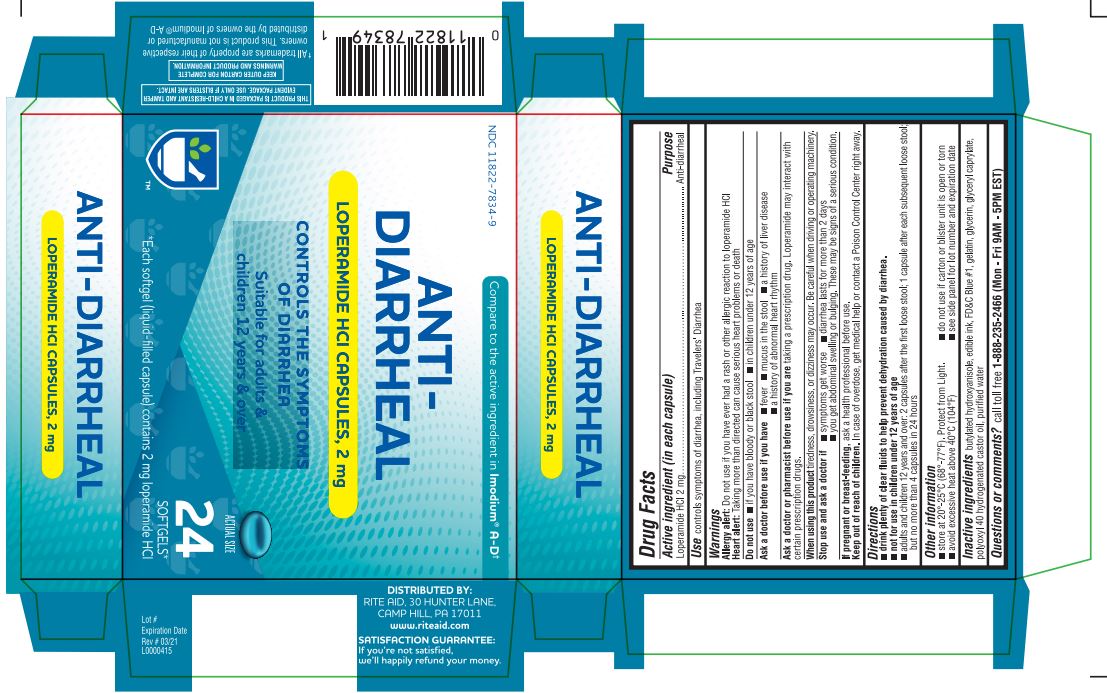
Label: ANTI-DIARRHEAL- loperamide hydrochloride capsule, liquid filled
- NDC Code(s): 11822-7834-2, 11822-7834-9
- Packager: Rite Aid Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 24, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each capsule)
- Purpose
- Use
- Warnings
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
SPL UNCLASSIFIED SECTION
THIS PRODUCT IS PACKAGED IN A CHILD-RESISTANT AND TAMPER EVIDENT PACKAGE. USE ONLY IF BLISTERS ARE INTACT.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
†All trademarks are property of their respective owners. This product is not manufactured or distributed by the owners of Imodium ® A-D
DISTRIBUTED BY:
RITE AID, 30 HUNTER LANE,
CAMP HILL, PA 17011
www.riteaid.com
SATISFACTION GUARANTEE:
If you're not satisfied, we'll happily refund your money.
Lot #
Expiration Date
Rev # 03/21
L0000415
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ANTI-DIARRHEAL
loperamide hydrochloride capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11822-7834 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 2 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) WATER (UNII: 059QF0KO0R) Product Characteristics Color blue Score no score Shape CAPSULE Size 10mm Flavor Imprint Code LP2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11822-7834-2 1 in 1 CARTON 01/24/2020 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:11822-7834-9 2 in 1 CARTON 01/24/2020 2 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021855 01/24/2020 Labeler - Rite Aid Corporation (014578892) Registrant - Bionpharma Inc. (079637826) Establishment Name Address ID/FEI Business Operations Patheon Softgels Inc. 002193829 manufacture(11822-7834)