COMBI-PEN-48- penicillin g benzathine and penicillin g procaine injection, suspension
Bimeda, Inc.
----------
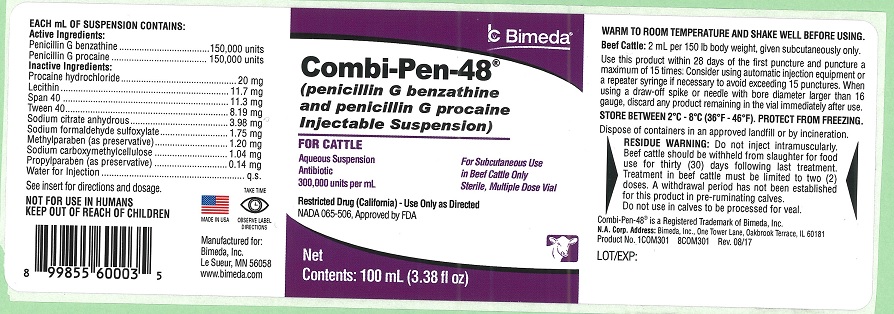
Combi-Pen-48®
(penicillin G benzathine and penicillin G procaine Injectable Suspension)
FOR CATTLE
Aqueous Suspension For Subcutaneous use in Beef Cattle Only
Antibiotic Sterile, Multiple Dose Vial
300,000 units per mL
Restricted Drug (California) - Use Only as Directed
NADA 065-506, Approved by FDA
Net Contents: 100 mL
Each mL of suspension contains:
Active Ingredients:
Penicillin G benzathine................150,000 units
Penicillin G procaine....................150,000 units
Inactive Ingredients:
Procaine hydrochloride............................20 mg
Lecithin..............................................11.7 mg
Span 40.............................................11.3 mg
Tween 40...........................................8.19 mg
Sodium citrate anhydrous.....................3.98 mg
Sodium formaldehyde sulfoxylate..........1.75 mg
Methylparaben (as preservative)...........1.20 mg
Sodium carboxymethylcellulose............1.04 mg
Propylparaben (as preservative)...........0.14 mg
Water for Injection....................................q.s.
See Insert for directions and dosage.
NOT FOR USE IN HUMANS
KEEP OUT OF REACH OF CHILDREN
WARM TO ROOM TEMPERATURE AND SHAKE WELL BEFORE USING
Beef Cattle: 2 mL per 150 lb. body weight, given subcutaneously only.
DESCRIPTION: Penicillin G Benzathine and Penicillin G Procaine Injectable Suspension is available as an aqueous suspension in 100-mL, 250-mL, and 500-mL multiple-dose vials. Each mL contains: Active Ingredients: 150,000 units penicillin G benzathine, 150,000 units penicillin G procaine. Inactive Ingredients: 11.7 mg lecithin, 1.75 mg sodium formaldehyde sulfoxylate, 1.20 mg methylparaben, 0.14 mg propylparaben, 8.19 mg Tween 40, 11.3 mg Span 40, 3.98 mg sodium citrate (anhydrous), 20 mg procaine hydrochloride, 1.04 mg sodium carboxymethylcellulose, Water for Injection, q.s.
ACTION: Penicillin G is an antibiotic which shows a marked bactericidal effect against certain organisms during their growth phase. It is relatively specific in its action against gram-positive bacteria but is usually ineffective against gram-negative organisms.
It is normally recommended that any bacterial infection be treated as early as possible and with a dosage that will give effective blood levels. Although the recommended dosage of this product will give longer detectable penicillin blood levels than penicillin G procaine alone, it is recommended that a second dose be administered at 48 hours when treating a penicillin-susceptible bacterial infection.
The use of antibiotics in the management of disease is based on an accurate diagnosis and an adequate course of treatment. When properly used in treatment of diseases caused be penicillin-susceptible organisms, most animals treated with the product will show a noticeable improvement within 24 to 48 hours. If improvement does not occur within this period of time, the diagnosis and course of treatment should be re-evaluated. It is recommended that the diagnosis and treatment of animal diseases be carried out by a veterinarian. Since many diseases look alike but require different types of treatment, the use of professional veterinary and laboratory services can reduce treatment time, costs and needless losses. Good housing, sanitation and nutrition are important in the maintenance of healthy animals and are essential in the treatment of disease.
RESIDUE WARNING: Beef cattle should be withheld from slaughter for food use for 30 days following the last treatment. Treatment in beef cattle must be limited to two (2) doses, by subcutaneous injection only. Do not inject intramuscularly.
A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in calves to be processed for veal.
INDICATIONS: This product is indicated for the treatment of the following bacterial infections in beef cattle due to penicillin-susceptible microorganisms that are susceptible to the serum levels common to this particular dosage form, such as:
1. Bacterial Pneumonia (shipping fever complex) (Streptococcus spp., Actinomyces pyogenes, Staphylococcus aureus).
2. Upper Respiratory Infections such as rhinitis or pharyngitis (Actinomyces pyogenes).
3. Blackleg (Clostridium chauvoei).
PRECAUTIONS: Exceeding the recommended doses and dosage levels may result in antibiotic residues beyond the withdrawal time. Do not inject this product intramuscularly.
Penicillin G is a substance of low toxicity. However, side effects, or so-called allergic or anaphylactic reactions - sometimes fatal, have been known to occur in animals hypersensitive to penicillin and procaine. Such reactions can occur unpredictably with varying intensity. Animals administered penicillin G should be kept under close observation for at least one-half hour. Should allergic or anaphylactic reactions occur, discontinue use of the product and immediately administer epinephrine following the manufacturer's recommendation and call a veterinarian.
As with all antibiotic preparations, use of this drug may result in overgrowth of non-susceptible organisms, including fungi. A lack of response by the treated animal, or the development of the new signs or symptoms suggests that an overgrowth of non-susceptible organisms has occurred. In such instances, consult a veterinarian.
Since bacterial drugs may interfere with the bacteriostatic action of tetracyclines, it is advisable to avoid giving penicillin in conjunction with tetracyclines.
ADMINISTRATION: The recommended dosage for beef cattle should be administered by subcutaneous injection only. Failure to use the subcutaneous route of administration may result in antibiotic residues in meat beyond the withdrawal time.
DOSAGE: Beef cattle: 2 mL per 150 lb body weight given subcutaneously only (2000 units penicillin G procaine and 2000 units penicillin G benzathine per lb body weight). Treatment should be repeated in 48 hours.
IMPORTANT: Treatment in beef cattle should be limited to two (2) doses given by subcutaneous injection only.
DIRECTIONS FOR USE: A thoroughly cleaned, sterile needle and syringe should be used for each injection (needles and syringes may be sterilized by boiling in water for 15 minutes).
Before withdrawing the solution from the bottle, disinfect the the rubber cap on the bottle with a suitable disinfectant, such as 70 percent alcohol. The injection site should be similarly cleaned with the disinfectant. Use a 16 gauge needle, not more than 1 inch long. Use this product within 28 days of the first puncture and puncture a maximum of 15 times. Consider using automatic injection equipment or a repeater syringe if necessary to avoid exceeding 15 punctures. When using a draw-off spike or needle with bore diameter larger than 16 gauge, discard any product remaining in the vial immediately after use.
A subcutaneous injection should be made by pinching up a fold of the skin between the thumb and forefinger. The mid-neck region is the preferred injection site. Insert the needle under the fold in a direction approximately parallel to the surface of the body. When the needle is inserted in this manner, the medication will be delivered underneath the skin between the skin and the muscles. Proper restraint, such as the use of a chute and nose lead is needed for proper administration of the product.
STORE BETWEEN 2°C - 8°C (36°F- 46°F). PROTECT FROM FREEZING.
WARM TO ROOM TEMPERATURE AND SHAKE WELL BEFORE USING.
| COMBI-PEN-48
penicillin g benzathine and penicillin g procaine injection, suspension |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Bimeda, Inc. (060492923) |
| Registrant - Bimeda, Inc. (060492923) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Constant Irwindale | 010569463 | manufacture | |