccinated very early in pregnancy before human chorionic gonadotropin (HCG) was detectable. An analysis was conducted to evaluate pregnancy outcomes for pregnancies with onset within 30 days before or after vaccination with GARDASIL 9 or GARDASIL. Among such pregnancies, there were 62 and 55 with known outcomes (excluding ectopic pregnancies and elective terminations) for GARDASIL 9 and GARDASIL, respectively, including 44 and 48 live births, respectively. The rates of pregnancies that resulted in a miscarriage were 27.4% (17/62) and 12.7% (7/55) in subjects who received GARDASIL 9 or GARDASIL, respectively. The rates of live births with major birth defects were 0% (0/44) and 2.1% (1/48) in subjects who received GARDASIL 9 or GARDASIL, respectively.
A six-year pregnancy registry for GARDASIL 9 enrolled 185 women who were exposed to GARDASIL 9 within one month prior to the last menstrual period (LMP) or at any time during pregnancy, 180 of whom were prospectively followed. After excluding elective terminations (n=1), ectopic pregnancies (n=0) and those lost to follow-up (n=110), there were 69 pregnancies (including one twin pregnancy) with known outcomes. Of the 69 pregnancies, 5 were in women exposed twice during pregnancy. Therefore, there were 74 exposures to GARDASIL 9 during pregnancy: 3 occurred during the 30 days prior to LMP, 34 during the first trimester, 13 during the second trimester, 19 during the third trimester, and 5 in an unknown trimester. Adverse pregnancy outcomes included 3 miscarriages (all in pregnancy exposures during the first trimester) and 3 major birth defects (one each for pregnancy exposure during the 30 days prior to LMP, first trimester, and second trimester). Women who were exposed twice during pregnancy had 0 miscarriages and 0 major birth defects. These data do not suggest an increased risk of major birth defects and miscarriage in women who received GARDASIL 9.
A five-year pregnancy registry enrolled 2,942 women who were exposed to GARDASIL within one month prior to the LMP or at any time during pregnancy, 2,566 of whom were prospectively followed. After excluding elective terminations (n=107), ectopic pregnancies (n=5) and those lost to follow-up (n=814), there were 1,640 pregnancies with known outcomes. Rates of miscarriage and major birth defects were 6.8% of pregnancies (111/1,640) and 2.4% of live born infants (37/1,527), respectively. These rates of assessed outcomes in the prospective population were consistent with estimated background rates.
In two postmarketing studies of GARDASIL (one conducted in the U.S., and the other in Nordic countries), pregnancy outcomes among subjects who received GARDASIL during pregnancy were evaluated retrospectively. Among the 1,740 pregnancies included in the U.S. study database, outcomes were available to assess the rates of major birth defects and miscarriage. Among the 499 pregnancies included in the Nordic study database, outcomes were available to assess the rates of major birth defects. In both studies, rates of assessed outcomes did not suggest an increased risk with the administration of GARDASIL during pregnancy.
Animal Data
Developmental toxicity studies were conducted in female rats. In one study, animals were administered 0.5 mL of a vaccine formulation containing between 1 and 1.5 –fold of each of the 9 HPV antigen types 5 and 2 weeks prior to mating, and on gestation day 6. In a second study, animals were administered a single human dose (0.5 mL) of GARDASIL 9, 5 and 2 weeks prior to mating, on gestation day 6, and on lactation day 7. No adverse effects on pre- and post-weaning development were observed. There were no vaccine-related fetal malformations or variations, or effects on female fertility.
8.2 Lactation
Risk Summary
Available data are not sufficient to assess the effects of GARDASIL 9 on the breastfed infant or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for GARDASIL 9 and any potential adverse effects on the breastfed child from GARDASIL 9 or from the underlying maternal condition. For preventive vaccines, the underlying maternal condition is susceptibility to disease prevented by the vaccine.
8.4 Pediatric Use
Safety and effectiveness have not been established in pediatric patients below 9 years of age.
8.5 Geriatric Use
The safety and effectiveness of GARDASIL 9 have not been evaluated in a geriatric population, defined as individuals aged 65 years and over.
8.6 Immunocompromised Individuals
The immunologic response to GARDASIL 9 may be diminished in immunocompromised individuals [see Drug Interactions (7.1)].
-
11 DESCRIPTION
GARDASIL 9, Human Papillomavirus 9-valent Vaccine, Recombinant, is a non-infectious recombinant 9-valent vaccine prepared from the purified virus-like particles (VLPs) of the major capsid (L1) protein of HPV Types 6, 11, 16, 18, 31, 33, 45, 52, and 58. The L1 proteins are produced by separate fermentations using recombinant Saccharomyces cerevisiae and self-assembled into VLPs. The fermentation process involves growth of S. cerevisiae on chemically-defined fermentation media which include vitamins, amino acids, mineral salts, and carbohydrates. The VLPs are released from the yeast cells by cell disruption and purified by a series of chemical and physical methods. The purified VLPs are adsorbed on preformed aluminum-containing adjuvant (Amorphous Aluminum Hydroxyphosphate Sulfate or AAHS). The 9-valent HPV VLP vaccine is a sterile liquid suspension that is prepared by combining the adsorbed VLPs of each HPV type and additional amounts of the aluminum-containing adjuvant and the final purification buffer.
GARDASIL 9 is a sterile suspension for intramuscular administration. Each 0.5-mL dose contains approximately 30 mcg of HPV Type 6 L1 protein, 40 mcg of HPV Type 11 L1 protein, 60 mcg of HPV Type 16 L1 protein, 40 mcg of HPV Type 18 L1 protein, 20 mcg of HPV Type 31 L1 protein, 20 mcg of HPV Type 33 L1 protein, 20 mcg of HPV Type 45 L1 protein, 20 mcg of HPV Type 52 L1 protein, and 20 mcg of HPV Type 58 L1 protein.
Each 0.5-mL dose of the vaccine also contains approximately 500 mcg of aluminum (provided as AAHS), 9.56 mg of sodium chloride, 0.78 mg of L-histidine, 50 mcg of polysorbate 80, 35 mcg of sodium borate, <7 mcg yeast protein, and water for injection. The product does not contain a preservative or antibiotics.
After thorough agitation, GARDASIL 9 is a white, cloudy liquid.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
HPV only infects human beings. Animal studies with analogous animal papillomaviruses suggest that the efficacy of L1 VLP vaccines may involve the development of humoral immune responses. Efficacy of GARDASIL 9 against anogenital diseases related to the vaccine HPV types in human beings is thought to be mediated by humoral immune responses induced by the vaccine, although the exact mechanism of protection is unknown.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
In these studies, seropositive is defined as anti-HPV titer greater than or equal to the pre-specified serostatus cutoff for a given HPV type. Seronegative is defined as anti-HPV titer less than the pre-specified serostatus cutoff for a given HPV type. The serostatus cutoff is the antibody titer level above the assay's lower limit of quantification that reliably distinguishes sera samples classified by clinical likelihood of HPV infection and positive or negative status by previous versions of competitive Luminex Immunoassay (cLIA). The lower limits of quantification and serostatus cutoffs for each of the 9 vaccine HPV types are shown in Table 5 below. PCR positive is defined as DNA detected for a given HPV type. PCR negative is defined as DNA not detected for a given HPV type. The lower limit of detection for the multiplexed HPV PCR assays ranged from 5 to 34 copies per test across the 9 vaccine HPV types.
Table 5: Competitive Luminex Immunoassay (cLIA) Limits of Quantification and Serostatus Cutoffs for GARDASIL 9 HPV Types HPV Type cLIA Lower Limit of Quantification (mMU*/mL) cLIA Serostatus Cutoff
(mMU*/mL)- *
- mMU=milli-Merck Units
HPV 6 16 30 HPV 11 6 16 HPV 16 12 20 HPV 18 8 24 HPV 31 4 10 HPV 33 4 8 HPV 45 3 8 HPV 52 3 8 HPV 58 4 8 14.1 Efficacy and Effectiveness Data for GARDASIL
Efficacy and effectiveness of GARDASIL are relevant to GARDASIL 9 since the vaccines are manufactured similarly and contain four of the same HPV L1 VLPs.
Individuals 16 through 26 Years of Age
Efficacy of GARDASIL was assessed in five AAHS-controlled, double-blind, randomized clinical trials evaluating 24,596 individuals 16 through 26 years of age (20,541 girls and women and 4,055 boys and men). The results of these trials are shown in Table 6 below.
Table 6: Analysis of Efficacy of GARDASIL in the PPE* Population for Vaccine HPV Types Disease Endpoints GARDASIL AAHS Control % Efficacy (95% CI) N Number of cases N Number of cases N=Number of individuals with at least one follow-up visit after Month 7 CI=Confidence Interval Note 1: Point estimates and confidence intervals are adjusted for person-time of follow-up. Note 2: Table 6 does not include cases due to HPV types not covered by the vaccine. AAHS = Amorphous Aluminum Hydroxyphosphate Sulfate, CIN = Cervical Intraepithelial Neoplasia, VIN = Vulvar Intraepithelial Neoplasia, VaIN=Vaginal Intraepithelial Neoplasia, PIN=Penile Intraepithelial Neoplasia, AIN=Anal Intraepithelial Neoplasia, AIS=Adenocarcinoma In Situ - *
- The PPE population consisted of individuals who received all three vaccinations within one year of enrollment, did not have major deviations from the study protocol, were naïve (PCR negative and seronegative) to the relevant HPV type(s) (Types 6, 11, 16, and 18) prior to dose 1 and who remained PCR negative to the relevant HPV type(s) through one month post-dose 3 (Month 7).
- †
- Analyses of the combined trials were prospectively planned and included the use of similar study entry criteria.
16- through 26-Year-Old Girls and Women† HPV 16- or 18-related CIN 2/3 or AIS 8493 2 8464 112 98.2 (93.5, 99.8) HPV 16- or 18-related VIN 2/3 7772 0 7744 10 100.0 (55.5, 100.0) HPV 16- or 18-related VaIN 2/3 7772 0 7744 9 100.0 (49.5, 100.0) HPV 6-, 11-, 16-, or 18-related CIN (CIN 1, CIN 2/3) or AIS 7864 9 7865 225 96.0 (92.3, 98.2) HPV 6-, 11-, 16-, or 18-related Genital Warts 7900 2 7902 193 99.0 (96.2, 99.9) HPV 6- and 11-related Genital Warts 6932 2 6856 189 99.0 (96.2, 99.9) 16- through 26-Year-Old Boys and Men External Genital Lesions HPV 6-, 11-, 16-, or 18-related External Genital Lesions 1394 3 1404 32 90.6 (70.1, 98.2) Condyloma 1394 3 1404 28 89.3 (65.3, 97.9) PIN 1/2/3 1394 0 1404 4 100.0 (-52.1, 100.0) HPV 6-, 11-, 16-, or 18-related Endpoint AIN 1/2/3 194 5 208 24 77.5 (39.6, 93.3) AIN 2/3 194 3 208 13 74.9 (8.8, 95.4) AIN 1
Condyloma Acuminatum
Non-acuminate194
194
1944
0
4208
208
20816
6
1173.0 (16.3, 93.4)
100.0 (8.2, 100.0)
60.4 (-33.5, 90.8)In an extension study in females 16 through 26 years of age at enrollment, prophylactic efficacy of GARDASIL through Month 60 against overall cervical and genital disease related to HPV 6, 11, 16, and 18 was 100% (95% CI: 12.3%, 100%) compared to AAHS control.
An extension study in girls and women 16 through 23 years of age used national health care registries in Denmark, Iceland, Norway, and Sweden to monitor endpoint cases of HPV 6-, 11-, 16-, or 18-related CIN (any grade), AIS, cervical cancer, vulvar cancer, or vaginal cancer among 2,650 girls and women 16 through 23 years of age at enrollment who were randomized to vaccination with GARDASIL. An interim analysis of the per-protocol effectiveness population included 1,902 subjects who completed the GARDASIL vaccination series within one year, were naïve to the relevant HPV type through 1 month post-dose 3, had no protocol violations, and had follow-up data available. The median follow-up from the first dose of vaccine was 6.7 years with a range of 2.8 to 8.4 years. At the time of interim analysis, no cases of HPV 6-, 11-, 16-, or 18-related CIN (any grade), AIS, cervical cancer, vulvar cancer, or vaginal cancer were observed over a total of 5,765 person-years at risk.
Girls and Boys 9 through 15 Years of Age
An extension study of 614 girls and 565 boys 9 through 15 years of age at enrollment who were randomized to vaccination with GARDASIL actively followed subjects for endpoint cases of HPV 6-, 11-, 16-, or 18-related persistent infection, CIN (any grade), AIS, VIN, VaIN, cervical cancer, vulvar cancer, vaginal cancer, and external genital lesions from the initiation of sexual activity or age 16 onwards. An interim analysis of the per-protocol effectiveness population included 246 girls and 168 boys who completed the GARDASIL vaccination series within one year, were seronegative to the relevant HPV type at initiation of the vaccination series, and had not initiated sexual activity prior to receiving the third dose of GARDASIL. The median follow-up from the first dose of vaccine was 7.2 years with a range of 0.5 to 8.5 years. At the time of interim analysis, no cases of persistent infection of at least 12 months' duration and no cases of HPV 6-, 11-, 16-, or 18-related CIN (any grade), AIS, VIN, VaIN, cervical cancer, vulvar cancer, vaginal cancer, or external genital lesions were observed over a total 1,105 person-years at risk. There were 4 cases of HPV 6-, 11-, 16-, or 18-related persistent infection of at least 6 months' duration, including 3 cases related to HPV 16 and 1 case related to HPV 6, none of which persisted to 12 months' duration.
Individuals 27 through 45 Years of Age
A clinical trial evaluated efficacy of GARDASIL in 3,253 women 27 through 45 years of age, based on a combined endpoint of HPV 6-, 11-, 16- or 18-related persistent infection, genital warts, vulvar and vaginal dysplastic lesions of any grade, CIN of any grade, AIS, and cervical cancer. These women were randomized 1:1 to receive either GARDASIL or AAHS control. The clinical trial was conducted in two phases: a base study and a long-term study extension. The per-protocol efficacy (PPE) population received all three vaccinations within one year of enrollment, did not have major deviations from the study protocol, were naïve (PCR negative and seronegative) to the relevant HPV type(s) (Types 6, 11, 16 and 18) prior to dose 1 and remained PCR negative to the relevant HPV type(s) through one month post-dose 3 (Month 7).
In the base study (median duration of follow-up of 3.5 years post-dose 3), the efficacy of GARDASIL against the combined incidence of HPV 6-, 11-, 16-, and 18-related persistent infection, genital warts, VIN, VaIN, vulvar cancer, vaginal cancer, cervical dysplasia (any grade CIN), AIS and cervical cancer in the PPE population was 87.7% (95% CI: 75.4%, 94.6%). The efficacy estimate for the combined endpoint was driven primarily by prevention of persistent infection. The efficacy of GARDASIL against the combined incidence of HPV 6-, 11-, 16-, and 18-related genital warts or cervical dysplasia was 95.0% (95% CI: 68.7%, 99.9%) in the PPE population. While no statistically significant efficacy was demonstrated for GARDASIL in the base study for prevention of cervical intraepithelial neoplasia grades 2 and 3 (CIN 2/3), adenocarcinoma in situ (AIS) or cervical cancer related to HPV types 16 and 18, there was 1 case of CIN 2/3 observed in the GARDASIL group and 5 cases in the placebo group. The CIN 2 case in the GARDASIL group tested positive by PCR for HPV 16 and HPV 51.
In the long-term extension of this study, subjects from Colombia (n=600) randomized to the GARDASIL group in the base study were monitored for HPV 6-, 11-, 16-, and 18-related genital warts or cervical dysplasia. The median follow-up post-dose 3 was 8.9 years with a range of 0.1 to 10.1 years over a total of 3,518 person-years. During the long-term extension phase, no cases of HPV 6-, 11-, 16-, or 18-related CIN (any grade) or genital warts were observed in the PPE population.
Effectiveness of GARDASIL in men 27 through 45 years of age is inferred from efficacy data in women 27 through 45 years of age as described above and supported by immunogenicity data from a clinical trial in which 150 men, 27 through 45 years of age, received a 3-dose regimen of GARDASIL (0, 2, 6 months). A cross-study analysis of per-protocol immunogenicity populations compared Month 7 anti-HPV 6, 11, 16, and 18 GMTs of these 27- through 45-year-old men (Study A) to those of 16- through 26-year old boys and men (Study B) in whom efficacy of GARDASIL had been established (see Table 6). GMT ratios (Study A/Study B) for HPV 6, 11, 16, and 18 were 0.82 (95%CI: 0.65, 1.03), 0.79 (95%CI: 0.66, 0.93), 0.91 (95%CI: 0.72, 1.13), and 0.74 (95%CI: 0.59, 0.92), respectively.
14.2 Clinical Trials for GARDASIL 9
Efficacy and/or immunogenicity of the 3-dose regimen of GARDASIL 9 were assessed in seven clinical trials. Study 1 evaluated the efficacy of GARDASIL 9 to prevent HPV-related cervical, vulvar, and vaginal disease using GARDASIL as a comparator.
The analysis of efficacy for GARDASIL 9 was evaluated in the per-protocol efficacy (PPE) population of 16- through 26-year-old girls and women, who received all three vaccinations within one year of enrollment, did not have major deviations from the study protocol, and were naïve to the relevant HPV type(s) by serology and PCR of cervicovaginal specimens prior to dose one and who remained PCR negative for the relevant HPV type(s) through one month post-dose 3 (Month 7). Overall, approximately 52% of subjects were negative to all vaccine HPV types by both PCR and serology at Day 1.
The primary analysis of efficacy against HPV Types 31, 33, 45, 52, and 58 is based on a combined endpoint of Cervical Intraepithelial Neoplasia (CIN) 2, CIN 3, Adenocarcinoma in situ (AIS), invasive cervical carcinoma, Vulvar Intraepithelial Neoplasia (VIN) 2/3, Vaginal Intraepithelial Neoplasia (VaIN) 2/3, vulvar cancer, or vaginal cancer. Other endpoints evaluated include cervical, vulvar and vaginal disease of any grade, persistent infection, cytological abnormalities and invasive procedures. For all endpoints, the efficacy against the HPV Types 31, 33, 45, 52 and 58 in GARDASIL 9 was evaluated compared with GARDASIL. Efficacy of GARDASIL 9 against anal lesions caused by HPV Types 31, 33, 45, 52, and 58 was not assessed due to low incidence. Effectiveness of GARDASIL 9 against anal lesions was inferred from the efficacy of GARDASIL against anal lesions caused by HPV types 6, 11, 16 and 18 in men and antibody responses elicited by GARDASIL 9 against the HPV types covered by the vaccine.
Effectiveness against disease caused by HPV Types 6, 11, 16, and 18 was assessed by comparison of geometric mean titers (GMTs) of type-specific antibodies following vaccination with GARDASIL 9 with those following vaccination with GARDASIL (Study 1 and Study 3). The effectiveness of GARDASIL 9 in girls and boys 9 through 15 years old and in boys and men 16 through 26 years old was inferred based on a comparison of type-specific antibody GMTs to those of 16 through 26-year-old girls and women following vaccination with GARDASIL 9. Immunogenicity analyses were performed in the per-protocol immunogenicity (PPI) population consisting of individuals who received all three vaccinations within pre-defined day ranges, did not have major deviations from the study protocol, met pre-defined day range for serum collection for assessment of antibody response and were naïve [PCR negative (in girls and women 16 through 26 years of age; Studies 1 and 2) and seronegative (Studies 1, 2, 3, 5, 7 and 8)] to the relevant HPV type(s) prior to dose 1 and among 16- through 26-year-old girls and women (Studies 1 and 2) remained PCR negative to the relevant HPV type(s) through Month 7. Pre-defined day ranges for vaccinations were relative to Day 1 (dose 1). For the 3-dose schedule, dose 2 was at 2 months (± 3 weeks) and dose 3 was at 6 months (± 4 weeks). For the 2-dose schedule, dose 2 was at 6 or 12 months (± 4 weeks). Pre-defined day range for serum collection for assessment of antibody response was 21 to 49 days after the last dose.
Study 1 evaluated immunogenicity of GARDASIL 9 and efficacy to prevent infection and disease caused by HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58 in 16- through 26-year-old girls and women. Study 2 evaluated immunogenicity of GARDASIL 9 in girls and boys 9 through 15 years of age and women 16 through 26 years of age. Study 3 evaluated immunogenicity of GARDASIL 9 compared with GARDASIL in girls 9 through 15 years of age. Study 4 evaluated administration of GARDASIL 9 to girls and women 12 through 26 years of age previously vaccinated with GARDASIL. Study 5 evaluated GARDASIL 9 concomitantly administered with Menactra and Adacel in girls and boys 11 through 15 years of age. Together, these five clinical trials evaluated 12,233 individuals who received GARDASIL 9 (8,048 girls and women 16 through 26 years of age at enrollment with a mean age of 21.8 years; 2,927 girls 9 through 15 years of age at enrollment with a mean age of 11.9 years; and 1,258 boys 9 through 15 years of age at enrollment with a mean age of 11.9 years. Study 7 evaluated immunogenicity of GARDASIL 9 in boys and men, including 1,106 self-identified as heterosexual men (HM) and 313 self-identified as men having sex with men (MSM), 16 through 26 years of age at enrollment (mean ages 20.8 years and 22.2 years, respectively) and 1,101 girls and women 16 through 26 years of age at enrollment (mean age 21.3 years). Study 9 evaluated immunogenicity of GARDASIL 9 in 640 women 27 through 45 years of age and 570 girls and women 16 through 26 years of age (mean ages 35.8 years and 21.6 years, respectively).
The race distribution of the 16- through 26-year-old girls and women in the clinical trials was as follows: 56.8% White; 25.2% Other; 14.1% Asian; and 3.9% Black. The race distribution of the 9- through 15-year-old girls in the clinical trials was as follows: 60.3% White; 19.3% Other; 13.5% Asian; and 7.0% Black. The race distribution of the 9- through 15-year-old boys in the clinical trials was as follows: 46.6% White; 34.3% Other; 13.3% Asian; and 5.9% Black. The race distribution of the 16- through 26-year-old boys and men in the clinical trials was as follows: 62.1% White; 22.6% Other; 9.8% Asian; and 5.5% Black.
In Study 9 the race distribution of 27- through 45-year-old women was as follows: 97.7% White, 1.6% Asian, 0.3% Other or Multiracial, and 0.5% Black. The race distribution of girls and women 16 through 26 years of age in this study was as follows: 94.6% White, 3.0% Asian, 1.6% Other or Multiracial, and 0.9% Black.
One clinical trial (Study 8) assessed the 2-dose regimen of GARDASIL 9. Study 8 evaluated the immunogenicity of 2 doses of GARDASIL 9 in girls and boys 9 through 14 years of age and 3 doses of GARDASIL 9 in girls 9 through 14 years of age and women 16 through 26 years of age; (N=1,518; 753 girls; 451 boys and 314 women). The mean age for the girls and boys 9 through 14 years of age was 11.5 years; the mean age for girls and women 16 through 26 years of age was 21.0 years. In Study 8, the race distribution was as follows: 61.1% White; 16.3% Asian; 13.3% Other; and 8.9% Black.
14.3 Efficacy – HPV Types 31, 33, 45, 52 and 58 in Girls and Women 16 through 26 Years of Age
Studies Supporting the Efficacy of GARDASIL 9 against HPV Types 31, 33, 45, 52, and 58
The efficacy of GARDASIL 9 in 16- through 26-year-old girls and women was assessed in an active comparator-controlled, double-blind, randomized clinical trial (Study 1) that included a total of 14,204 women (GARDASIL 9 = 7,099; GARDASIL = 7,105) who were enrolled and vaccinated without pre-screening for the presence of HPV infection. Subjects were followed up with a median duration of 40 months (range 0 to 64 months) after the last vaccination.
The primary efficacy evaluation was conducted in the PPE population based on a composite clinical endpoint of HPV 31-, 33-, 45-, 52-, and 58-related cervical cancer, vulvar cancer, vaginal cancer, CIN 2/3 or AIS, VIN 2/3, and VaIN 2/3. Efficacy was further evaluated with the clinical endpoints of HPV 31-, 33-, 45-, 52-, and 58-related CIN 1, vulvar and vaginal disease of any grade, and persistent infection. In addition, the study also evaluated the impact of GARDASIL 9 on the rates of HPV 31-, 33-, 45-, 52-, and 58-related abnormal Papanicolaou (Pap) tests, cervical and external genital biopsy, and definitive therapy [including loop electrosurgical excision procedure (LEEP) and conization]. Efficacy for all endpoints was measured starting after the Month 7 visit.
GARDASIL 9 prevented HPV 31-, 33-, 45-, 52-, and 58-related persistent infection and disease and also reduced the incidence of HPV 31-, 33-, 45-, 52-, and 58-related Pap test abnormalities, cervical and external genital biopsy, and definitive therapy (Table 7).
Table 7: Analysis of Efficacy of GARDASIL 9 against HPV Types 31, 33, 45, 52, and 58 in the PPE* Population of 16- through 26-Year-old Girls and Women (Study 1) Disease Endpoint GARDASIL 9
N†=7099GARDASIL
N†=7105GARDASIL 9 Efficacy
%
(95% CI)n‡ Number of cases n‡ Number of cases CI=Confidence Interval CIN=Cervical Intraepithelial Neoplasia, VIN=Vulvar Intraepithelial Neoplasia, VaIN=Vaginal Intraepithelial Neoplasia, AIS=Adenocarcinoma In Situ, ASC-US=Atypical squamous cells of undetermined significance HR=High Risk - *
- The PPE population consisted of individuals who received all three vaccinations within one year of enrollment, did not have major deviations from the study protocol, were naïve (PCR negative and seronegative) to the relevant HPV type(s) (Types 31, 33, 45, 52, and 58) prior to dose 1, and who remained PCR negative to the relevant HPV type(s) through one month post-dose 3 (Month 7); data from Study 1 (NCT00543543).
- †
- N=Number of individuals randomized to the respective vaccination group who received at least one injection
- ‡
- n=Number of individuals contributing to the analysis
- §
- Persistent infection detected in samples from two or more consecutive visits at least six months apart
- ¶
- Persistent infection detected in samples from two or more consecutive visits over 12 months or longer
- #
- Papanicolaou test
- Þ
- Including loop electrosurgical excision procedure (LEEP) and conization
HPV 31-, 33-, 45-, 52-, 58-related CIN 2/3, AIS, Cervical Cancer, VIN 2/3, VaIN 2/3, Vulvar Cancer, and Vaginal Cancer 6016 1 6017 30 96.7
(80.9, 99.8)HPV 31-, 33-, 45-, 52-, 58-related CIN 1 5948 1 5943 69 98.6
(92.4, 99.9)HPV 31-, 33-, 45-, 52-, 58-related CIN 2/3 or AIS 5948 1 5943 27 96.3
(79.5, 99.8)HPV 31-, 33-, 45-, 52-, 58-related Vulvar or Vaginal Disease 6009 1 6012 16 93.8
(61.5, 99.7)HPV 31-, 33-, 45-, 52-, 58-related Persistent Infection ≥6 Months§ 5939 26 5953 642 96.2
(94.4, 97.5)HPV 31-, 33-, 45-, 52-, 58-related Persistent Infection ≥12 Months¶ 5939 15 5953 375 96.1
(93.7, 97.9)HPV 31-, 33-, 45-, 52-, 58-related ASC-US HR-HPV Positive or Worse Pap# Abnormality 5881 35 5882 462 92.6
(89.7, 94.8)HPV 31-, 33-, 45-, 52-, 58-related Biopsy 6016 7 6017 222 96.9
(93.6, 98.6)HPV 31-, 33-, 45-, 52-, 58-related Definitive TherapyÞ 6012 4 6014 32 87.5
(65.7, 96.0)14.4 Long term Follow-up of Individuals Vaccinated with GARDASIL 9
In an extension study of individuals in Study 2, 971 girls and 301 boys 9 through 15 years of age at enrollment who received a 3-dose regimen of GARDASIL 9 were actively followed from age 16 onwards for endpoint cases of HPV 6-, 11-, 16-, 18-, 31-, 33-, 45-, 52-, and 58-related persistent infection and disease. For girls, disease endpoints assessed included HPV 6-, 11-, 16-, 18-, 31-, 33-, 45-, 52-, and 58-related CIN (any grade), AIS, VIN, VaIN, external genital warts, cervical cancer, vulvar cancer and vaginal cancer. For boys, the disease endpoints assessed included HPV 6-, 11-, 16-, 18-, 31-, 33-, 45-, 52-, and 58-related PIN, external genital warts, penile cancer, perineal cancer and perianal cancer.
Analysis of the per-protocol population included 872 girls and 262 boys who completed the GARDASIL 9 vaccination series within one year, were seronegative to the relevant HPV type at initiation of the vaccination series and had not initiated sexual activity prior to receiving the third dose of GARDASIL 9. The median follow-up from the last dose of vaccine was 10.0 years with a range of 3.0 to 11.0 years in girls 9 through 15 years of age and 9.9 years with a range of 3.0 to 10.6 years in boys 9 through 15 years of age.
In girls, no cases of HPV 6-, 11-, 16-, 18-, 31-, 33-, 45-, 52-, and 58-related CIN 2/3, AIS, VIN, VaIN, external genital warts, cervical cancer, vulvar cancer or vaginal cancer were observed over a total of 4,576.1 person-years at risk. One case of CIN1 that tested positive for HPV 16, 39 and 59 by PCR was observed. In boys, no cases of HPV 6-, 11-, 16-, 18-, 31-, 33-, 45-, 52-, and 58-related PIN, external genital warts, penile cancer, perineal cancer or perianal cancer were observed over a total of 1,278.6 person-years at risk.
Incidence rates of vaccine HPV types-related persistent infections of at least 6 months duration in girls and boys observed during the study were 52.4 (95% CI: 33.6, 78.0) and 54.6 (95% CI: 21.9, 112.4) cases per 10,000 person-years, respectively, and within the range of incidence rates reported in vaccinated cohorts of similar age based on results from previous efficacy studies of GARDASIL 9, (which were 36.6 and 21.5 per 10,000 person years for HPV 6-, 11-, 16- and 18-related and HPV 31-, 33-, 45-, 52-, and 58-related persistent infections, respectively, in females in Study 1) and GARDASIL (which were 30 and 59 per 10,000 person-years, for HPV 6-, 11-, 16- and 18-related persistent infections in GARDASIL studies in females and males, respectively).
14.5 Effectiveness in Prevention of HPV-Related Oropharyngeal and Other Head and Neck Cancers
The effectiveness of GARDASIL 9 against oropharyngeal and other head and neck cancers caused by HPV types 16, 18, 31, 33, 45, 52 and 58, is based on the effectiveness of GARDASIL and GARDASIL 9 to prevent anogenital disease caused by HPV types covered by the vaccine [see Clinical Studies (14.1, 14.2, 14.3)].
14.6 Immunogenicity of a 3-Dose Regimen
The minimum anti-HPV titer that confers protective efficacy has not been determined.
Type-specific immunoassays (i.e., cLIA) with type-specific standards were used to assess immunogenicity to each vaccine HPV type. These assays measured antibodies against neutralizing epitopes for each HPV type. The scales for these assays are unique to each HPV type; thus, comparisons across types and to other assays are not appropriate. Immunogenicity was measured by (1) the percentage of individuals who were seropositive for antibodies against the relevant vaccine HPV type, and (2) the Geometric Mean Titer (GMT).
Studies Supporting the Effectiveness of GARDASIL 9 against HPV Types 6, 11, 16, and 18
Effectiveness of GARDASIL 9 against persistent infection and disease related to HPV Types 6, 11, 16, or 18 was inferred from non-inferiority comparisons in Study 1 (16- through 26-year-old girls and women) and Study 3 (9- through 15-year-old girls) of GMTs following vaccination with GARDASIL 9 with those following vaccination with GARDASIL. A low number of efficacy endpoint cases related to HPV types 6, 11, 16 and 18 in both vaccination groups precluded a meaningful assessment of efficacy using disease endpoints associated with these HPV types. The primary analyses were conducted in the per-protocol population, which included subjects who received all three vaccinations within one year of enrollment, did not have major deviations from the study protocol, and were HPV-naïve. HPV-naïve individuals were defined as seronegative to the relevant HPV type(s) prior to dose 1 and among female subjects 16 through 26 years of age in Study 1 PCR negative to the relevant HPV type(s) in cervicovaginal specimens prior to dose 1 through Month 7.
Anti-HPV 6, 11, 16 and 18 GMTs at Month 7 for GARDASIL 9 among girls 9 through 15 years of age and young women 16 through 26 years of age were non-inferior to those among the corresponding populations for GARDASIL (Table 8). At least 99.7% of individuals included in the analyses for each HPV type became seropositive by Month 7.
Table 8: Comparison of Immune Responses (Based on cLIA) Between GARDASIL 9 and GARDASIL for HPV Types 6, 11, 16, and 18 in the PPI* Population of 9- through 26-Year-Old Girls and Women (Studies 1 and 3) Population GARDASIL 9 GARDASIL GARDASIL 9/
GARDASILN†
(n‡)GMT
mMU§/mLN†
(n‡)GMT
mMU§/mLGMT
Ratio(95% CI)¶ CI=Confidence Interval GMT=Geometric Mean Titer cLIA=competitive Luminex Immunoassay - *
- The PPI population consisted of individuals who received all three vaccinations within pre-defined day ranges, did not have major deviations from the study protocol, met predefined criteria for the interval between the Month 6 and Month 7 visit, were naïve (PCR negative [among 16- through 26-year old girls and women] and seronegative) to the relevant HPV type(s) (types 6, 11, 16, and 18) prior to dose 1, and among 16- through 26-year-old girls and women remained PCR negative to the relevant HPV type(s) through one month post-dose 3 (Month 7). The data for 16- through 26-year-old girls and women are from Study 1 (NCT00543543), and the data for 9- through 15-year-old girls are from Study 3 (NCT01304498).
- †
- N=Number of individuals randomized to the respective vaccination group who received at least one injection
- ‡
- n=Number of individuals contributing to the analysis
- §
- mMU=milli-Merck Units
- ¶
- Demonstration of non-inferiority required that the lower bound of the 95% CI of the GMT ratio be greater than 0.67
Anti-HPV 6 9- through 15-year-old girls 300
(273)1679.4 300
(261)1565.9 1.07 (0.93, 1.23) 16- through 26-year-old girls and women 6792
(3993)893.1 6795
(3975)875.2 1.02 (0.99, 1.06) Anti-HPV 11 9- through 15-year-old girls 300
(273)1315.6 300
(261)1417.3 0.93 (0.80, 1.08) 16- through 26-year-old girls and women 6792
(3995)666.3 6795
(3982)830.0 0.80 (0.77, 0.83) Anti-HPV 16 9- through 15-year-old girls 300
(276)6739.5 300
(270)6887.4 0.97 (0.85, 1.11) 16- through 26-year-old girls and women 6792
(4032)3131.1 6795
(4062)3156.6 0.99 (0.96, 1.03) Anti-HPV 18 9- through 15-year-old girls 300
(276)1956.6 300
(269)1795.6 1.08 (0.91, 1.29) 16- through 26-year-old girls and women 6792
(4539)804.6 6795
(4541)678.7 1.19 (1.14, 1.23) Study Supporting the Effectiveness of GARDASIL 9 against Vaccine HPV Types in 9- through 15-Year-Old Girls and Boys
Effectiveness of GARDASIL 9 against persistent infection and disease related to vaccine HPV types in 9- through 15-year-old girls and boys was inferred from non-inferiority comparison conducted in the PPI population in Study 2 of GMTs following vaccination with GARDASIL 9 among 9- through 15-year-old girls and boys with those among 16- through 26-year-old girls and women. Anti-HPV GMTs at Month 7 among 9- through 15-year-old girls and boys were non-inferior to anti-HPV GMTs among 16- through 26-year-old girls and women (Table 9).
Table 9: Comparison of Immune Responses (Based on cLIA) between the PPI* Populations of 16- through 26-Year-Old Girls and Women, 9- through 15-Year-Old Girls, and 9- through 15-Year-Old Boys for All GARDASIL 9 Vaccine HPV Types (Study 2) Population N† n‡ GMT
mMU§/mLGMT Ratio relative to 16-through 26-year-old girls and women
(95% CI)¶cLIA=competitive Luminex Immunoassay CI=Confidence Interval GMT=Geometric Mean Titer - *
- The PPI population consisted of individuals who received all three vaccinations within pre-defined day ranges, did not have major deviations from the study protocol, met predefined criteria for the interval between the Month 6 and Month 7 visit, were naïve (PCR negative [among 16- through 26-year old girls and women] and seronegative) to the relevant HPV type(s) prior to dose 1 and among 16- through 26-year-old girls and women remained PCR negative to the relevant HPV types through one month post-dose 3 (Month 7). The data are from Study 2 (NCT00943722).
- †
- N=Number of individuals randomized to the respective vaccination group who received at least one injection
- ‡
- n=Number of individuals contributing to the analysis
- §
- mMU=milli-Merck Units
- ¶
- Demonstration of non-inferiority required that the lower bound of the 95% CI of the GMT ratio be greater than 0.67
Anti-HPV 6 9- through 15-year-old girls 630 503 1703.1 1.89 (1.68, 2.12) 9- through 15-year-old boys 641 537 2083.4 2.31 (2.06, 2.60) 16- through 26-year-old girls and women 463 328 900.8 1 Anti-HPV 11 9- through 15-year-old girls 630 503 1291.5 1.83 (1.63, 2.05) 9- through 15-year-old boys 641 537 1486.3 2.10 (1.88, 2.36) 16- through 26-year-old girls and women 463 332 706.6 1 Anti-HPV 16 9- through 15-year-old girls 630 513 6933.9 1.97 (1.75, 2.21) 9- through 15-year-old boys 641 546 8683.0 2.46 (2.20, 2.76) 16- through 26-year-old girls and women 463 329 3522.6 1 Anti-HPV 18 9- through 15-year-old girls 630 516 2148.3 2.43 (2.12, 2.79) 9- through 15-year-old boys 641 544 2855.4 3.23 (2.83, 3.70) 16- through 26-year-old girls and women 463 345 882.7 1 Anti-HPV 31 9- through 15-year-old girls 630 506 1894.7 2.51 (2.21, 2.86) 9- through 15-year-old boys 641 543 2255.3 2.99 (2.63, 3.40) 16- through 26-year-old girls and women 463 340 753.9 1 Anti-HPV 33 9- through 15-year-old girls 630 518 985.8 2.11 (1.88, 2.37) 9- through 15-year-old boys 641 544 1207.4 2.59 (2.31, 2.90) 16- through 26-year-old girls and women 463 354 466.8 1 Anti-HPV 45 9- through 15-year-old girls 630 518 707.7 2.60 (2.25, 3.00) 9- through 15-year-old boys 641 547 912.1 3.35 (2.90, 3.87) 16- through 26-year-old girls and women 463 368 272.2 1 Anti-HPV 52 9- through 15-year-old girls 630 517 962.2 2.21 (1.96, 2.49) 9- through 15-year-old boys 641 545 1055.5 2.52 (2.22, 2.84) 16- through 26-year-old girls and women 463 337 419.6 1 Anti-HPV 58 9- through 15-year-old girls 630 516 1288.0 2.18 (1.94, 2.46) 9- through 15-year-old boys 641 544 1593.3 2.70 (2.40, 3.03) 16- through 26-year-old girls and women 463 332 590.5 1 Study Supporting the Effectiveness of GARDASIL 9 against Vaccine HPV Types in 16- through 26-Year-Old Boys and Men
Effectiveness of GARDASIL 9 against persistent infection and disease related to vaccine HPV types in 16- through 26-year-old boys and men was inferred from non-inferiority comparison conducted in the PPI population in Study 7 of GMTs following vaccination with GARDASIL 9 among 16- through 26-year-old HM with those among 16- through 26-year-old girls and women. Anti-HPV GMTs at Month 7 among 16- through 26-year-old HM were non-inferior to anti-HPV GMTs among 16- through 26-year-old girls and women (Table 10). Study 7 also enrolled 313 16- through 26-year-old HIV-negative MSM. At Month 7, anti-HPV GMT ratios for MSM relative to HM ranged from 0.6 to 0.8, depending on HPV type. The GMT ratios for MSM relative to HM were generally similar to those previously observed in clinical trials with GARDASIL.
Table 10: Comparison of Immune Responses (Based on cLIA) between the PPI* Populations of 16- through 26-Year-Old Girls and Women and 16- through 26-Year-Old Boys and Men Self-Identified as Heterosexual (HM) for All GARDASIL 9 Vaccine HPV Types (Study 7) Population N† n‡ GMT
mMU§/mLGMT Ratio relative to 16- through 26-year-old girls and women
(95% CI)¶cLIA=competitive Luminex Immunoassay CI=Confidence Interval GMT=Geometric Mean Titer - *
- The PPI population consisted of individuals who received all three vaccinations within pre-defined day ranges, did not have major deviations from the study protocol, met predefined criteria for the interval between the Month 6 and Month 7 visit, and were seronegative to the relevant HPV type(s) (types 6, 11, 16, 18, 31, 33, 45, 52, and 58) prior to dose 1. The data are from Study 7 (NCT01651949).
- †
- Number of individuals randomized to the respective vaccination group who received at least one injection
- ‡
- Number of individuals contributing to the analysis
- §
- mMU=milli-Merck Units
- ¶
- Demonstration of non-inferiority required that the lower bound of the 95% CI of the GMT ratio be greater than 0.67
Anti-HPV 6 16- through 26-year-old HM 1103 847 782.0 1.11 (1.02, 1.21) 16- through 26-year-old girls and women 1099 708 703.9 1 Anti-HPV 11 16- through 26-year-old HM 1103 851 616.7 1.09 (1.00, 1.19) 16- through 26-year-old girls and women 1099 712 564.9 1 Anti-HPV 16 16- through 26-year-old HM 1103 899 3346.0 1.20 (1.10, 1.30) 16- through 26-year-old girls and women 1099 781 2788.3 1 Anti-HPV 18 16- through 26-year-old HM 1103 906 808.2 1.19 (1.08, 1.31) 16- through 26-year-old girls and women 1099 831 679.8 1 Anti-HPV 31 16- through 26-year-old HM 1103 908 708.5 1.24 (1.13, 1.37) 16- through 26-year-old girls and women 1099 826 570.1 1 Anti-HPV 33 16- through 26-year-old HM 1103 901 384.8 1.19 (1.10, 1.30) 16- through 26-year-old girls and women 1099 853 322.0 1 Anti-HPV 45 16- through 26-year-old HM 1103 909 235.6 1.27 (1.14, 1.41) 16- through 26-year-old girls and women 1099 871 185.7 1 Anti-HPV 52 16- through 26-year-old HM 1103 907 386.8 1.15 (1.05, 1.26) 16- through 26-year-old girls and women 1099 849 335.2 1 Anti-HPV 58 16- through 26-year-old HM 1103 897 509.8 1.25 (1.14, 1.36) 16- through 26-year-old girls and women 1099 839 409.3 1 Study Supporting the Effectiveness of GARDASIL 9 against Vaccine HPV Types in 27- through 45-Year-Old Women
Effectiveness of GARDASIL 9 against persistent infection and disease related to vaccine HPV types in 27- through 45-year-old women was supported by immunobridging comparisons conducted in the PPI population in Study 9. In Study 9, the GMT ratios of anti-HPV responses at Month 7 among 27- through 45-year-old women relative to anti-HPV responses among 16- through 26-year-old girls and women met the success criteria of having the lower bound of the 95% CI of the GMT ratios greater than 0.50 for HPV 16, 18, 31, 33, 45, 52, and 58 (Table 11).
Table 11: Comparison of Immune Responses (Based on cLIA) Between the PPI* Populations of 27- through 45 Year-Old Women and 16- through 26-Year-Old Girls and Women for GARDASIL 9 Vaccine HPV Types (Study 9) Population N† n‡ GMT
mMU§/mLGMT Ratio relative to 16-through 26-year-old girls and women (95% CI)¶ cLIA=Competitive Luminex Immunoassay CI=Confidence Interval GMT=Geometric Mean Titers - *
- The PPI population consisted of individuals who received all 3 vaccinations within pre-defined day ranges, did not have major deviations from the study protocol, met predefined criteria for the interval between the Month 6 and Month 7 visit, and were seronegative to the relevant HPV type(s) (types 16, 18, 31, 33, 45, 52, and 58) prior to dose 1. The data are from Study 9 (NCT03158220).
- †
- Number of individuals randomized to the respective vaccination group who received at least 1 injection
- ‡
- Number of individuals contributing to the analysis
- §
- mMU=milli-Merck Units
- ¶
- Immunobridging required that the lower bound of the 95% CI of the GMT ratio be greater than 0.50
- #
- N.D=Not Determined. GMT ratios were not calculated because immunobridging comparison was not specified in the study protocol for HPV types 6 and 11.
Anti-HPV 6 27- through 45-year-old women 640 448 638.4 N.D# 16- through 26-year-old girls and women 570 421 787.8 N.D# Anti-HPV 11 27- through 45-year-old women 640 448 453.5 N.D# 16- through 26-year-old girls and women 570 421 598.7 N.D# Anti-HPV 16 27- through 45-year-old women 640 448 2,147.5 0.70 (0.63, 0.77) 16- through 26-year-old girls and women 570 436 3,075.8 1 Anti-HPV 18 27- through 45-year-old women 640 471 532.1 0.71 (0.64, 0.80) 16- through 26-year-old girls and women 570 421 744.5 1 Anti-HPV 31 27- through 45-year-old women 640 488 395.7 0.66 (0.60, 0.74) 16- through 26-year-old girls and women 570 447 596.1 1 Anti-HPV 33 27- through 45-year-old women 640 493 259.0 0.73 (0.67, 0.80) 16- through 26-year-old girls and women 570 457 354.5 1 Anti-HPV 45 27- through 45-year-old women 640 515 145.6 0.68 (0.60, 0.76) 16- through 26-year-old girls and women 570 470 214.9 1 Anti-HPV 52 27- through 45-year-old women 640 496 244.7 0.71 (0.64, 0.78) 16- through 26-year-old girls and women 570 456 346.5 1 Anti-HPV 58 27- through 45-year-old women 640 478 296.4 0.69 (0.63, 0.76) 16- through 26-year-old girls and women 570 451 428.0 1 Immune Response to GARDASIL 9 across All Clinical Trials
Across all clinical trials, at least 99.2% of individuals included in the analyses for each of the nine vaccine HPV types became seropositive by Month 7. Anti-HPV GMTs at Month 7 among 9- through 15-year-old girls and boys and 16- through 26-year-old boys and men were comparable to anti-HPV responses among 16- through 26-year-old girls and women in the combined database of immunogenicity studies for GARDASIL 9.
Persistence of Immune Response to GARDASIL 9
In an extension study of individuals in Study 2, among girls and boys 9 through 15 years of age at enrollment (range of 494 to 525 subjects with evaluable data across HPV types) and followed for 10 years post dose 3, anti-HPV 6, 11, 16, 18, 31, 33, 45, 52 and 58 GMTs as measured by cLIA were decreased compared with corresponding values at one-month post-dose 3. The proportion of seropositive subjects ranged from 99.6% to 100% at one month post-dose 3 and from 81.3% to 97.7% at 10 years post-dose 3, depending on HPV type.
Administration of GARDASIL 9 to Individuals Previously Vaccinated with GARDASIL
Study 4 evaluated the immunogenicity of 3 doses of GARDASIL 9 in 921 girls and women (12 through 26 years of age) who had previously been vaccinated with 3 doses of GARDASIL. Prior to enrollment in the study, over 99% of subjects had received three injections of GARDASIL within a one year period. The time interval between the last injection of GARDASIL and the first injection of GARDASIL 9 ranged from approximately 12 to 36 months.
Seropositivity to HPV Types 6, 11, 16, 18, 31, 33, 45, 52, and 58 in the per protocol population ranged from 98.3 to 100% by Month 7 in individuals who received GARDASIL 9. The anti-HPV 31, 33, 45, 52 and 58 GMTs for the population previously vaccinated with GARDASIL were 25-63% of the GMTs in the combined populations from Studies 1, 2, 3, and 5, who had not previously received GARDASIL, although the clinical relevance of these differences is unknown. Efficacy of GARDASIL 9 in preventing infection and disease related to HPV Types 31, 33, 45, 52, and 58 in individuals previously vaccinated with GARDASIL has not been assessed.
Concomitant Use of Hormonal Contraceptives
Among 7,269 female recipients of GARDASIL 9 (16 through 26 years of age), 60.2% used hormonal contraceptives during the vaccination period of clinical studies 1 and 2. Use of hormonal contraceptives did not appear to affect the type of specific immune responses to GARDASIL 9.
14.7 Immune Responses to GARDASIL 9 Using a 2-Dose Regimen in Individuals 9 through 14 Years of Age
Effectiveness of GARDASIL 9 against persistent infection and disease related to vaccine HPV types in 9- through 14-year-old girls and boys who received a 2-dose regimen was inferred from non-inferiority comparison conducted in the PPI population in Study 8 of GMTs following vaccination with GARDASIL 9 among 9- through 14-year-old girls and boys who received a 2-dose regimen (at 0, 6 months or 0, 12 months) with those among 16- through 26-year-old girls and women who received a 3-dose regimen (at 0, 2, 6 months). Anti-HPV GMTs at one month after the last dose among 9- through 14-year-old girls and boys who received 2 doses of GARDASIL 9 were non-inferior to anti-HPV GMTs among 16- through 26-year-old girls and women who received 3 doses of GARDASIL 9 (Table 12).
One month following the last dose of the assigned regimen, between 97.9% and 100% of subjects across all groups became seropositive for antibodies against the 9 vaccine HPV types (Table 12).
In the same study, in girls and boys 9 through 14 years old, GMTs at one month after the last vaccine dose were numerically lower for some vaccine types after a 2-dose schedule than in girls 9 through 14 years old after a 3-dose schedule (HPV types 18, 31, 45, and 52 after 0, 6 months and HPV type 45 after 0, 12 months; Table 12). The clinical relevance of these findings is unknown.
Duration of immunity of a 2-dose schedule of GARDASIL 9 has not been established.
Table 12: Summary of Anti-HPV cLIA Geometric Mean Titers in the PPI* Population at One Month After the Last Vaccine Dose Among Subjects Who Received 2 Doses† or 3 Doses† of GARDASIL 9 (Study 8) Population (Regimen) N n GMT
mMU‡/mLGMT Ratio relative to 3-dose regimen in 16- through 26-year-old girls and women
(95% CI)N = Number of individuals randomized to the respective vaccination group who received at least 1 injection n = Number of individuals contributing to the analysis CI=Confidence Interval cLIA=competitive Luminex Immunoassay GMT=Geometric Mean Titer - *
- The PPI population consisted of individuals who received all assigned vaccinations within pre-defined day ranges, did not have major deviations from the study protocol, met predefined criteria for the interval between the last vaccination dose and blood collection for immunogenicity assessment, and were seronegative to the relevant HPV type(s) (types 6, 11, 16, 18, 31, 33, 45, 52, and 58) prior to dose 1.
- †
- 2-dose regimen (0, 6): vaccination at Day 1 and Month 6; 2-dose regimen (0, 12): vaccination at Day 1 and Month 12; 3-dose regimen (0, 2, 6): vaccination at Day 1, Month 2, and Month 6. The data are from Study 8 (NCT01984697).
- ‡
- mMU=milli-Merck Units
- §
- Demonstration of non-inferiority required that the lower bound of the 95% CI of the GMT ratio be greater than 0.67
- ¶
- Exploratory analysis; criterion for non-inferiority was not pre-specified
Anti-HPV 6 9- to 14-year-old girls (0, 6)† 301 258 1657.9 2.15 (1.83, 2.53)§ 9- to 14-year-old boys (0, 6)† 301 263 1557.4 2.02 (1.73, 2.36)§ 9- to 14-year-old girls and boys (0, 12)† 300 257 2678.8 3.47 (2.93, 4.11)§ 9- to 14-year-old girls (0, 2, 6)† 300 254 1496.1 1.94 (1.65, 2.29)¶ 16- to 26-year-old women (0, 2, 6)† 314 238 770.9 1 Anti-HPV 11 9- to 14-year-old girls (0, 6)† 301 258 1388.9 2.39 (2.03, 2.82)§ 9- to 14-year-old boys (0, 6)† 301 264 1423.9 2.45 (2.09, 2.88)§ 9- to 14-year-old girls and boys (0, 12)† 300 257 2941.8 5.07 (4.32, 5.94)§ 9- to 14-year-old girls (0, 2, 6)† 300 254 1306.3 2.25 (1.90, 2.66)¶ 16- to 26-year-old women (0, 2, 6)† 314 238 580.5 1 Anti-HPV 16 9- to 14-year-old girls (0, 6)† 301 272 8004.9 2.54 (2.14, 3.00)§ 9- to 14-year-old boys (0, 6)† 301 273 8474.8 2.69 (2.29, 3.15)§ 9- to 14-year-old girls and boys (0, 12)† 300 264 14329.3 4.54 (3.84, 5.37)§ 9- to 14-year-old girls (0, 2, 6)† 300 269 6996.0 2.22 (1.89, 2.61)¶ 16- to 26-year-old women (0, 2, 6)† 314 249 3154.0 1 Anti-HPV 18 9- to 14-year-old girls (0, 6)† 301 272 1872.8 2.46 (2.05, 2.96)§ 9- to 14-year-old boys (0, 6)† 301 272 1860.9 2.44 (2.04, 2.92)§ 9- to 14-year-old girls and boys (0, 12)† 300 266 2810.4 3.69 (3.06, 4.45)§ 9- to 14-year-old girls (0, 2, 6)† 300 270 2049.3 2.69 (2.24, 3.24)¶ 16- to 26-year-old women (0, 2, 6)† 314 267 761.5 1 Anti-HPV 31 9- to 14-year-old girls (0, 6)† 301 272 1436.3 2.51 (2.10, 3.00)§ 9- to 14-year-old boys (0, 6)† 301 271 1498.2 2.62 (2.20, 3.12)§ 9- to 14-year-old girls and boys (0, 12)† 300 268 2117.5 3.70 (3.08, 4.45)§ 9- to 14-year-old girls (0, 2, 6)† 300 271 1748.3 3.06 (2.54, 3.67)¶ 16- to 26-year-old women (0, 2, 6)† 314 264 572.1 1 Anti-HPV 33 9- to 14-year-old girls (0, 6)† 301 273 1030.0 2.96 (2.50, 3.50)§ 9- to 14-year-old boys (0, 6)† 301 271 1040.0 2.99 (2.55, 3.50)§ 9- to 14-year-old girls and boys (0, 12)† 300 269 2197.5 6.31 (5.36, 7.43)§ 9- to 14-year-old girls (0, 2, 6)† 300 275 796.4 2.29 (1.95, 2.68) ¶ 16- to 26-year-old women (0, 2, 6)† 314 279 348.1 1 Anti-HPV 45 9- to 14-year-old girls (0, 6)† 301 274 357.6 1.67 (1.38, 2.03)§ 9- to 14-year-old boys (0, 6)† 301 273 352.3 1.65 (1.37, 1.99)§ 9- to 14-year-old girls and boys (0, 12)† 300 268 417.7 1.96 (1.61, 2.37)§ 9- to 14-year-old girls (0, 2, 6)† 300 275 661.7 3.10 (2.54, 3.77)¶ 16- to 26-year-old women (0, 2, 6)† 314 280 213.6 1 Anti-HPV 52 9- to 14-year-old girls (0, 6)† 301 272 581.1 1.60 (1.36, 1.87)§ 9- to 14-year-old boys (0, 6)† 301 273 640.4 1.76 (1.51, 2.05)§ 9- to 14-year-old girls and boys (0, 12)† 300 268 1123.4 3.08 (2.64, 3.61)§ 9- to 14-year-old girls (0, 2, 6)† 300 275 909.9 2.50 (2.12, 2.95)¶ 16- to 26-year-old women (0, 2, 6)† 314 271 364.2 1 Anti-HPV 58 9- to 14-year-old girls (0, 6)† 301 270 1251.2 2.55 (2.15, 3.01)§ 9- to 14-year-old boys (0, 6)† 301 270 1325.7 2.70 (2.30, 3.16)§ 9- to 14-year-old girls and boys (0, 12)† 300 265 2444.6 4.98 (4.23, 5.86)§ 9- to 14-year-old girls (0, 2, 6)† 300 273 1229.3 2.50 (2.11, 2.97)¶ 16- to 26-year-old women (0, 2, 6)† 314 261 491.1 1 14.8 Studies with Menactra and Adacel
In Study 5, the safety and immunogenicity of co-administration of GARDASIL 9 with Menactra [Meningococcal (Groups A, C, Y and W-135) Polysaccharide Diphtheria Toxoid Conjugate Vaccine] and Adacel [Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine Adsorbed (Tdap)] (same visit, injections at separate sites) were evaluated in 1,237 boys and girls 11 through 15 years of age at enrollment.
One group received GARDASIL 9 in one limb and both Menactra and Adacel, as separate injections, in the opposite limb concomitantly on Day 1 (n = 619). The second group received the first dose of GARDASIL 9 on Day 1 in one limb then Menactra and Adacel, as separate injections, at Month 1 in the opposite limb (n = 618). Subjects in both vaccination groups received the second dose of GARDASIL 9 at Month 2 and the third dose at Month 6. Immunogenicity was assessed for all vaccines one month post vaccination (one dose for Menactra and Adacel and three doses for GARDASIL 9).
Assessments of post-vaccination immune responses included type-specific antibody GMTs for each of the vaccine HPV types at four weeks following the last dose of GARDASIL 9; GMTs for anti-filamentous hemagglutinin, anti-pertactin, and anti-fimbrial antibodies at four weeks following Adacel; percentage of subjects with anti-tetanus toxin and anti-diphtheria toxin antibody concentrations ≥0.1 IU/mL at four weeks following Adacel; and percentage of subjects with ≥4-fold rise from pre-vaccination baseline in antibody titers against N. meningitidis serogroups A, C, Y, and W-135 at four weeks following Menactra. Based on these measures, concomitant administration of GARDASIL 9 with Menactra and Adacel did not interfere with the antibody responses to any of the vaccines when compared with non-concomitant administration of GARDASIL 9 with Menactra and Adacel.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
GARDASIL 9 is supplied in vials and syringes.
Carton of ten 0.5-mL single-dose vials. NDC 0006-4119-03
Carton of ten 0.5-mL single-dose prefilled Luer Lock syringes with tip caps. NDC 0006-4121-02
Store refrigerated at 2 to 8°C (36 to 46°F). Do not freeze. Protect from light.
GARDASIL 9 should be administered as soon as possible after being removed from refrigeration. GARDASIL 9 can be administered provided total (cumulative multiple excursion) time out of refrigeration (at temperatures between 8°C and 25°C) does not exceed 72 hours. Cumulative multiple excursions between 0°C and 2°C are also permitted as long as the total time between 0°C and 2°C does not exceed 72 hours. These are not, however, recommendations for storage.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Inform the patient, parent, or guardian:
- Vaccination does not eliminate the necessity for women to continue to undergo recommended cervical cancer screening. Women who receive GARDASIL 9 should continue to undergo cervical cancer screening per standard of care.
- Recipients of GARDASIL 9 should not discontinue anal cancer screening if it has been recommended by a health care provider.
- GARDASIL 9 has not been demonstrated to provide protection against disease from vaccine and non-vaccine HPV types to which a person has previously been exposed through sexual activity.
- Since syncope has been reported following HPV vaccination sometimes resulting in falling with injury, observation for 15 minutes after administration is recommended.
- Vaccine information is required to be given with each vaccination to the patient, parent, or guardian.
- Provide information regarding benefits and risks associated with vaccination.
- Safety and effectiveness of GARDASIL 9 have not been established in pregnant women.
- It is important to complete the full vaccination series unless contraindicated.
- Report any adverse reactions to their health care provider.
-
SPL UNCLASSIFIED SECTION
Manuf. and Dist. by: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USAFor patent information: www.msd.com/research/patent
The trademarks depicted herein are owned by their respective companies.
Copyright © 2006-2024 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.uspi-v503-i-2403r016
-
Patient Information about GARDASIL®9 (pronounced "gard-Ah-sill nīn") (Human Papillomavirus 9-valent Vaccine, Recombinant)
Read this information with care before getting GARDASIL®9. You or your child (the person getting GARDASIL 9) will need 2 or 3 doses of the vaccine, depending on how old you are. It is important to read this information before getting each dose. This information does not take the place of talking with your health care professional about GARDASIL 9.
What is GARDASIL 9?
GARDASIL 9 is a vaccine (injection/shot) given to individuals 9 through 45 years of age to help protect against diseases caused by some types of Human Papillomavirus (HPV).
What diseases can GARDASIL 9 help protect against?
In girls and women 9 through 45 years of age, GARDASIL 9 helps protect against:
- Cervical cancer
- Vulvar and vaginal cancers
- Anal cancer
- Certain head and neck cancers, such as throat and back of mouth cancers
- Precancerous cervical, vulvar, vaginal and anal lesions
- Genital warts
In boys and men 9 through 45 years of age, GARDASIL 9 helps protect against:
- Anal cancer
- Certain head and neck cancers, such as throat and back of mouth cancers
- Precancerous anal lesions
- Genital warts
These diseases may have many causes, including HPV Types 6, 11, 16, 18, 31, 33, 45, 52, and 58. GARDASIL 9 only protects against diseases caused by these nine types of HPV.
People cannot get HPV or any of these diseases from GARDASIL 9.
What important information about GARDASIL 9 should I know?
GARDASIL 9:
- Does not remove the need for screening for cervical, vulvar, vaginal, anal, and certain head and neck cancers, such as throat and back of mouth cancers as recommended by a health care professional; women should still get routine cervical cancer screening.
- Does not protect the person getting GARDASIL 9 from a disease that is caused by other types of HPV, other viruses or bacteria.
- Does not treat HPV infection.
- Does not protect the person getting GARDASIL 9 from HPV types that he/she may already have.
GARDASIL 9 may not fully protect each person who gets it.
Who should not get GARDASIL 9?
Anyone with an allergic reaction to:
- A previous dose of GARDASIL 9
- A previous dose of GARDASIL®
- Yeast (severe allergic reaction)
- Amorphous aluminum hydroxyphosphate sulfate
- Polysorbate 80
What should I tell the health care professional before getting GARDASIL 9?
Tell the health care professional if you or your child (the person getting GARDASIL 9):
- Are pregnant or planning to get pregnant.
- Have immune problems, like HIV or cancer.
- Take medicines that affect the immune system.
- Have a fever over 100°F (37.8°C).
- Might have had an allergic reaction to a previous dose of GARDASIL 9 or GARDASIL.
- Take any medicines, even those you can buy over the counter.
The health care professional will help decide if you or your child should get the vaccine.
How is GARDASIL 9 given?
GARDASIL 9 is a shot that is usually given in the arm muscle. GARDASIL 9 may be given as 2 or 3 shots.
For persons who are You will need Given as 9 through 14 years old 2-shots* Dose 1: first shot
Dose 2: second shot given between 6 and 12 months after the first shotor 3-shots† Dose 1: first shot
Dose 2: second shot given 2 months after the first shot
Dose 3: third shot given 6 months after the first shot15 through 45 years old 3-shots Dose 1: first shot
Dose 2: second shot given 2 months after the first shot
Dose 3: third shot given 6 months after the first shotMake sure that you or your child gets all doses recommended by your health care professional so that you or your child gets the best protection. If the person getting GARDASIL 9 misses a dose, tell the health care professional and they will decide when to give the missed dose. It is important that you follow the instructions of your health care professional regarding return visits for the follow-up doses.
Fainting can happen after getting an HPV vaccine. Sometimes people who faint can fall and hurt themselves. For this reason, the health care professional may ask the person getting GARDASIL 9 to sit or lie down for 15 minutes after getting the vaccine. Some people who faint might shake or become stiff. The health care professional may need to treat the person getting GARDASIL 9.
Can I get GARDASIL 9 if I have already gotten GARDASIL?
If you have already gotten GARDASIL, talk to your health care professional to see if GARDASIL 9 is right for you.
Can I get GARDASIL 9 with other vaccines?
GARDASIL 9 can be given at the same time as:
- Menactra [Meningococcal (Groups A, C, Y and W-135) Polysaccharide Diphtheria Toxoid Conjugate Vaccine]
- Adacel [Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine Adsorbed (Tdap)]
What are the possible side effects of GARDASIL 9?
The most common side effects seen with GARDASIL 9 are:
- pain, swelling, redness, itching, bruising, bleeding, and a lump where you got the shot
- headache
- fever
- nausea
- dizziness
- tiredness
- diarrhea
- abdominal pain
- sore throat
Studies show that there was more swelling where the shot was given when GARDASIL 9 was given at the same time as Menactra and/or Adacel.
Tell the health care professional if you have any of these problems because these may be signs of an allergic reaction:
- difficulty breathing
- wheezing (bronchospasm)
- hives
- rash
Additional side effects that have been reported during general use for GARDASIL 9 are shown below. Side effects reported during the general use of GARDASIL are also shown below. GARDASIL side effects are reported as they may be relevant to GARDASIL 9 since the vaccines are similar in composition.
GARDASIL 9
- vomiting
- hives
Additionally, these side effects have been seen with the general use of GARDASIL.
GARDASIL
- swollen glands (neck, armpit, or groin)
- joint pain
- unusual tiredness, weakness, or confusion
- chills
- generally feeling unwell
- leg pain
- shortness of breath
- chest pain
- aching muscles
- muscle weakness
- seizure
- bad stomachache
- bleeding or bruising more easily than normal
- skin infection
You should contact your health care professional right away if you get any symptoms that bother you.
For a more complete list of side effects, ask the health care professional.
Call your health care professional for medical advice about side effects. You may also report any side effects to your doctor or directly to Vaccine Adverse Event Reporting System (VAERS). The VAERS toll-free number is 1-800-822-7967 or report online to www.vaers.hhs.gov .
GARDASIL 9 was not studied in women who knew they were pregnant. Tell the doctor or health care professional if you or your child (the person getting GARDASIL 9), is pregnant or is planning to get pregnant.
What is in GARDASIL 9?
GARDASIL 9 contains:
- Proteins of HPV Types 6, 11, 16, 18, 31, 33, 45, 52, and 58
- Amorphous aluminum hydroxyphosphate sulfate
- Yeast protein
- Sodium chloride
- L-histidine
- Polysorbate 80
- Sodium borate
- Water
This document is a summary of information about GARDASIL 9.
To learn more about GARDASIL 9, please talk to the health care professional or visit www.GARDASIL9.com .
-
SPL UNCLASSIFIED SECTION
Manuf. and Dist. by: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USAFor patent information: www.msd.com/research/patent
The trademarks depicted herein are owned by their respective companies.
Copyright © 2006-2024 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.Revised: 03/2024
usppi-v503-i-2403r007
-
PRINCIPAL DISPLAY PANEL - 0.5 mL Vial Carton
NDC 0006-4119-03
10 Single-dose 0.5-mL Vials
REFRIGERATE
Human Papillomavirus 9-valent Vaccine, Recombinant
GARDASIL® 9
Suspension for intramuscular injection
Each 0.5-mL dose contains human papillomavirus L1 protein of each type adsorbed on amorphous aluminum
hydroxyphosphate sulfate adjuvant as follows:
Type 6 (30 mcg), Type 11 (40 mcg), Type 16 (60 mcg), Type 18 (40 mcg), Type 31 (20 mcg), Type 33 (20 mcg),
Type 45 (20 mcg), Type 52 (20 mcg), Type 58 (20 mcg).Vaccine is prepared from fermentation cultures of a recombinant strain of yeast Saccharomyces cerevisiae
containing the genes for the human papillomavirus L1 protein of each type (6, 11, 16, 18, 31, 33, 45, 52, 58).Contains no preservative or antibiotics.
Rx only
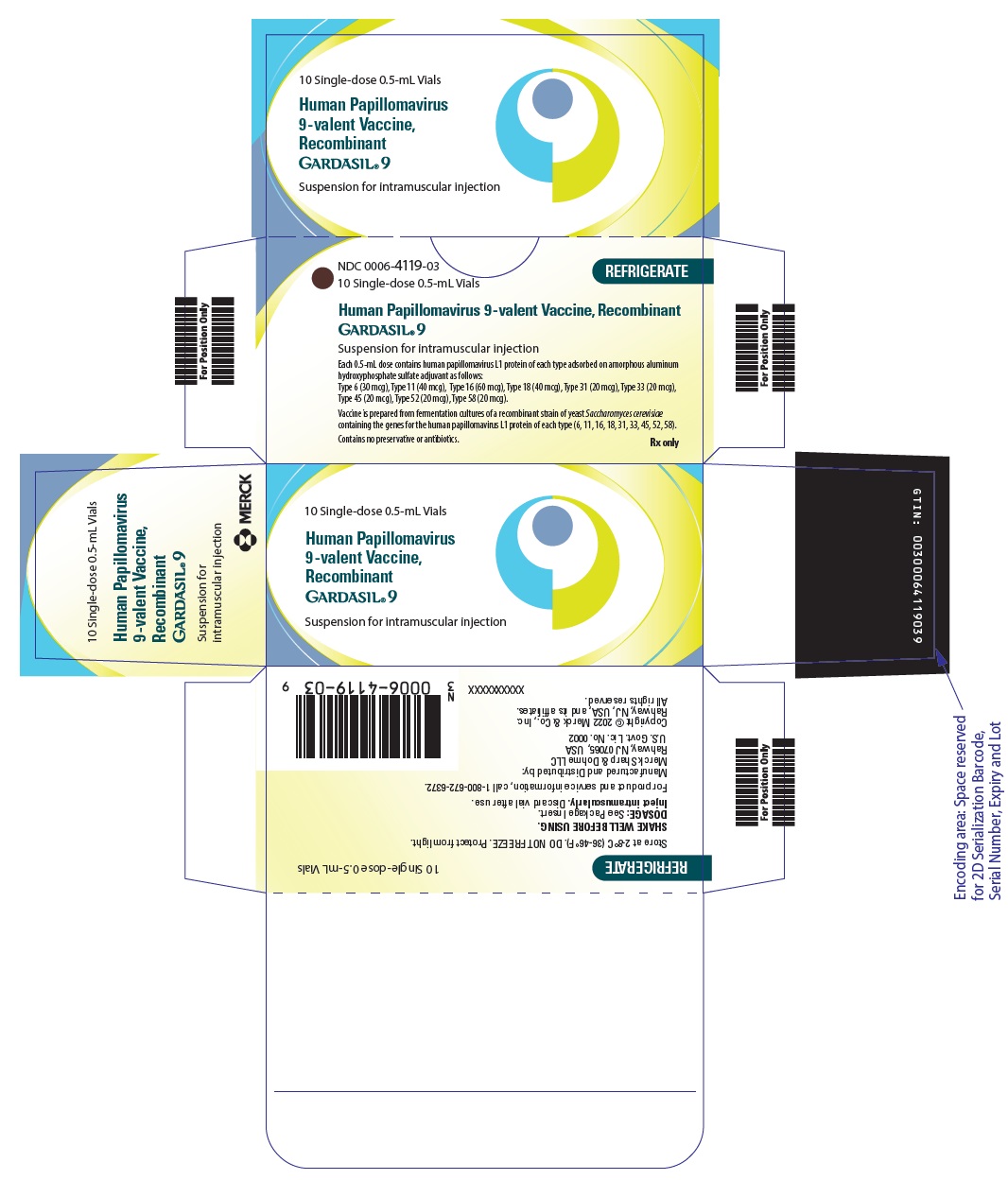
-
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Carton
NDC 0006-4121-02
10 Single-dose 0.5-mL Syringes
REFRIGERATE
Human Papillomavirus
9-valent Vaccine, RecombinantGARDASIL® 9
Suspension for intramuscular injection
Each 0.5-mL dose contains human papillomavirus L1 protein of each type
adsorbed on amorphous aluminum hydroxyphosphate sulfate adjuvant
as follows:
Type 6 (30 mcg), Type 11 (40 mcg), Type 16 (60 mcg), Type 18 (40 mcg),
Type 31 (20 mcg), Type 33 (20 mcg), Type 45 (20 mcg), Type 52 (20 mcg),
Type 58 (20 mcg).Vaccine is prepared from fermentation cultures of a recombinant strain of yeast
Saccharomyces cerevisiae containing the genes for the human papillomavirus
L1 protein of each type (6, 11, 16, 18, 31, 33, 45, 52, 58).Contains no preservative or antibiotics.
Rx only

-
INGREDIENTS AND APPEARANCE
GARDASIL 9
human papillomavirus 9-valent vaccine, recombinant injection, suspensionProduct Information Product Type VACCINE Item Code (Source) NDC:0006-4119 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Human Papillomavirus Type 6 L1 Capsid Protein Antigen (UNII: 61746O90DY) (Human Papillomavirus Type 6 L1 Capsid Protein Antigen - UNII:61746O90DY) Human Papillomavirus Type 6 L1 Capsid Protein Antigen 30 ug in 0.5 mL Human Papillomavirus Type 11 L1 Capsid Protein Antigen (UNII: Z845VHQ61P) (Human Papillomavirus Type 11 L1 Capsid Protein Antigen - UNII:Z845VHQ61P) Human Papillomavirus Type 11 L1 Capsid Protein Antigen 40 ug in 0.5 mL Human Papillomavirus Type 16 L1 Capsid Protein Antigen (UNII: 6LTE2DNX63) (Human Papillomavirus Type 16 L1 Capsid Protein Antigen - UNII:6LTE2DNX63) Human Papillomavirus Type 16 L1 Capsid Protein Antigen 60 ug in 0.5 mL Human Papillomavirus Type 18 L1 Capsid Protein Antigen (UNII: J2D279PEM5) (Human Papillomavirus Type 18 L1 Capsid Protein Antigen - UNII:J2D279PEM5) Human Papillomavirus Type 18 L1 Capsid Protein Antigen 40 ug in 0.5 mL Human Papillomavirus Type 31 L1 Capsid Protein Antigen (UNII: 53JIL371NS) (Human Papillomavirus Type 31 L1 Capsid Protein Antigen - UNII:53JIL371NS) Human Papillomavirus Type 31 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 33 L1 Capsid Protein Antigen (UNII: 759RAC446C) (Human Papillomavirus Type 33 L1 Capsid Protein Antigen - UNII:759RAC446C) Human Papillomavirus Type 33 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 45 L1 Capsid Protein Antigen (UNII: 68S8VCN34F) (Human Papillomavirus Type 45 L1 Capsid Protein Antigen - UNII:68S8VCN34F) Human Papillomavirus Type 45 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 52 L1 Capsid Protein Antigen (UNII: 55644W68FD) (Human Papillomavirus Type 52 L1 Capsid Protein Antigen - UNII:55644W68FD) Human Papillomavirus Type 52 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 58 L1 Capsid Protein Antigen (UNII: 94Y15HP7LF) (Human Papillomavirus Type 58 L1 Capsid Protein Antigen - UNII:94Y15HP7LF) Human Papillomavirus Type 58 L1 Capsid Protein Antigen 20 ug in 0.5 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 9.56 mg in 0.5 mL HISTIDINE (UNII: 4QD397987E) 0.78 mg in 0.5 mL POLYSORBATE 80 (UNII: 6OZP39ZG8H) 50 ug in 0.5 mL SODIUM BORATE (UNII: 91MBZ8H3QO) 35 ug in 0.5 mL WATER (UNII: 059QF0KO0R) Other Ingredients Ingredient Kind Ingredient Name Quantity ADJV ALUMINUM HYDROXYPHOSPHATE SULFATE (UNII: F41V936QZM) 500 ug in 0.5 mL Product Characteristics Color WHITE (white, cloudy) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0006-4119-03 10 in 1 CARTON 1 NDC:0006-4119-01 0.5 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125508 12/10/2014 GARDASIL 9
human papillomavirus 9-valent vaccine, recombinant injection, suspensionProduct Information Product Type VACCINE Item Code (Source) NDC:0006-4121 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Human Papillomavirus Type 6 L1 Capsid Protein Antigen (UNII: 61746O90DY) (Human Papillomavirus Type 6 L1 Capsid Protein Antigen - UNII:61746O90DY) Human Papillomavirus Type 6 L1 Capsid Protein Antigen 30 ug in 0.5 mL Human Papillomavirus Type 11 L1 Capsid Protein Antigen (UNII: Z845VHQ61P) (Human Papillomavirus Type 11 L1 Capsid Protein Antigen - UNII:Z845VHQ61P) Human Papillomavirus Type 11 L1 Capsid Protein Antigen 40 ug in 0.5 mL Human Papillomavirus Type 16 L1 Capsid Protein Antigen (UNII: 6LTE2DNX63) (Human Papillomavirus Type 16 L1 Capsid Protein Antigen - UNII:6LTE2DNX63) Human Papillomavirus Type 16 L1 Capsid Protein Antigen 60 ug in 0.5 mL Human Papillomavirus Type 18 L1 Capsid Protein Antigen (UNII: J2D279PEM5) (Human Papillomavirus Type 18 L1 Capsid Protein Antigen - UNII:J2D279PEM5) Human Papillomavirus Type 18 L1 Capsid Protein Antigen 40 ug in 0.5 mL Human Papillomavirus Type 31 L1 Capsid Protein Antigen (UNII: 53JIL371NS) (Human Papillomavirus Type 31 L1 Capsid Protein Antigen - UNII:53JIL371NS) Human Papillomavirus Type 31 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 33 L1 Capsid Protein Antigen (UNII: 759RAC446C) (Human Papillomavirus Type 33 L1 Capsid Protein Antigen - UNII:759RAC446C) Human Papillomavirus Type 33 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 45 L1 Capsid Protein Antigen (UNII: 68S8VCN34F) (Human Papillomavirus Type 45 L1 Capsid Protein Antigen - UNII:68S8VCN34F) Human Papillomavirus Type 45 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 52 L1 Capsid Protein Antigen (UNII: 55644W68FD) (Human Papillomavirus Type 52 L1 Capsid Protein Antigen - UNII:55644W68FD) Human Papillomavirus Type 52 L1 Capsid Protein Antigen 20 ug in 0.5 mL Human Papillomavirus Type 58 L1 Capsid Protein Antigen (UNII: 94Y15HP7LF) (Human Papillomavirus Type 58 L1 Capsid Protein Antigen - UNII:94Y15HP7LF) Human Papillomavirus Type 58 L1 Capsid Protein Antigen 20 ug in 0.5 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 9.56 mg in 0.5 mL HISTIDINE (UNII: 4QD397987E) 0.78 mg in 0.5 mL POLYSORBATE 80 (UNII: 6OZP39ZG8H) 50 ug in 0.5 mL SODIUM BORATE (UNII: 91MBZ8H3QO) 35 ug in 0.5 mL WATER (UNII: 059QF0KO0R) Other Ingredients Ingredient Kind Ingredient Name Quantity ADJV ALUMINUM HYDROXYPHOSPHATE SULFATE (UNII: F41V936QZM) 500 ug in 0.5 mL Product Characteristics Color WHITE (white, cloudy) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0006-4121-02 10 in 1 CARTON 1 NDC:0006-4121-01 0.5 mL in 1 SYRINGE, GLASS; Type 3: Prefilled Biologic Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125508 12/10/2014 Labeler - Merck Sharp & Dohme LLC (118446553)