CHLORTHALIDONE- chlorthalidone tablet
Bryant Ranch Prepack
----------
Chlorthalidone Tablets, USP
VistaPharm, Inc.
DESCRIPTION
Chlorthalidone is an oral antihypertensive/diuretic. It is a monosulfamyl diuretic that differs chemically from thiazide diuretics in that a double-ring system is incorporated in its structure. It is 2-Chloro-5(1-hydroxy- 3-oxo-1- isoindolinyl) benzenesulfonamide with the following structural formula:
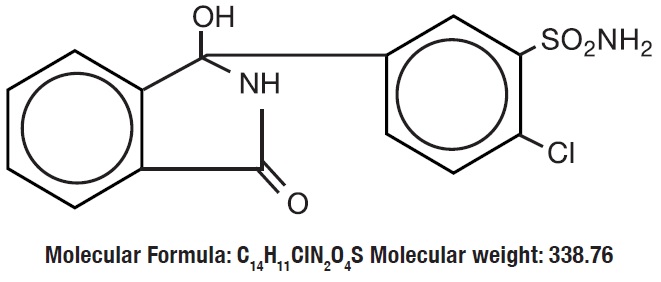
Chlorthalidone, USP is practically insoluble in water, in ether, and in chloroform; soluble in methanol; slightly soluble in ethanol.
Chlorthalidone tablets are available containing either 25 mg or 50 mg of chlorthalidone, USP and the following inactive ingredients: colloidal silicon dioxide, microcrystalline cellulose, pregelatinized starch (corn), sodium starch glycolate, stearic acid.
CLINICAL PHARMACOLOGY
Chlorthalidone is an oral diuretic with prolonged action (48–72 hours) and low toxicity. The major portion of the drug is excreted unchanged by the kidneys. The diuretic effect of the drug occurs in approximately 2.6 hours and continues for up to 72 hours. The mean half-life following a 50 to 200 mg dose is 40 hours. In the first order of absorption, the elimination half-life is 53 hours following a 50 mg dose, and 60 hours following a 100 mg dose. Approximately 75 percent of the drug is bound to plasma proteins, 58 percent of the drug being bound to albumin. This is caused by an increased affinity of the drug to erythrocyte carbonic anhydrase. Nonrenal routes of elimination have yet to be clarified. Data are not available regarding percentage of dose as unchanged drug and metabolites, concentration of the drug in body fluids, degree of uptake by a particular organ or in the fetus, or passage across the blood-brain barrier.
The drug produces copious diuresis with greatly increased excretion of sodium and chloride. At maximal therapeutic dosage, chlorthalidone is approximately equal in its diuretic effect to comparable maximal therapeutic doses of benzothiadiazine diuretics. The site of action appears to be the cortical diluting segment of the ascending limb of Henle’s loop of the nephron.
INDICATIONS AND USAGE
Diuretics such as chlorthalidone are indicated in the management of hypertension either as the sole therapeutic agent or to enhance the effect of other antihypertensive drugs in the more severe forms of hypertension.
Chlorthalidone is indicated as adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
Chlorthalidone has also been found useful in edema due to various forms of renal dysfunction, such as nephrotic syndrome, acute glomerulonephritis, and chronic renal failure.
Usage in Pregnancy
The routine use of diuretics in an otherwise healthy woman is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy, and there is no satisfactory evidence that they are useful in the treatment of developed toxemia. Edema during pregnancy may arise from pathologic causes or from the physiologic and mechanical consequences of pregnancy. Chlorthalidone is indicated in pregnancy when edema is due to pathologic causes, just as it is in the absence of pregnancy (however, see PRECAUTIONS, below). Dependent edema in pregnancy, resulting from restriction of venous return by the expanded uterus, is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy that is harmful to neither the fetus nor the mother (in the absence of cardiovascular disease), but that is associated with edema, including generalized edema, in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances, this edema may cause extreme discomfort that is not relieved by rest. In these cases, a short course of diuretics may provide relief and be appropriate.
CONTRAINDICATIONS
Anuria.
Known hypersensitivity to chlorthalidone or other sulfonamide-derived drugs.
WARNINGS
Chlorthalidone should be used with caution in severe renal disease. In patients with renal disease, chlorthalidone or related drugs may precipitate azotemia. Cumulative effects of the drug may develop in patients with impaired renal function.
Chlorthalidone should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor alterations of fluid and electrolyte balance may precipitate hepatic coma. Sensitivity reactions may occur in patients with a history of allergy or bronchial asthma.
The possibility of exacerbation or activation of systemic lupus erythematosus has been reported with thiazide diuretics, which are structurally related to chlorthalidone. However, systemic lupus erythematosus has not been reported following chlorthalidone administration.
PRECAUTIONS
General
Hypokalemia may develop with chlorthalidone as with any other diuretic, especially with brisk diuresis when severe cirrhosis is present or during concomitant use of corticosteroids or ACTH.
Interference with adequate oral electrolyte intake will also contribute to hypokalemia. Digitalis therapy may exaggerate metabolic effects of hypokalemia especially with reference to myocardial activity.
Any chloride deficit is generally mild and usually does not require specific treatment except under extraordinary circumstances (as in liver disease or renal disease). Dilutional hyponatremia may occur in edematous patients in hot weather, appropriate therapy is water restriction, rather than administration of salt except in rare instances when the hyponatremia is life threatening. In actual salt depletion, appropriate replacement is the therapy of choice.
Hyperuricemia may occur or frank gout may be precipitated in certain patients receiving chlorthalidone. Thiazide-like diuretics have been shown to increase the urinary excretion of magnesium; this may result in hypomagnesemia.
The antihypertensive effects of the drug may be enhanced in the post-sympathectomy patient. If progressive renal impairment becomes evident, as indicated by a rising nonprotein nitrogen or blood urea nitrogen, a careful reappraisal of therapy is necessary with consideration given to withholding or discontinuing diuretic therapy.
Calcium excretion is decreased by thiazide-like drugs. Pathological changes in the parathyroid gland with hypercalcemia and hypophosphatemia have been observed in few patients on thiazide therapy. The common complications of hyperparathyroidism such as renal lithiasis, bone resorption and peptic ulceration have not been seen.
Information for Patients
Patients should inform their physician if they have: (1) had an allergic reaction to chlorthalidone or other diuretics or have asthma, (2) kidney disease, (3) liver disease, (4) gout, (5) systemic lupus erythematosus, or (6) been taking other drugs such as cortisone, digitalis, lithium carbonate, or drugs for diabetes.
Patients should be cautioned to contact their physician if they experience any of the following symptoms of potassium loss: excess thirst, tiredness, drowsiness, restlessness, muscle pains or cramps, nausea, vomiting, or increased heart rate or pulse.
Patients should also be cautioned that taking alcohol can increase the chance of dizziness occurring.
Laboratory Tests
Periodic determination of serum electrolytes to detect possible electrolyte imbalance should be performed at appropriate intervals.
All patients receiving chlorthalidone should be observed for clinical signs of fluid or electrolyte imbalance: namely, hyponatremia, hypochloremic alkalosis, and hypokalemia. Serum and urine electrolyte determinations are particularly important when the patient is vomiting excessively or receiving parenteral fluids.
Drug Interactions
Chlorthalidone may add to or potentiate the action of other antihypertensive drugs. Potentiation occurs with ganglionic peripheral adrenergic blocking drugs.
Medication such as digitalis may also influence serum electrolytes. Warning signs, irrespective of cause, are: dryness of mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances such as nausea and vomiting.
Insulin requirements in diabetic patients may be increased, decreased, or unchanged. Higher dosage of oral hypoglycemic agents may be required. Latent diabetes mellitus may become manifest during chlorthalidone administration.
Chlorthalidone and related drugs may increase the responsiveness to tubocurarine.
Chlorthalidone and related drugs may decrease arterial responsiveness to norepinephrine. This diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use.
Drug/Laboratory Test Interactions
Chlorthalidone and related drugs may decrease serum PBI levels without signs of thyroid disturbance.
Pregnancy
Teratogenic Effects. Pregnancy Category B
Reproduction studies have been performed in the rat and the rabbit at doses up to 420 times the human dose and have revealed no evidence of harm to the fetus due to chlorthalidone. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nonteratogenic Effects
Thiazides cross the placental barrier and appear in cord blood. The use of chlorthalidone and related drugs in pregnant women requires that the anticipated benefits of the drug be weighed against possible hazards to the fetus. These hazards include fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in the adult.
ADVERSE REACTIONS
The following adverse reactions have been observed, but there is not enough systematic collection of data to support an estimate of their frequency.
Gastrointestinal System Reactions: anorexia, gastric irritation, nausea, vomiting, cramping, diarrhea, constipation, jaundice (intrahepatic cholestatic jaundice), pancreatitis.
Central Nervous System Reactions: dizziness, vertigo, paresthesias, headache, xanthopsia.
Hematologic Reactions: leukopenia, agranulocytosis, thrombocytopenia, aplastic anemia.
Dermatologic-Hypersensitivity Reactions: purpura, photosensitivity, rash, urticaria, necrotizing angiitis (vasculitis, cutaneous vasculitis), Lyell’s syndrome (toxic epidermal necrolysis).
Cardiovascular Reactions: orthostatic hypotension may occur and may be aggravated by alcohol, barbiturates, or narcotics.
Other Adverse Reactions: hyperglycemia, glycosuria, hyperuricemia, muscle spasm, weakness, restlessness, impotence.
Whenever adverse reactions are moderate or severe, chlorthalidone dosage should be reduced or therapy withdrawn.
OVERDOSAGE
Symptoms of acute overdosage include nausea, weakness, dizziness, and disturbances of electrolyte balance. The oral LD50 of the drug in the mouse and the rat is more than 25,000 mg/kg body weight. The minimum lethal dose (MLD) in humans has not been established. There is no specific antidote, but gastric lavage is recommended, followed by supportive treatment. Where necessary, this may include intravenous dextrose-saline with potassium, administered with caution.
DOSAGE AND ADMINISTRATION
Therapy should be initiated with the lowest possible dose. This dose should be titrated according to individual patient response to gain maximal therapeutic benefit while maintaining lowest dosage possible. A single dose given in the morning with food is recommended; divided daily doses are unnecessary.
Hypertension
Initiation: Therapy, in most patients, should be initiated with a single daily dose of 25 mg. If the response is insufficient after a suitable trial, the dosage may be increased to a single daily dose of 50 mg. If additional control is required, the dosage of chlorthalidone may be increased to 100 mg once daily or a second antihypertensive drug (step 2 therapy) may be added. Dosage above 100 mg daily usually does not increase effectiveness. Increases in serum uric acid and decreases in serum potassium are dose-related over the 25 to 100 mg/day range.
Maintenance: Maintenance doses may be lower than initial doses and should be adjusted according to individual patient response. Effectiveness is well sustained during continued use.
Edema
Initiation: Adults, initially 50 to 100 mg daily, or 100 mg on alternate days. Some patients may require 150 to 200 mg at these intervals or up to 200 mg daily. Dosages above this level, however, do not usually produce a greater response.
Maintenance: Maintenance doses may often be lower than initial doses and should be adjusted according to individual patient response. Effectiveness is well sustained during continued use.
| CHLORTHALIDONE
chlorthalidone tablet |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Bryant Ranch Prepack (171714327) |
| Registrant - Bryant Ranch Prepack (171714327) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bryant Ranch Prepack | 171714327 | REPACK(71335-1708) , RELABEL(71335-1708) | |
