PRISTIQ EXTENDED-RELEASE- desvenlafaxine succinate tablet, extended release
Cardinal Health
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use PRISTIQ safely and effectively. See full prescribing information for PRISTIQ.
PRISTIQ® (desvenlafaxine) Extended-Release Tablets, for oral use Initial U.S. Approval: 2008 WARNING: SUICIDAL THOUGHTS AND BEHAVIORSSee full prescribing information for complete boxed warning.RECENT MAJOR CHANGESINDICATIONS AND USAGEPRISTIQ, is a serotonin and norepinephrine reuptake inhibitor (SNRI) indicated for the treatment of major depressive disorder (MDD) (1). DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSCONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions (incidence ≥5% and twice the rate of placebo in the 50 or 100 mg dose groups) were: nausea, dizziness, insomnia, hyperhidrosis, constipation, somnolence, decreased appetite, anxiety, and specific male sexual function disorders (6.1). To report SUSPECTED ADVERSE REACTIONS, contact Wyeth Pharmaceuticals Inc., a subsidiary of Pfizer Inc., at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 11/2016 |
FULL PRESCRIBING INFORMATION
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
Antidepressants increased the risk of suicidal thoughts and behavior in children, adolescents, and young adults in short-term studies. These studies did not show an increase in the risk of suicidal thoughts and behavior with antidepressant use in patients over age 24; there was a reduction in risk with antidepressant use in patients aged 65 and older [see Warnings and Precautions (5.1)].
In patients of all ages who are started on antidepressant therapy, monitor closely for worsening, and for emergence of suicidal thoughts and behaviors. Advise families and caregivers of the need for close observation and communication with the prescriber [see Warnings and Precautions (5.1)].
PRISTIQ is not approved for use in pediatric patients [see Use in Specific Populations (8.4)].
1 INDICATIONS AND USAGE
PRISTIQ, a serotonin and norepinephrine reuptake inhibitor (SNRI), is indicated for the treatment of major depressive disorder (MDD) [see Clinical Studies (14) and Dosage and Administration (2.1)]. The efficacy of PRISTIQ has been established in four short-term (8-week, placebo-controlled studies) and two maintenance studies in adult outpatients who met DSM-IV criteria for major depressive disorder.
2 DOSAGE AND ADMINISTRATION
2.1 GENERAL INSTRUCTION FOR USE
The recommended dose for PRISTIQ is 50 mg once daily, with or without food. The 50 mg dose is both a starting dose and the therapeutic dose. PRISTIQ should be taken at approximately the same time each day. Tablets must be swallowed whole with fluid and not divided, crushed, chewed, or dissolved.
In clinical studies, doses of 10 mg to 400 mg per day were studied. In clinical studies, doses of 50 mg to 400 mg per day were shown to be effective, although no additional benefit was demonstrated at doses greater than 50 mg per day and adverse reactions and discontinuations were more frequent at higher doses.
The 25 mg per day dose is intended for a gradual reduction in dose when discontinuing treatment. When discontinuing therapy, gradual dose reduction is recommended whenever possible to minimize discontinuation symptoms [see Dosage and Administration (2.4) and Warnings and Precautions (5.9)].
2.2 Special Populations
Patients with renal impairment
The maximum recommended dose in patients with moderate renal impairment (24-hr creatinine clearance [CrCl] = 30 to 50 mL/min, Cockcroft-Gault [C-G]) is 50 mg per day. The maximum recommended dose in patients with severe renal impairment (24-hr CrCl less than 30 mL/min, C-G) or end-stage renal disease (ESRD) is 25 mg every day or 50 mg every other day. Supplemental doses should not be given to patients after dialysis [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Patients with hepatic impairment
The recommended dose in patients with moderate to severe hepatic impairment is 50 mg per day. Dose escalation above 100 mg per day is not recommended [see Clinical Pharmacology (12.3)].
2.3 Maintenance/Continuation/Extended Treatment
It is generally agreed that acute episodes of major depressive disorder require several months or longer of sustained pharmacologic therapy. Longer-term efficacy of PRISTIQ (50–400 mg) was established in two maintenance trials [see Clinical Studies (14)]. Patients should be periodically reassessed to determine the need for continued treatment.
2.4 Discontinuing PRISTIQ
Symptoms associated with discontinuation of PRISTIQ, other SNRIs and SSRIs have been reported [see Warnings and Precautions (5.9)]. Patients should be monitored for these symptoms when discontinuing treatment. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the physician may continue decreasing the dose, but at a more gradual rate. The 25 mg dose is available for discontinuing therapy.
2.5 Switching Patients From Other Antidepressants to PRISTIQ
Discontinuation symptoms have been reported when switching patients from other antidepressants, including venlafaxine, to PRISTIQ. Tapering of the initial antidepressant may be necessary to minimize discontinuation symptoms.
2.6 Switching Patients To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders
At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with PRISTIQ. Conversely, at least 7 days should be allowed after stopping PRISTIQ before starting an MAOI intended to treat psychiatric disorders [see Contraindications (4.2)].
Use of PRISTIQ with other MAOIs such as Linezolid or Methylene Blue
Do not start PRISTIQ in a patient who is being treated with linezolid or intravenous methylene blue because there is increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered [see Contraindications (4.2)].
In some cases, a patient already receiving PRISTIQ therapy may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, PRISTIQ should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for 7 days or until 24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with PRISTIQ may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue [see Warnings and Precautions (5.2)].
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with PRISTIQ is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use [see Warnings and Precautions (5.2)].
3 DOSAGE FORMS AND STRENGTHS
PRISTIQ® (desvenlafaxine) Extended-Release Tablets are available as 25 mg, 50 mg and 100 mg tablets.
25 mg, tan, square pyramid tablet debossed with "W" over "25" on the flat side
50 mg, light pink, square pyramid tablet debossed with "W" over "50" on the flat side
100 mg, reddish-orange, square pyramid tablet debossed with "W" over "100" on the flat side
4 CONTRAINDICATIONS
- •
- Hypersensitivity to desvenlafaxine succinate, venlafaxine hydrochloride or to any excipients in the PRISTIQ formulation. Angioedema has been reported in patients treated with PRISTIQ [see Adverse Reactions (6.1)].
- •
- The use of MAOIs intended to treat psychiatric disorders with PRISTIQ or within 7 days of stopping treatment with PRISTIQ is contraindicated because of an increased risk of serotonin syndrome. The use of PRISTIQ within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated [see Dosage and Administration (2.6) and Warnings and Precautions (5.2)].
- •
- Starting PRISTIQ in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome [see Dosage and Administration (2.6) and Warnings and Precautions (5.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Suicidal Thoughts and Behaviors in Children, Adolescents and Young Adults
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the strongest predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment. Pooled analyses of short-term placebo-controlled studies of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18 to 24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled studies in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term studies of 9 antidepressant drugs in over 4,400 patients. The pooled analyses of placebo-controlled studies in adults with MDD or other psychiatric disorders included a total of 295 short-term studies (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs. placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1,000 patients treated) are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Cases of Suicidality per 1,000 Patients Treated |
|---|---|
|
Increases Compared to Placebo |
|
|
<18 |
|
|
18 to 24 |
5 additional cases |
|
Decreases Compared to Placebo |
|
|
25 to 64 |
1 fewer case |
|
≥65 |
6 fewer cases |
No suicides occurred in any of the pediatric studies. There were suicides in the adult studies, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer-term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance studies in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse, or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset, or were not part of the patient's presenting symptoms.
If the decision has been made to discontinue treatment, medication should be tapered, as rapidly as is feasible, but with recognition that abrupt discontinuation can be associated with certain symptoms [see Dosage and Administration (2.4) and Warnings and Precautions (5.7) for a description of the risks of discontinuation of PRISTIQ].
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to healthcare providers. Such monitoring should include daily observation by families and caregivers.
Prescriptions for PRISTIQ should be written for the smallest quantity of tablets consistent with good patient management, in order to reduce the risk of overdose.
Screening patients for bipolar disorder
A major depressive episode may be the initial presentation of bipolar disorder. It is generally believed (though not established in controlled studies) that treating such an episode with an antidepressant alone may increase the likelihood of precipitation of a mixed/manic episode in patients at risk for bipolar disorder. Whether any of the symptoms described above represent such a conversion is unknown. However, prior to initiating treatment with an antidepressant, patients with depressive symptoms should be adequately screened to determine if they are at risk for bipolar disorder; such screening should include a detailed psychiatric history, including a family history of suicide, bipolar disorder, and depression. It should be noted that PRISTIQ is not approved for use in treating bipolar depression.
5.2 Serotonin Syndrome
The development of a potentially life-threatening serotonin syndrome has been reported with SNRIs and SSRIs, including PRISTIQ, alone but particularly with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John's Wort), and with drugs that impair metabolism of serotonin (in particular, MAOIs, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue).
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome.
The concomitant use of PRISTIQ with MAOIs intended to treat psychiatric disorders is contraindicated. PRISTIQ should also not be started in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue. All reports with methylene blue that provided information on the route of administration involved intravenous administration in the dose range of 1 mg/kg to 8 mg/kg. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection) or at lower doses. There may be circumstances when it is necessary to initiate treatment with a MAOI such as linezolid or intravenous methylene blue in a patient taking PRISTIQ. PRISTIQ should be discontinued before initiating treatment with the MAOI [see Contraindications (4.2) and Dosage and Administration (2.6)].
If concomitant use of PRISTIQ with other serotonergic drugs, including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, buspirone, tryptophan, and St. John's Wort is clinically warranted, patients should be made aware of a potential increased risk for serotonin syndrome, particularly during treatment initiation and dose increases.
Treatment with PRISTIQ and any concomitant serotonergic agents should be discontinued immediately if the above events occur and supportive symptomatic treatment should be initiated.
5.3 Elevated Blood Pressure
Patients receiving PRISTIQ should have regular monitoring of blood pressure since increases in blood pressure were observed in clinical studies [see Adverse Reactions (6.1)]. Pre-existing hypertension should be controlled before initiating treatment with PRISTIQ. Caution should be exercised in treating patients with pre-existing hypertension, cardiovascular, or cerebrovascular conditions that might be compromised by increases in blood pressure. Cases of elevated blood pressure requiring immediate treatment have been reported with PRISTIQ.
Sustained blood pressure increases could have adverse consequences. For patients who experience a sustained increase in blood pressure while receiving PRISTIQ, either dose reduction or discontinuation should be considered [see Adverse Reactions (6.1)].
5.4 Abnormal Bleeding
SSRIs and SNRIs, including PRISTIQ, may increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs, warfarin, and other anticoagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Bleeding events related to SSRIs and SNRIs have ranged from ecchymosis, hematoma, epistaxis, and petechiae to life-threatening hemorrhages. Patients should be cautioned about the risk of bleeding associated with the concomitant use of PRISTIQ and NSAIDs, aspirin, or other drugs that affect coagulation or bleeding.
5.5 Angle Closure Glaucoma
Angle-Closure Glaucoma: The pupillary dilation that occurs following use of many antidepressant drugs including Pristiq may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy.
5.6 Activation of Mania/Hypomania
During all MDD phase 2 and phase 3 studies, mania was reported for approximately 0.02% of patients treated with PRISTIQ. Activation of mania/hypomania has also been reported in a small proportion of patients with major affective disorder who were treated with other marketed antidepressants. As with all antidepressants, PRISTIQ should be used cautiously in patients with a history or family history of mania or hypomania.
5.7 Discontinuation Syndrome
Discontinuation symptoms have been systematically and prospectively evaluated in patients treated with PRISTIQ during clinical studies in Major Depressive Disorder. Abrupt discontinuation or dose reduction has been associated with the appearance of new symptoms that include dizziness, nausea, headache, irritability, insomnia, diarrhea, anxiety, fatigue, abnormal dreams, and hyperhidrosis. In general, discontinuation events occurred more frequently with longer duration of therapy.
During marketing of SNRIs (Serotonin and Norepinephrine Reuptake Inhibitors), and SSRIs (Selective Serotonin Reuptake Inhibitors), there have been spontaneous reports of adverse events occurring upon discontinuation of these drugs, particularly when abrupt, including the following: dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g., paresthesia, such as electric shock sensations), anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. While these events are generally self-limiting, there have been reports of serious discontinuation symptoms.
Patients should be monitored for these symptoms when discontinuing treatment with PRISTIQ. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the physician may continue decreasing the dose, but at a more gradual rate [see Dosage and Administration (2.4) and Adverse Reactions (6.1)].
5.8 Seizure
Cases of seizure have been reported in pre-marketing clinical studies with PRISTIQ. PRISTIQ has not been systematically evaluated in patients with a seizure disorder. Patients with a history of seizures were excluded from pre-marketing clinical studies. PRISTIQ should be prescribed with caution in patients with a seizure disorder.
5.9 Hyponatremia
Hyponatremia may occur as a result of treatment with SSRIs and SNRIs, including PRISTIQ. In many cases, this hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH). Cases with serum sodium lower than 110 mmol/L have been reported. Elderly patients may be at greater risk of developing hyponatremia with SSRIs and SNRIs. Also, patients taking diuretics or who are otherwise volume depleted can be at greater risk [see Use in Specific Populations (8.5) and Clinical Pharmacology (12.6)]. Discontinuation of PRISTIQ should be considered in patients with symptomatic hyponatremia and appropriate medical intervention should be instituted.
Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which can lead to falls. Signs and symptoms associated with more severe and/or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death.
5.10 Interstitial Lung Disease and Eosinophilic Pneumonia
Interstitial lung disease and eosinophilic pneumonia associated with venlafaxine (the parent drug of PRISTIQ) therapy have been rarely reported. The possibility of these adverse events should be considered in patients treated with PRISTIQ who present with progressive dyspnea, cough, or chest discomfort. Such patients should undergo a prompt medical evaluation, and discontinuation of PRISTIQ should be considered.
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the label.
- •
- Hypersensitivity [see Contraindications (4)]
- •
- Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Warnings and Precautions (5.1)]
- •
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
- •
- Elevated Blood Pressure [see Warnings and Precautions (5.3)]
- •
- Abnormal Bleeding [see Warnings and Precautions (5.4)]
- •
- Angle Closure Glaucoma [see Warnings and Precautions (5.5)]
- •
- Activation of Mania/Hypomania [see Warnings and Precautions (5.6)]
- •
- Discontinuation Syndrome [see Warnings and Precautions (5.7)]
- •
- Seizure [see Warnings and Precautions (5.8)]
- •
- Hyponatremia [see Warnings and Precautions (5.9)]
- •
- Interstitial Lung Disease and Eosinophilic Pneumonia [see Warnings and Precautions (5.10)]
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
Patient exposure
PRISTIQ was evaluated for safety in 8,394 patients diagnosed with major depressive disorder who participated in multiple-dose pre-marketing studies, representing 2,784 patient-years of exposure. Of the total 8,394 patients exposed to at least one dose of PRISTIQ; 2,116 were exposed to PRISTIQ for 6 months, representing 1,658 patient-years of exposure, and 421 were exposed for one year, representing 416 patient-years of exposure.
Adverse reactions reported as reasons for discontinuation of treatment
In the pre-marketing pooled 8-week placebo-controlled studies in patients with MDD, 1,834 patients were exposed to PRISTIQ (50 to 400 mg). Of the 1,834 patients, 12% discontinued treatment due to an adverse reaction, compared with 3% of the 1,116 placebo-treated patients. At the recommended dose of 50 mg, the discontinuation rate due to an adverse reaction for PRISTIQ (4.1%) was similar to the rate for placebo (3.8%). For the 100 mg dose of PRISTIQ the discontinuation rate due to an adverse reaction was 8.7%.
The most common adverse reactions leading to discontinuation in at least 2% and at a rate greater than placebo of the PRISTIQ treated patients in the short-term studies, up to 8 weeks, were: nausea (4%); dizziness, headache and vomiting (2% each). In a longer-term study, up to 9 months, the most common was vomiting (2%).
Common adverse reactions in placebo-controlled MDD studies
The most commonly observed adverse reactions in PRISTIQ treated MDD patients in pre-marketing pooled 8-week, placebo-controlled, fixed-dose studies (incidence ≥ 5% and at least twice the rate of placebo in the 50 or 100 mg dose groups) were: nausea, dizziness, insomnia, hyperhidrosis, constipation, somnolence, decreased appetite, anxiety, and specific male sexual function disorders.
Table 2 shows the incidence of common adverse reactions that occurred in ≥ 2% of PRISTIQ treated MDD patients and twice the rate of placebo at any dose in the pre-marketing pooled 8-week, placebo-controlled, fixed dose clinical studies
| Percentage of Patients Reporting Reaction | |||||
|---|---|---|---|---|---|
| PRISTIQ | |||||
| System Organ Class
Preferred Term | Placebo
(n=636) | 50 mg
(n=317) | 100 mg
(n=424) | 200 mg
(n=307) | 400 mg
(n=317) |
|
Cardiac disorders |
|||||
|
Blood pressure increased |
1 |
1 |
1 |
2 |
2 |
|
Gastrointestinal disorders |
|||||
|
Nausea |
10 |
22 |
26 |
36 |
41 |
|
Dry mouth |
9 |
11 |
17 |
21 |
25 |
|
Constipation |
4 |
9 |
9 |
10 |
14 |
|
Vomiting |
3 |
3 |
4 |
6 |
9 |
|
General disorders and administration site conditions |
|||||
|
Fatigue |
4 |
7 |
7 |
10 |
11 |
|
Chills |
1 |
1 |
<1 |
3 |
4 |
|
Feeling jittery |
1 |
1 |
2 |
3 |
3 |
|
Metabolism and nutrition disorders |
|||||
|
Decreased appetite |
2 |
5 |
8 |
10 |
10 |
|
Nervous system disorders |
|||||
|
Dizziness |
5 |
13 |
10 |
15 |
16 |
|
Somnolence |
4 |
4 |
9 |
12 |
12 |
|
Tremor |
2 |
2 |
3 |
9 |
9 |
|
Disturbance in attention |
<1 |
<1 |
1 |
2 |
1 |
|
Psychiatric disorders |
|||||
|
Insomnia |
6 |
9 |
12 |
14 |
15 |
|
Anxiety |
2 |
3 |
5 |
4 |
4 |
|
Nervousness |
1 |
<1 |
1 |
2 |
2 |
|
Abnormal dreams |
1 |
2 |
3 |
2 |
4 |
|
Renal and urinary disorders |
|||||
|
Urinary hesitation |
0 |
<1 |
1 |
2 |
2 |
|
Respiratory, thoracic and mediastinal disorders |
|||||
|
Yawning |
<1 |
1 |
1 |
4 |
3 |
|
Skin and subcutaneous tissue disorders |
|||||
|
Hyperhidrosis |
4 |
10 |
11 |
18 |
21 |
|
Special Senses |
|||||
|
Vision blurred |
1 |
3 |
4 |
4 |
4 |
|
Mydriasis |
<1 |
2 |
2 |
6 |
6 |
|
Vertigo |
1 |
2 |
1 |
5 |
3 |
|
Tinnitus |
1 |
2 |
1 |
1 |
2 |
|
Dysgeusia |
1 |
1 |
1 |
1 |
2 |
|
Vascular disorders |
|||||
|
Hot flush |
<1 |
1 |
1 |
2 |
2 |
Sexual function adverse reactions
Table 3 shows the incidence of sexual function adverse reactions that occurred in ≥ 2% of PRISTIQ treated MDD patients in any fixed-dose group (pre-marketing pooled 8-week, placebo-controlled, fixed -dose, clinical studies).
|
PRISTIQ |
|||||
|
Placebo
|
50 mg
|
100 mg
|
200 mg
|
400 mg
|
|
|
Men only |
|||||
|
Anorgasmia |
0 |
0 |
3 |
5 |
8 |
|
Libido decreased |
1 |
4 |
5 |
6 |
3 |
|
Orgasm abnormal |
0 |
0 |
1 |
2 |
3 |
|
Ejaculation delayed |
<1 |
1 |
5 |
7 |
6 |
|
Erectile dysfunction |
1 |
3 |
6 |
8 |
11 |
|
Ejaculation disorder |
0 |
0 |
1 |
2 |
5 |
|
Ejaculation failure |
0 |
1 |
0 |
2 |
2 |
|
Sexual dysfunction |
0 |
1 |
0 |
0 |
2 |
|
PRISTIQ |
|||||
|
Placebo
|
50 mg
|
100 mg
|
200 mg
|
400 mg
|
|
|
Women only |
|||||
|
Anorgasmia |
0 |
1 |
1 |
0 |
3 |
Other adverse reactions observed in premarketing and postmarketing clinical studies
Other infrequent adverse reactions, not described elsewhere in the label, occurring at an incidence of < 2% in MDD patients treated with PRISTIQ were:
Cardiac disorders – Tachycardia.
General disorders and administration site conditions – Asthenia.
Investigations – Weight increased, liver function test abnormal, blood prolactin increased.
Musculoskeletal and connective tissue disorders – Musculoskeletal stiffness.
Nervous system disorders –Syncope, convulsion, dystonia.
Psychiatric disorders – Depersonalization, bruxism.
Renal and urinary disorders – Urinary retention.
Skin and subcutaneous tissue disorders – Rash, alopecia, photosensitivity reaction, angioedema.
In clinical studies, there were uncommon reports of ischemic cardiac adverse reactions, including myocardial ischemia, myocardial infarction, and coronary occlusion requiring revascularization; these patients had multiple underlying cardiac risk factors. More patients experienced these events during PRISTIQ treatment as compared to placebo.
Laboratory, ECG and vital sign changes observed in MDD clinical studies
The following changes were observed in pre-marketing placebo-controlled, short-term MDD studies with PRISTIQ.
Lipids
Elevations in fasting serum total cholesterol, LDL (low density lipoproteins) cholesterol, and triglycerides occurred in the controlled studies. Some of these abnormalities were considered potentially clinically significant.
The percentage of patients who exceeded a predetermined threshold value is shown in Table 4.
| PRISTIQ | |||||
|---|---|---|---|---|---|
| Placebo | 50 mg | 100 mg | 200 mg | 400 mg | |
|
Total Cholesterol |
2 |
3 |
4 |
4 |
10 |
|
LDL Cholesterol |
0 |
1 |
0 |
1 |
2 |
|
Triglycerides, fasting |
3 |
2 |
1 |
4 |
6 |
Proteinuria
Proteinuria, greater than or equal to trace, was observed in the pre-marketing fixed-dose controlled studies (see Table 5). This proteinuria was not associated with increases in BUN or creatinine and was generally transient.
| PRISTIQ | |||||
|---|---|---|---|---|---|
| Placebo | 50 mg | 100 mg | 200 mg | 400 mg | |
|
Proteinuria |
4 |
6 |
8 |
5 |
7 |
Vital sign changes
Table 6 summarizes the changes that were observed in placebo-controlled, short-term, pre-marketing studies with PRISTIQ in patients with MDD (doses 50 to 400 mg).
| PRISTIQ | |||||
|---|---|---|---|---|---|
| Placebo | 50 mg | 100 mg | 200 mg | 400 mg | |
|
Blood pressure |
|||||
|
Supine systolic bp (mm Hg) |
-1.4 |
1.2 |
2.0 |
2.5 |
2.1 |
|
Supine diastolic bp (mm Hg) |
-0.6 |
0.7 |
0.8 |
1.8 |
2.3 |
|
Pulse rate |
|||||
|
Supine pulse (bpm) |
-0.3 |
1.3 |
1.3 |
0.9 |
4.1 |
|
Weight (kg) |
0.0 |
-0.4 |
-0.6 |
-0.9 |
-1.1 |
Treatment with PRISTIQ at all doses from 50 mg per day to 400 mg per day in controlled studies was associated with sustained hypertension, defined as treatment-emergent supine diastolic blood pressure (SDBP) ≥90 mm Hg and ≥10 mm Hg above baseline for 3 consecutive on-therapy visits (see Table 7). Analyses of patients in PRISTIQ pre-marketing short-term controlled studies who met criteria for sustained hypertension revealed a consistent increase in the proportion of patients who developed sustained hypertension. This was seen at all doses with a suggestion of a higher rate at 400 mg per day.
| Treatment Group | Proportion of Patients with Sustained Hypertension |
|---|---|
|
Placebo |
0.5% |
|
PRISTIQ 50 mg per day |
1.3% |
|
PRISTIQ 100 mg per day |
0.7% |
|
PRISTIQ 200 mg per day |
1.1% |
|
PRISTIQ 400 mg per day |
2.3% |
Orthostatic hypotension
In the pre-marketing short-term, placebo-controlled clinical studies with doses of 50 to 400 mg, systolic orthostatic hypotension (decrease ≥30 mm Hg from supine to standing position) occurred more frequently in patients ≥65 years of age receiving PRISTIQ (8%, 7/87) versus placebo (2.5%, 1/40), compared to patients <65 years of age receiving PRISTIQ (0.9%, 18/1,937) versus placebo (0.7%, 8/1,218).
6.2 Postmarketing Experience
The following adverse reaction has been identified during post-approval use of PRISTIQ. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Skin and subcutaneous tissue disorders – Stevens-Johnson syndrome.
7 DRUG INTERACTIONS
7.1 Monoamine Oxidase Inhibitors (MAOI)
Do not use MAOIs intended to treat psychiatric disorders with desvenlafaxine or within 7 days of stopping treatment with desvenlafaxine. Do not use desvenlafaxine within 14 days of stopping an MAOI intended to treat psychiatric disorders. In addition, do not start desvenlafaxine in a patient who is being treated with linezolid or intravenous methylene blue [see Dosage and Administration (2.6 ), Contraindications (4) and Warnings and Precautions (5.2)].
7.2 Serotonergic Drugs
Based on the mechanism of action of desvenlafaxine and the potential for serotonin syndrome, caution is advised when desvenlafaxine is co-administered with other drugs that may affect the serotonergic neurotransmitter systems [see Dosage and Administration (2.6), Contraindications (4) and Warnings and Precautions (5.2)].
7.3 Drugs that Interfere with Hemostasis (e.g., NSAIDs, Aspirin, and Warfarin)
Serotonin release by platelets plays an important role in hemostasis. Epidemiological studies of case-control and cohort design that have demonstrated an association between use of psychotropic drugs that interfere with serotonin reuptake and the occurrence of upper gastrointestinal bleeding. These studies have also shown that concurrent use of an NSAID or aspirin may potentiate this risk of bleeding. Altered anticoagulant effects, including increased bleeding, have been reported when SSRIs and SNRIs are co-administered with warfarin. Patients receiving warfarin therapy should be carefully monitored when PRISTIQ is initiated or discontinued [see Warnings and Precautions (5.4)].
7.4 Potential for Other Drugs to Affect Desvenlafaxine
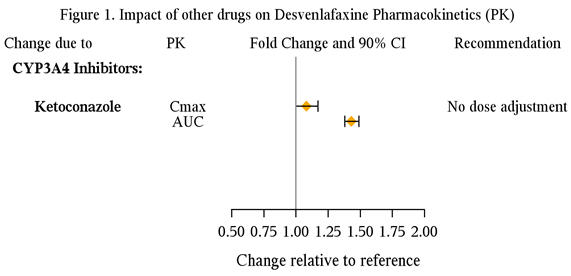
Based on in vitro data, no dose adjustment is required for PRISTIQ when used concomitantly with inhibitors of CYP3A4 and CYP1A1, 1A2, 2A6, 2D6, 2C8, 2C9, 2C19, 2E1, and the P-glycoprotein transporter. Clinical studies have demonstrated no clinically significant pharmacokinetic interaction between PRISTIQ and strong CYP 3A4 inhibitors (Figure 1).
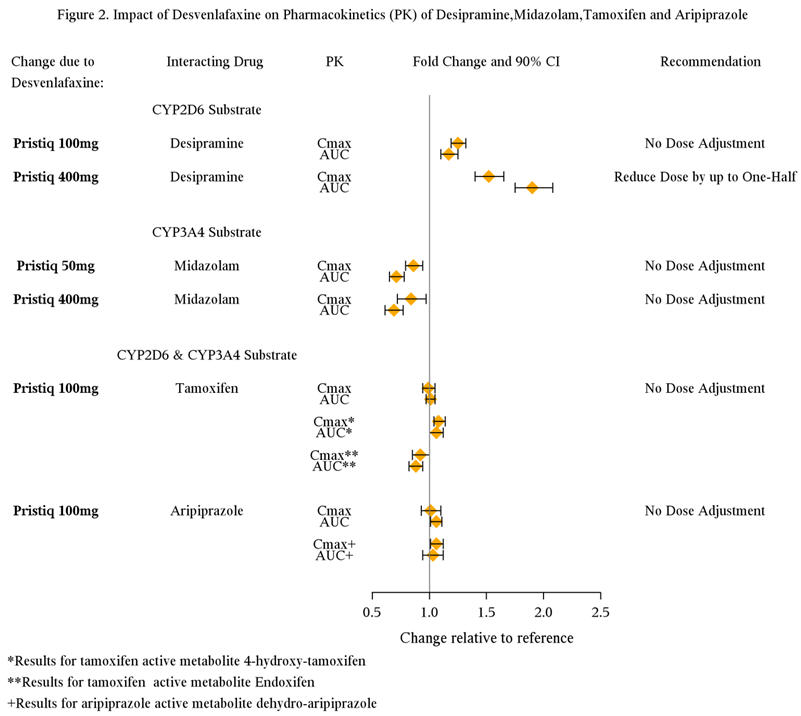
7.5 Potential for Desvenlafaxine to Affect Other Drugs
Clinical studies have shown that desvenlafaxine does not have a clinically relevant effect on CYP2D6 metabolism at the dose of 100 mg daily (Figure 2). Substrates primarily metabolized by CYP2D6 (e.g., desipramine , atomoxetine, dextromethorphan, metoprolol, nebivolol, perphenazine, tolterodine) should be dosed at the original level when co-administered with PRISTIQ 100 mg or lower or when PRISTIQ is discontinued. Reduce the dose of these substrates by up to one-half if co-administered with 400 mg of PRISTIQ.
No additional dose adjustment is required for concomitant use of substrates of CYP3A4, 1A2, 2A6, 2C8, 2C9, and 2C19 isozymes, and P-glycoprotein transporter. Clinical studies have demonstrated no clinically significant pharmacokinetic interaction between PRISTIQ and CYP3A4 substrates (Figure 2).
Clinical studies have shown that desvenlafaxine (100 mg daily) does not have a clinically relevant effect on tamoxifen and aripiprazole, compounds that are metabolized by a combination of both CYP2D6 and CYP3A4 enzymes (Figure 2).
In vitro studies showed minimal inhibitory effect of desvenlafaxine on the CYP2D6 isoenzyme.
In vitro, desvenlafaxine does not inhibit or induce the CYP3A4 isozyme.
In vitro, desvenlafaxine does not inhibit CYP1A2, 2A6, 2C8, 2C9, and 2C19, isozymes, and P-glycoprotein transporter and would not be expected to affect the pharmacokinetics of drugs that are substrates of these CYP isozymes and transporter.
7.6 Other Drugs Containing Desvenlafaxine or Venlafaxine
Avoid use of PRISTIQ with other desvenlafaxine-containing products or venlafaxine products. The concomitant use of PRISTIQ with other desvenlafaxine-containing products or venlafaxine will increase desvenlafaxine blood levels and increase dose-related adverse reactions [see Adverse Reactions (6)].
7.7 Ethanol
A clinical study has shown that PRISTIQ does not increase the impairment of mental and motor skills caused by ethanol. However, as with all CNS-active drugs, patients should be advised to avoid alcohol consumption while taking PRISTIQ.
7.8 Drug-Laboratory Test Interactions
False-positive urine immunoassay screening tests for phencyclidine (PCP) and amphetamine have been reported in patients taking desvenlafaxine. This is due to lack of specificity of the screening tests. False positive test results may be expected for several days following discontinuation of desvenlafaxine therapy. Confirmatory tests, such as gas chromatography/mass spectrometry, will distinguish desvenlafaxine from PCP and amphetamine.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
Risk summary
There are no adequate and well-controlled studies of PRISTIQ in pregnant women. In reproductive developmental studies in rats and rabbits with desvenlafaxine succinate, evidence of teratogenicity was not observed at doses up to 30 times a human dose of 100 mg per day (on a mg/m2 basis) in rats, and up to 15 times a human dose of 100 mg per day (on a mg/m2 basis) in rabbits. An increase in rat pup deaths was seen during the first 4 days of lactation when dosing occurred during gestation and lactation, at doses greater than 10 times a human dose of 100 mg per day (on a mg/m2 basis). PRISTIQ should be used during pregnancy only if the potential benefits justify the potential risks to the fetus.
Clinical considerations
A prospective longitudinal study of 201 women with history of major depression who were euthymic at the beginning of pregnancy, showed women who discontinued antidepressant medication during pregnancy were more likely to experience a relapse of major depression than women who continued antidepressant medication.
Human data
Neonates exposed to SNRIs (Serotonin and Norepinephrine Reuptake Inhibitors), or SSRIs (Selective Serotonin Reuptake Inhibitors), late in the third trimester have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding. Such complications can arise immediately upon delivery. Reported clinical findings have included respiratory distress, cyanosis, apnea, seizures, temperature instability, feeding difficulty, vomiting, hypoglycemia, hypotonia, hypertonia, hyperreflexia, tremor, jitteriness, irritability, and constant crying. These features are consistent with either a direct toxic effect of SSRIs and SNRIs or, possibly, a drug discontinuation syndrome. It should be noted that, in some cases, the clinical picture is consistent with serotonin syndrome [see Warnings and Precautions (5.2)].
Animal data
When desvenlafaxine succinate was administered orally to pregnant rats and rabbits during the period of organogenesis at doses up to 300 mg/kg/day and 75 mg/kg/day, respectively, no teratogenic effects were observed. These doses are 30 times a human dose of 100 mg per day (on a mg/m2 basis) in rats and 15 times a human dose of 100 mg per day (on a mg/m2 basis) in rabbits. However, fetal weights were decreased and skeletal ossification was delayed in rats in association with maternal toxicity at the highest dose, with a no-effect dose 10 times a human dose of 100 mg per day (on a mg/m2 basis).
When desvenlafaxine succinate was administered orally to pregnant rats throughout gestation and lactation, there was a decrease in pup weights and an increase in pup deaths during the first four days of lactation at the highest dose of 300 mg/kg/day. The cause of these deaths is not known. The no-effect dose for rat pup mortality was 10 times a human dose of 100 mg per day (on a mg/m2 basis). Post-weaning growth and reproductive performance of the progeny were not affected by maternal treatment with desvenlafaxine succinate at a dose 30 times a human dose of 100 mg per day (on a mg/m2 basis).
8.3 Nursing Mothers
Desvenlafaxine (O-desmethylvenlafaxine) is excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from PRISTIQ, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established [see Boxed Warning and Warnings and Precautions (5.1)]. Anyone considering the use of PRISTIQ in a child or adolescent must balance the potential risks with the clinical need.
8.5 Geriatric Use
Of the 4,158 patients in pre-marketing clinical studies with PRISTIQ, 6% were 65 years of age or older. No overall differences in safety or efficacy were observed between these patients and younger patients; however, in the short-term placebo-controlled studies, there was a higher incidence of systolic orthostatic hypotension in patients ≥65 years of age compared to patients <65 years of age treated with PRISTIQ [see Adverse Reactions (6)]. For elderly patients, possible reduced renal clearance of PRISTIQ should be considered when determining dose [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
SSRIs and SNRIs, including PRISTIQ, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event [see Warnings and Precautions (5.9)].
8.6 Other Patient Factors
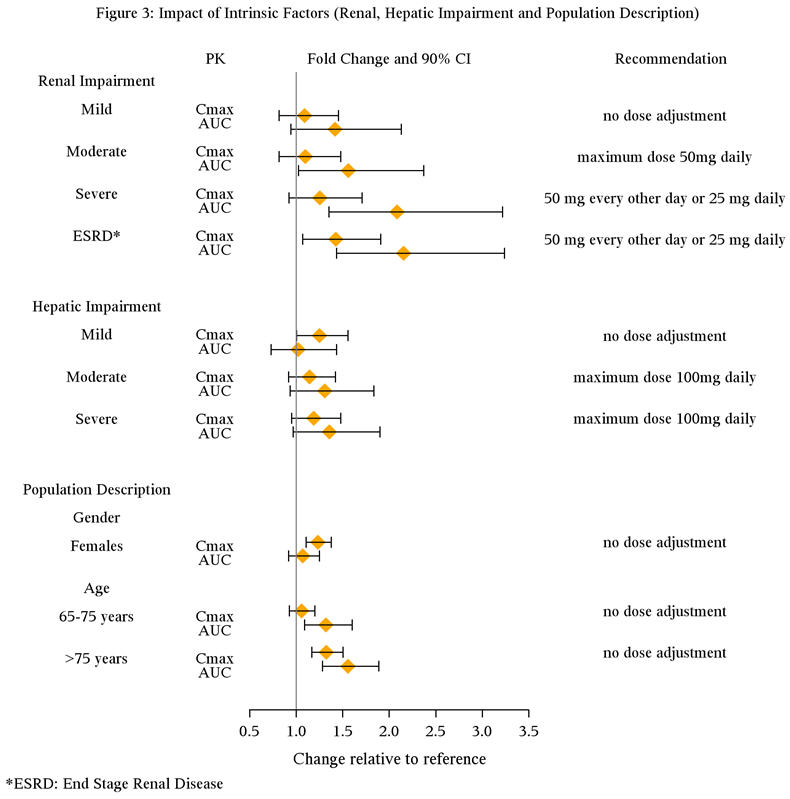
The effect of intrinsic patient factors on the pharmacokinetics of PRISTIQ is presented in Figure 3.
Renal Impairment:
In subjects with renal impairment the clearance of PRISTIQ was decreased. In subjects with severe renal impairment (24-hr CrCl <30 mL/min, Cockcroft-Gault) and end-stage renal disease, elimination half-lives were significantly prolonged, increasing exposures to PRISTIQ; therefore, dosage adjustment is recommended in these patients [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
Hepatic Impairment:
The mean terminal half-life (t1/2) changed from approximately 10 hours in healthy subjects and subjects with mild hepatic impairment to 13 and 14 hours in moderate and severe hepatic impairment, respectively. The recommended dose in patients with moderate to severe hepatic impairment is 50 mg per day. Dose escalation above 100 mg per day is not recommended [see Clinical Pharmacology (12.3)].
10 OVERDOSAGE
10.1 Human Experience with Overdosage
There is limited clinical trial experience with desvenlafaxine succinate overdosage in humans. However, desvenlafaxine (PRISTIQ) is the major active metabolite of venlafaxine. Overdose experience reported with venlafaxine (the parent drug of PRISTIQ) is presented below; the identical information can be found in the Overdosage section of the venlafaxine package insert.
In postmarketing experience, overdose with venlafaxine (the parent drug of PRISTIQ) has occurred predominantly in combination with alcohol and/or other drugs. The most commonly reported events in overdosage include tachycardia, changes in level of consciousness (ranging from somnolence to coma), mydriasis, seizures, and vomiting. Electrocardiogram changes (e.g., prolongation of QT interval, bundle branch block, QRS prolongation), sinus and ventricular tachycardia, bradycardia, hypotension, rhabdomyolysis, vertigo, liver necrosis, serotonin syndrome, and death have been reported.
Published retrospective studies report that venlafaxine overdosage may be associated with an increased risk of fatal outcomes compared to that observed with SSRI antidepressant products, but lower than that for tricyclic antidepressants. Epidemiological studies have shown that venlafaxine-treated patients have a higher pre-existing burden of suicide risk factors than SSRI-treated patients. The extent to which the finding of an increased risk of fatal outcomes can be attributed to the toxicity of venlafaxine in overdosage, as opposed to some characteristic(s) of venlafaxine-treated patients, is not clear.
11 DESCRIPTION
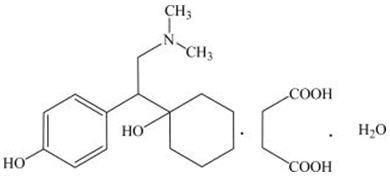
PRISTIQ is an extended-release tablet for oral administration that contains desvenlafaxine succinate, a structurally novel SNRI for the treatment of MDD. Desvenlafaxine (O-desmethylvenlafaxine) is the major active metabolite of the antidepressant venlafaxine, a medication used to treat major depressive disorder.
Desvenlafaxine is designated RS-4-[2-dimethylamino-1-(1-hydroxycyclohexyl)ethyl]phenol and has the empirical formula of C16H25NO2 (free base) and C16H25NO2∙C4H6O4∙H2O (succinate monohydrate). Desvenlafaxine succinate monohydrate has a molecular weight of 399.48. The structural formula is shown below.
Desvenlafaxine succinate is a white to off-white powder that is soluble in water. The solubility of desvenlafaxine succinate is pH dependent. Its octanol:aqueous system (at pH 7.0) partition coefficient is 0.21.
PRISTIQ is formulated as an extended-release tablet for once-a-day oral administration.
Each tablet contains 38 mg, 76 mg or 152 mg of desvenlafaxine succinate equivalent to 25 mg, 50 mg or 100 mg of desvenlafaxine, respectively.
Inactive ingredients for the 25 mg tablet consist of hypromellose, microcrystalline cellulose, talc, magnesium stearate, a film coating which consists of polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, and iron oxides.
Inactive ingredients for the 50 mg tablet consist of hypromellose, microcrystalline cellulose, talc, magnesium stearate and film coating, which consists of polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, and iron oxides.
Inactive ingredients for the 100 mg tablet consist of hypromellose, microcrystalline cellulose, talc, magnesium stearate and film coating, which consists of polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, iron oxide and FD&C yellow #6.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The exact mechanism of the antidepressant action of desvenlafaxine is unknown, but is thought to be related to the potentiation of serotonin and norepinephrine in the central nervous system, through inhibition of their reuptake. Non-clinical studies have shown that desvenlafaxine is a potent and selective serotonin and norepinephrine reuptake inhibitor (SNRI).
12.2 Pharmacodynamics
Desvenlafaxine lacked significant affinity for numerous receptors, including muscarinic-cholinergic, H1-histaminergic, or α1-adrenergic receptors in vitro. Desvenlafaxine also lacked monoamine oxidase (MAO) inhibitory activity.
ECG changes
Electrocardiograms were obtained from 1,492 desvenlafaxine treated patients with major depressive disorder and 984 placebo-treated patients in clinical studies lasting up to 8 weeks. No clinically relevant differences were observed between desvenlafaxine treated and placebo-treated patients for QT, QTc, PR, and QRS intervals. In a thorough QTc study with prospectively determined criteria, desvenlafaxine did not cause QT prolongation. No difference was observed between placebo and desvenlafaxine treatments for the QRS interval.
12.3 Pharmacokinetics
The single-dose pharmacokinetics of desvenlafaxine are linear and dose-proportional in a dose range of 50 to 600 mg per day. With once-daily dosing, steady-state plasma concentrations are achieved within approximately 4 to 5 days. At steady-state, multiple-dose accumulation of desvenlafaxine is linear and predictable from the single-dose pharmacokinetic profile.
Absorption and distribution
The absolute oral bioavailability of PRISTIQ after oral administration is about 80%.
A food-effect study involving administration of PRISTIQ to healthy subjects under fasting and fed conditions (high-fat meal, 800 to 1000 calories) indicated that desvenlafaxine Cmax was increased about 16% in the fed state, while the AUCs were similar. This difference is not expected to be clinically significant; therefore, PRISTIQ can be taken without regard to meals [see Dosage and Administration (2.1)].
The plasma protein binding of desvenlafaxine is low (30%) and is independent of drug concentration. The desvenlafaxine volume of distribution at steady-state following intravenous administration is 3.4 L/kg, indicating distribution into nonvascular compartments.
Metabolism and elimination
Desvenlafaxine is primarily metabolized by conjugation (mediated by UGT isoforms) and, to a minor extent, through oxidative metabolism. CYP3A4 is the cytochrome P450 isozyme mediating the oxidative metabolism (N-demethylation) of desvenlafaxine. The CYP2D6 metabolic pathway is not involved, and after administration of 100 mg, the pharmacokinetics of desvenlafaxine was similar in subjects with CYP2D6 poor and extensive metabolizer phenotype. Approximately 45% of desvenlafaxine is excreted unchanged in urine at 72 hours after oral administration. Approximately 19% of the administered dose is excreted as the glucuronide metabolite and <5% as the oxidative metabolite (N,O-didesmethylvenlafaxine) in urine.
Drug interaction studies
Inhibitors of CYP3A4 (ketoconazole)
CYP3A4 is a minor pathway for the metabolism of desvenlafaxine. In a clinical study, ketoconazole (200 mg BID) increased the area under the concentration vs. time curve (AUC) of desvenlafaxine (400 mg single dose) by about 43% and Cmax by about 8%. Concomitant use of desvenlafaxine with potent inhibitors of CYP3A4 may result in higher concentrations of desvenlafaxine.
Inhibitors of other CYP enzymes
Based on in vitro data, drugs that inhibit CYP isozymes 1A1, 1A2, 2A6, 2D6, 2C8, 2C9, 2C19, and 2E1 are not expected to have significant impact on the pharmacokinetic profile of desvenlafaxine.
Drugs metabolized by CYP2D6 (e.g. desipramine, dextromethorphan, metoprolol, atomoxetine)
In vitro studies showed minimal inhibitory effect of desvenlafaxine on CYP2D6. Clinical studies have shown that desvenlafaxine does not have a clinically relevant effect on CYP2D6 metabolism at the dose of 100 mg daily. When desvenlafaxine succinate was administered at a dose of 100 mg daily in conjunction with a single 50 mg dose of desipramine, a CYP2D6 substrate, the Cmax and AUC of desipramine increased approximately 25% and 17%, respectively. When 400 mg (8 times the recommended 50 mg dose) was administered, the Cmax and AUC of desipramine increased approximately 50% and 90%, respectively. Concomitant use of desvenlafaxine with a drug metabolized by CYP2D6 can result in higher concentrations of that drug [see Drug Interactions (7.5)].
Drugs metabolized by CYP3A4 (midazolam)
In vitro, desvenlafaxine does not inhibit or induce the CYP3A4 isozyme. In a clinical study, desvenlafaxine 400 mg daily (8 times the recommended 50 mg dose) was co-administered with a single 4 mg dose of midazolam (a CYP3A4 substrate). The AUC and Cmax of midazolam decreased by approximately 31% and 16%, respectively. Concomitant use of desvenlafaxine with a drug metabolized by CYP3A4 can result in lower exposures to that drug.
Drugs metabolized by CYP1A2, 2A6, 2C8, 2C9 and 2C19
In vitro, desvenlafaxine does not inhibit CYP1A2, 2A6, 2C8, 2C9, and 2C19 isozymes and would not be expected to affect the pharmacokinetics of drugs that are metabolized by these CYP isozymes.
In vitro, desvenlafaxine is not a substrate or an inhibitor for the P-glycoprotein transporter. The pharmacokinetics of desvenlafaxine are unlikely to be affected by drugs that inhibit the P-glycoprotein transporter, and desvenlafaxine is not likely to affect the pharmacokinetics of drugs that are substrates of the P-glycoprotein transporter.
Special populations
Age
In a study of healthy subjects administered doses of up to 300 mg, there was an approximate 32% increase in Cmax and a 55% increase in AUC in subjects older than 75 years of age (n = 17), compared with subjects 18 to 45 years of age (n = 16). Subjects 65 to 75 years of age (n = 15) had no change in Cmax, but an approximately 32% increase in AUC, compared to subjects 18 to 45 years of age [see Dosage and Administration (2.2)].
Gender
In a study of healthy subjects administered doses of up to 300 mg, women had an approximately 25% higher Cmax and an approximately 10% higher AUC than age-matched men. No adjustment of dosage on the basis of gender is needed.
Race
Pharmacokinetic analysis showed that race (White, n = 466; Black, n = 97; Hispanic, n = 39; Other, n = 33) had no apparent effect on the pharmacokinetics of PRISTIQ. No adjustment of dosage on the basis of race is needed.
Hepatic insufficiency
The disposition of desvenlafaxine succinate after administration of 100 mg was studied in subjects with mild (Child-Pugh A, n = 8), moderate (Child-Pugh B, n = 8), and severe (Child-Pugh C, n = 8) hepatic impairment and to healthy subjects (n = 12).
Average AUC was increased by approximately 31% and 35% in patients with moderate and severe hepatic impairment, respectively, as compared to healthy subjects. Average AUC values were similar in subjects with mild hepatic impairment and healthy subjects (< 5% difference).
Systemic clearance (CL/F) was decreased by approximately 20% and 36% in patients with moderate and severe hepatic impairment, respectively, as compared to healthy subjects. CL/F values were comparable in mild hepatic impairment and healthy subjects (< 5% difference).
The mean t1/2 changed from approximately 10 hours in healthy subjects and subjects with mild hepatic impairment to 13 and 14 hours in moderate and severe hepatic impairment, respectively. The recommended dose in patients with hepatic impairment is 50 mg per day. Dose escalation above 100 mg per day is not recommended [see Use in Specific Populations (8.6)].
Renal insufficiency
The disposition of desvenlafaxine after administration of 100 mg was studied in subjects with mild (n = 9), moderate (n = 8), severe (n = 7) and end-stage renal disease (ESRD) (n = 9) requiring dialysis and in healthy, age-matched control subjects (n = 8). Elimination was significantly correlated with creatinine clearance. Increases in AUCs of about 42% in mild renal impairment (24-hr CrCl = 50 to 80 mL/min, Cockcroft-Gault [C-G]), about 56% in moderate renal impairment (24-hr CrCl = 30 to 50 mL/min, C-G), about 108% in severe renal impairment (24-hr CrCl ≤30 mL/min, C-G), and about 116% in ESRD subjects were observed, compared with healthy, age-matched control subjects.
The mean terminal half-life (t1/2) was prolonged from 11.1 hours in the control subjects to approximately 13.5, 15.5, 17.6, and 22.8 hours in mild, moderate, severe renal impairment and ESRD subjects, respectively. Less than 5% of the drug in the body was cleared during a standard 4-hour hemodialysis procedure.
The maximum recommended dose in patients with moderate renal impairment is 50 mg per day. Dosage adjustment of 50 mg every other day is recommended in patients with severe renal impairment or ESRD. [see Dosage and Administration (2.2) and Use in Specific Populations (8.6)].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Desvenlafaxine succinate administered by oral gavage to mice and rats for 2 years did not increase the incidence of tumors in either study.
Mice received desvenlafaxine succinate at dosages up to 500/300 mg/kg/day (dosage lowered after 45 weeks of dosing). The 300 mg/kg/day dose is 15 times a human dose of 100 mg per day on a mg/m2 basis.
Rats received desvenlafaxine succinate at dosages up to 300 mg/kg/day (males) or 500 mg/kg/day (females). The highest dose is 29 (males) or 48 (females) times a human dose of 100 mg per day on a mg/m2 basis.
Mutagenesis
Desvenlafaxine was not mutagenic in the in vitro bacterial mutation assay (Ames test) and was not clastogenic in an in vitro chromosome aberration assay in cultured CHO cells, an in vivo mouse micronucleus assay, or an in vivo chromosome aberration assay in rats. Additionally, desvenlafaxine was not genotoxic in the in vitro CHO mammalian cell forward mutation assay and was negative in the in vitro BALB/c-3T3 mouse embryo cell transformation assay.
Impairment of fertility
When desvenlafaxine succinate was administered orally to male and female rats, fertility was reduced at the high dose of 300 mg/kg/day, which is 30 times a human dose of 100 mg per day (on a mg/m2 basis). There was no effect on fertility at 100 mg/kg/day, approximately 10 times a human dose of 100 mg per day (on a mg/m2 basis).
14 CLINICAL STUDIES
The efficacy of PRISTIQ as a treatment for depression was established in four 8-week, randomized, double-blind, placebo-controlled, fixed-dose studies (at doses of 50 mg per day to 400 mg per day) in adult outpatients who met the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) criteria for major depressive disorder. In the first study, patients received 100 mg (n = 114), 200 mg (n = 116), or 400 mg (n = 113) of PRISTIQ once daily, or placebo (n = 118). In a second study, patients received either 200 mg (n = 121) or 400 mg (n = 124) of PRISTIQ once daily, or placebo (n = 124). In two additional studies, patients received 50 mg (n = 150 and n = 164) or 100 mg (n = 147 and n = 158) of PRISTIQ once daily, or placebo (n = 150 and n = 161).
PRISTIQ showed superiority over placebo as measured by improvement in the 17-item Hamilton Rating Scale for Depression (HAM-D17) total score in four studies and overall improvement, as measured by the Clinical Global Impressions Scale - Improvement (CGI-I), in three of the four studies. In studies directly comparing 50 mg per day and 100 mg per day there was no suggestion of a greater effect with the higher dose and adverse reactions and discontinuations were more frequent at higher doses [see Dosage and Administration (2.1)].
| PRISTIQ | ||||||
|---|---|---|---|---|---|---|
| Study No. | Primary Endpoint: HAM-D17 | Placebo | 50 mg/day | 100 mg/day | 200 mg/day | 400 mg/day |
|
1 |
Baseline Score (SD*) |
23.1 (2.5) |
23.2 (2.5) |
22.9 (2.4) |
23.0 (2.2) |
|
|
Difference from Placebo (95% CI †) |
-2.9‡
|
-2.0 |
-3.1‡
|
|||
|
2 |
Baseline Score (SD*) |
25.3 (3.3) |
24.8 (2.9) |
25.2 (3.2) |
||
|
Difference from Placebo (95% CI †) |
-3.3‡
|
-2.8‡
|
||||
|
3 |
Baseline Score (SD*) |
23.0 (2.6) |
23.4 (2.6) |
23.4 (2.6) | ||
|
Difference from Placebo (95% CI †) |
-1.9‡
|
-1.5 | ||||
|
4 |
Baseline Score (SD*) |
24.3 (2.6) |
24.3 (2.4) |
24.4 (2.7) | ||
|
Difference from Placebo (95% CI †) |
-2.5‡
|
-3.0‡
| ||||
Analyses of the relationships between treatment outcome and age and treatment outcome and gender did not suggest any differential responsiveness on the basis of these patient characteristics. There was insufficient information to determine the effect of race on outcome in these studies.
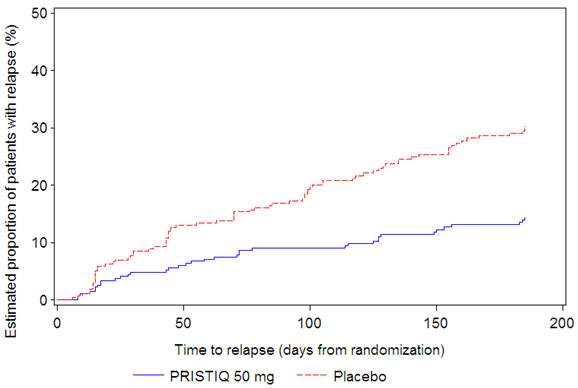
In a longer-term trial (Study 5), adult outpatients meeting DSM-IV criteria for major depressive disorder, who responded to 8 weeks of open-label acute treatment with 50 mg per day desvenlafaxine and subsequently remained stable for 12 weeks on desvenlafaxine, were assigned randomly in a double-blind manner to remain on active treatment or switch to placebo for up to 26 weeks of observation for relapse. Response during the open-label phase was defined as a HAM-D17 total score of ≤ 11 and CGI-I ≤ 2 at the day 56 evaluation; stability was defined as HAM-D17 total score of ≤ 11 and CGI-I ≤ 2 at week 20 and not having a HAM-D17 total score of ≥ 16 or a CGI-I score ≥ 4 at any office visit. Relapse during the double-blind phase was defined as follows: (1) a HAM-D17 total score of ≥ 16 at any office visit, (2) discontinuation for unsatisfactory efficacy response, (3) hospitalized for depression, (4) suicide attempt, or (5) suicide. Patients receiving continued desvenlafaxine treatment experienced statistically significantly longer time to relapse compared with placebo. At 26 weeks, the Kaplan-Meier estimated proportion of relapse was 14% with desvenlafaxine treatment versus 30% with placebo.
|
Figure 4. Estimated Proportion of Relapses vs. Number of Days since Randomization (Study 5) |
 |
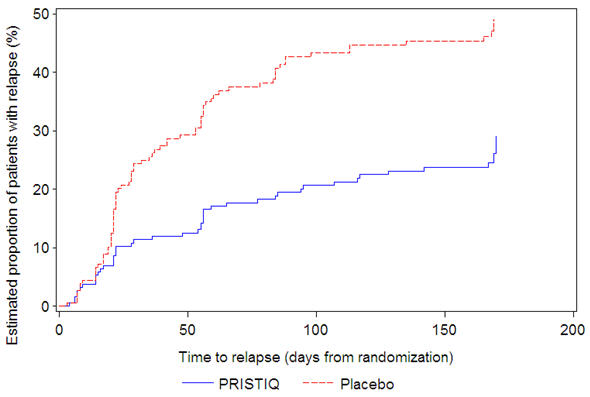
In another longer-term trial (Study 6), adult outpatients meeting DSM-IV criteria for major depressive disorder and who responded to 12 weeks of acute treatment with desvenlafaxine were assigned randomly to the same dose (200 or 400 mg per day) they had received during acute treatment or to placebo for up to 26 weeks of observation for relapse. Response during the open-label phase was defined as a HAM-D17 total score of ≤ 11 at the day 84 evaluation. Relapse during the double-blind phase was defined as follows: (1) a HAM-D17 total score of ≥ 16 at any office visit, (2) a CGI-I score of ≥ 6 (versus day 84) at any office visit, or (3) discontinuation from the trial due to unsatisfactory response. Patients receiving continued desvenlafaxine treatment experienced statistically significantly longer time to relapse over the subsequent 26 weeks compared with those receiving placebo. At 26 weeks, the Kaplan-Meier estimated proportion of relapse was 29% with desvenlafaxine treatment versus 49% with placebo.
|
Figure 5. Estimated Proportion of Relapses vs. Number of Days since Randomization (Study 6) |
 |
In a postmarketing study, the efficacy of PRISTIQ at a dose lower than 50 mg per day was evaluated in an 8-week, multicenter, randomized, double-blind, placebo-controlled, fixed-dose study in adult outpatients with Major Depressive Disorder. The treatment arms were 25 mg (n=232), 50 mg (n=236), and placebo (n=231). The 50 mg dose was superior to placebo, as measured by the mean change from baseline on the HAMD-17. The 25 mg dose was not superior to placebo.
16 HOW SUPPLIED/STORAGE AND HANDLING
PRISTIQ (desvenlafaxine) Extended-Release Tablets are available as follows:
50 mg, light pink, square pyramid tablet debossed with "W" (over) "50" on the flat side
- Available in bottles of approximately 2700 tablets, NDC 55154-4230-8
- Available in bottles of approximately 1350 tablets, NDC 55154-4230-2
100 mg, reddish-orange, square pyramid tablet debossed with "W" (over) "100" on the flat side
- Available in bottles of approximately 2520 tablets, NDC 55154-4235-8
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Each tablet contains 38 mg, 76 mg or 152 mg of desvenlafaxine succinate equivalent to 25 mg, 50 mg or 100 mg of desvenlafaxine, respectively.
The unit-of-use package is intended to be dispensed as a unit.
The appearance of these tablets is a trademark of Wyeth Pharmaceuticals.
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Medication Guide).
Advise patients, their families, and their caregivers about the benefits and risks associated with treatment with PRISTIQ and counsel them in its appropriate use.
Advise patients, their families, and their caregivers to read the Medication Guide and assist them in understanding its contents. The complete text of the Medication Guide is reprinted at the end of this document.
Suicide Risk
Advise patients, their families and caregivers to look for the emergence of suicidality, especially early during treatment and when the dose is adjusted up or down [see Boxed Warning and Warnings and Precautions (5.1)].
Concomitant Medication
Advise patients taking PRISTIQ not to use concomitantly other products containing desvenlafaxine or venlafaxine. Healthcare professionals should instruct patients not to take PRISTIQ with an MAOI or within 14 days of stopping an MAOI and to allow 7 days after stopping PRISTIQ before starting an MAOI [see Contraindications (4)].
Serotonin Syndrome
Caution patients about the risk of serotonin syndrome, particularly with the concomitant use of PRISTIQ with other serotonergic agents (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John's Wort supplements) [see Warnings and Precautions (5.2)].
Elevated Blood Pressure
Advise patients that they should have regular monitoring of blood pressure when taking PRISTIQ [see Warnings and Precautions (5.3)].
Abnormal Bleeding
Patients should be cautioned about the concomitant use of PRISTIQ and NSAIDs, aspirin, warfarin, or other drugs that affect coagulation since combined use of psychotropic drugs that interfere with serotonin reuptake and these agents has been associated with an increased risk of bleeding [see Warnings and Precautions (5.4)].
Angle Closure Glaucoma
Patients should be advised that taking Pristiq can cause mild pupillary dilation, which in susceptible individuals, can lead to an episode of angle closure glaucoma. Pre-existing glaucoma is almost always open-angle glaucoma because angle closure glaucoma, when diagnosed, can be treated definitively with iridectomy. Open-angle glaucoma is not a risk factor for angle closure glaucoma. Patients may wish to be examined to determine whether they are susceptible to angle closure, and have a prophylactic procedure (e.g., iridectomy), if they are susceptible. [see Warnings and Precautions (5.5)]
Activation of Mania/Hypomania
Advise patients, their families and caregivers to observe for signs of activation of mania/hypomania [see Warnings and Precautions (5.6)].
Discontinuation
Advise patients not to stop taking PRISTIQ without talking first with their healthcare professional. Patients should be aware that discontinuation effects may occur when stopping PRISTIQ, and a dose of 25 mg per day is available for discontinuing therapy [see Warnings and Precautions (5.7) and Adverse Reactions (6.1)].
Switching Patients From Other Antidepressants to PRISTIQ
Discontinuation symptoms have been reported when switching patients from other antidepressants, including venlafaxine, to PRISTIQ. Tapering of the initial antidepressant may be necessary to minimize discontinuation symptoms.
Interference with Cognitive and Motor Performance
Caution patients about operating hazardous machinery, including automobiles, until they are reasonably certain that PRISTIQ therapy does not adversely affect their ability to engage in such activities.
Allergic Reactions
Advise patients to notify their physician if they develop allergic phenomena such as rash, hives, swelling, or difficulty breathing.
Pregnancy
Advise patients to notify their physician if they become pregnant or intend to become pregnant during therapy [see Use in Specific Populations (8.1)].
Nursing
Advise patients to notify their physician if they are breastfeeding an infant [see Use in Specific Populations (8.3)].
Residual Inert Matrix Tablet
Patients receiving PRISTIQ may notice an inert matrix tablet passing in the stool or via colostomy. Patients should be informed that the active medication has already been absorbed by the time the patient sees the inert matrix tablet.
Repackaged By:
Cardinal Health
Zanesville, OH 43701
L7756I0916A
L77551016
MEDICATION GUIDE
PRISTIQ® (pris-TEEK)
(desvenlafaxine)
Extended-Release Tablets
Read this Medication Guide before you start taking PRISTIQ and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
Antidepressant Medicines, Depression and Other Serious Mental Illnesses, and Suicidal Thoughts or Actions
Talk to your healthcare provider about:
- •
- all risks and benefits of treatment with antidepressant medicines
- •
- all treatment choices for depression or other serious mental illness
What is the most important information I should know about antidepressant medicines, depression and other serious mental illnesses, and suicidal thoughts or actions?
- 1.
- Antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, and young adults within the first few months of treatment.
- 2.
- Depression and other serious mental illnesses are the most important causes of suicidal thoughts and actions. Some people may have a particularly high risk of having suicidal thoughts or actions. These include people who have (or have a family history of) bipolar illness (also called manic-depressive illness) or suicidal thoughts or actions.
- 3.
-
How can I watch for and try to prevent suicidal thoughts and actions?
- •
- Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings. This is very important when an antidepressant medicine is started or when the dose is changed.
- •
- Call the healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings.
- •
- Keep all follow-up visits with the healthcare provider as scheduled. Call the healthcare provider between visits as needed, especially if you have concerns about symptoms.
Call a healthcare provider right away if you have any of the following symptoms, especially if they are new, worse, or worry you:
|
|
What else do I need to know about antidepressant medicines?
- •
- Never stop an antidepressant medicine without first talking to a healthcare provider. Stopping an antidepressant medicine suddenly can cause other symptoms.
- •
- Antidepressants are medicines used to treat depression and other illnesses. It is important to discuss all the risks of treating depression and also the risks of not treating it. Patients should discuss all treatment choices with the healthcare provider, not just the use of antidepressants.
- •
- Antidepressant medicines have other side effects. Talk to the healthcare provider about the side effects of this medicine.
- •
- Antidepressant medicines can interact with other medicines. Know all of the medicines that you take. Keep a list of all medicines to show the healthcare provider. Do not start new medicines without first checking with your healthcare provider.
- •
- Not all antidepressant medicines prescribed for children are FDA approved for use in children. Talk to your child's healthcare provider for more information.
Important Information about PRISTIQ Extended-Release Tablets
Read the patient information that comes with PRISTIQ before you take PRISTIQ and each time you refill your prescription. There may be new information. If you have questions, ask your healthcare provider. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
What is PRISTIQ?
- •
- PRISTIQ is a prescription medicine used to treat depression. PRISTIQ belongs to a class of medicines known as SNRIs (or serotonin-norepinephrine reuptake inhibitors).
Who should not take PRISTIQ?
Do not take PRISTIQ if you:
- •
- are allergic to desvenlafaxine, venlafaxine or any of the ingredients in PRISTIQ. See the end of this Medication Guide for a complete list of ingredients in PRISTIQ.
- •
- take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid and the intravenous medicine methylene blue.
- •
- have taken an MAOI within 7 days of stopping PRISTIQ unless directed by your healthcare provider.
- •
- have started PRISTIQ and if you stopped taking an MAOI in the last 14 days unless directed by your healthcare provider.
What should I tell my healthcare provider before taking PRISTIQ?
Tell your healthcare provider about all your medical conditions, including if you:
- •
- have high blood pressure
- •
- have heart problems
- •
- have high cholesterol or high triglycerides
- •
- have a history of a stroke
- •
- have or had depression, suicidal thoughts or behavior
- •
- have kidney problems
- •
- have liver problems
- •
- have or had bleeding problems
- •
- have or had seizures or convulsions
- •
- have mania or bipolar disorder
- •
- have low sodium levels in your blood
- •
- are pregnant or plan to become pregnant. It is not known if PRISTIQ will harm your unborn baby.
- •
- are breastfeeding. PRISTIQ can pass into your breast milk and may harm your baby. Talk with your healthcare provider about the best way to feed your baby if you take PRISTIQ.
Serotonin syndrome
Rare, but potentially life-threatening conditions called serotonin syndrome can happen when medicines such as PRISTIQ are taken with certain other medicines. Serotonin syndrome can cause serious changes in how your brain, muscles, heart and blood vessels, and digestive system work. Especially tell your healthcare provider if you take the following:
- •
- medicines to treat migraine headaches known as triptans
- •
- medicines used to treat mood, anxiety, psychotic, or thought disorders, including tricyclics, lithium, selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), or other dopamine antagonists, such as metoclopramide
- •
- silbutramine
- •
- tramadol
- •
- St. John's Wort
- •
- MAOIs (including linezolid, an antibiotic and intravenous methylene blue)
- •
- tryptophan supplements
Ask your healthcare provider if you are not sure if you are taking any of these medicines.
Before you take PRISTIQ with any of these medicines, talk to your healthcare provider about serotonin syndrome. See "What are the possible side effects of PRISTIQ?"
Do not take PRISTIQ with other medicines containing venlafaxine or desvenlafaxine.
How should I take PRISTIQ?
- •
- Take PRISTIQ exactly as your healthcare provider has told you.
- •
- Take PRISTIQ at about the same time each day.
- •
- PRISTIQ may be taken either with or without food.
- •
- Swallow PRISTIQ tablets whole, with fluid. Do not crush, cut, chew, or dissolve PRISTIQ tablets because the tablets are time-released.
- •
- When you take PRISTIQ, you may see something in your stool that looks like a tablet. This is the empty shell from the tablet after the medicine has been absorbed by your body.
- •
- It is common for antidepressant medicines such as PRISTIQ to take several weeks before you start to feel better. Do not stop taking PRISTIQ if you do not feel results right away.
- •
- Do not stop taking or change the dose of PRISTIQ without talking with your healthcare provider, even if you feel better.
- •
- Talk with your healthcare provider about how long you should use PRISTIQ. Take PRISTIQ for as long as your healthcare provider tells you to.
- •
- If you miss a dose of PRISTIQ, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Do not try to "make up" for the missed dose by taking two doses at the same time.
- •
- Do not take more PRISTIQ than prescribed by your healthcare provider. If you take more PRISTIQ than the amount prescribed, contact your healthcare provider right away.
- •
- If you take too much PRISTIQ, call the Poison Control Center at 1-800-222-1222 or go to the nearest hospital emergency room right away.
Switching from other antidepressants
Side effects from discontinuing antidepressant medication have occurred when patients switched from other antidepressants, including venlafaxine, to PRISTIQ. Your doctor may gradually reduce the dose of your initial antidepressant medication to help to reduce these side effects.
What should I avoid while taking PRISTIQ?
- •
- Do not drive a car or operate machinery until you know how PRISTIQ affects you.
- •
- Avoid drinking alcohol while taking PRISTIQ.
What are the possible side effects of PRISTIQ?
PRISTIQ can cause serious side effects, including:
- •
- See the beginning of this Medication Guide -Antidepressant Medicines, Depression and other Serious Mental Illnesses, and Suicidal Thoughts or Actions.
- •
-
Serotonin syndrome. See "What should I tell my healthcare provider before taking PRISTIQ?"
Get medical help right away if you think that you have these syndromes. Signs and symptoms of these syndromes may include one or more of the following:
|
|
PRISTIQ may also cause other serious side effects, including:
- •
- New or worsened high blood pressure (hypertension). Your healthcare provider should monitor your blood pressure before and while you are taking PRISTIQ. If you have high blood pressure, it should be controlled before you start taking PRISTIQ.
- •
- Abnormal bleeding or bruising. PRISTIQ and other SNRIs/SSRIs may cause you to have an increased chance of bleeding. Taking aspirin, NSAIDs (non-steroidal anti-inflammatory drugs), or blood thinners may add to this risk. Tell your healthcare provider right away about any unusual bleeding or bruising.
- •
-
Visual problemsOnly some people are at risk for these problems. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are.
Symptoms when stopping PRISTIQ (discontinuation symptoms). Side effects may occur when stopping PRISTIQ (discontinuation symptoms), especially when therapy is stopped suddenly. Your healthcare provider may want to decrease your dose slowly to help avoid side effects. Some of these side effects may include:- •
- eye pain
- •
- changes in vision
- •
- swelling or redness in or around the eye
|
|
- •
- Seizures (convulsions)
- •
- Low sodium levels in your blood. Symptoms of this may include: headache, difficulty concentrating, memory changes, confusion, weakness and unsteadiness on your feet. In severe or more sudden cases, symptoms can include: hallucinations (seeing or hearing things that are not real), fainting, seizures and coma. If not treated, severe low sodium levels could be fatal.
- •
- Lung problems. Some people who have taken the medicine venlafaxine which is the same kind of medicine as the medicine in PRISTIQ have had lung problems. Symptoms of lung problems include difficulty breathing, cough, or chest discomfort. Tell your healthcare provider right away if you have any of these symptoms.
Common side effects with PRISTIQ include:
|
|
These are not all the possible side effects of PRISTIQ. Tell your healthcare provider about any side effect that bothers you or does not go away.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store PRISTIQ?
- •
- Store PRISTIQ at 68°F to 77°F (20°C to 25°C).
- •
- Do not use PRISTIQ after the expiration date (EXP), which is on the container. The expiration date refers to the last day of that month.
- •
- Keep PRISTIQ and all medicines out of the reach of children.
General Information about the safe and effective use of PRISTIQ
Medicines are sometimes used for conditions that are not mentioned in Medication Guides. Do not use PRISTIQ for a condition for which it was not prescribed. Do not give PRISTIQ to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about PRISTIQ. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about PRISTIQ that is written for healthcare professionals.
For more information, go to www.pristiq.com or call 1-888-PRISTIQ (774-7847).
What are the ingredients in PRISTIQ?
Active ingredient: desvenlafaxine
Inactive ingredients: For the 25 mg tablet, hypromellose, microcrystalline cellulose, talc, magnesium stearate, a film coating which consists of polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, and iron oxides.
For the 50 mg tablet, hypromellose, microcrystalline cellulose, talc, magnesium stearate and film coating, which consists of polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, and iron oxides.
For the 100 mg tablet, hypromellose, microcrystalline cellulose, talc, magnesium stearate, a film coating which consists of polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, iron oxide and FD&C yellow #6.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
LAB-0539-7
Repackaged By:
Cardinal Health
Zanesville, OH 43701
L7756I0916A
L77551016
August 2014
| PRISTIQ
EXTENDED-RELEASE
desvenlafaxine succinate tablet, extended release |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| PRISTIQ
EXTENDED-RELEASE
desvenlafaxine succinate tablet, extended release |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Cardinal Health (188557102) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Cardinal Health | 188557102 | REPACK(55154-4230, 55154-4235) | |