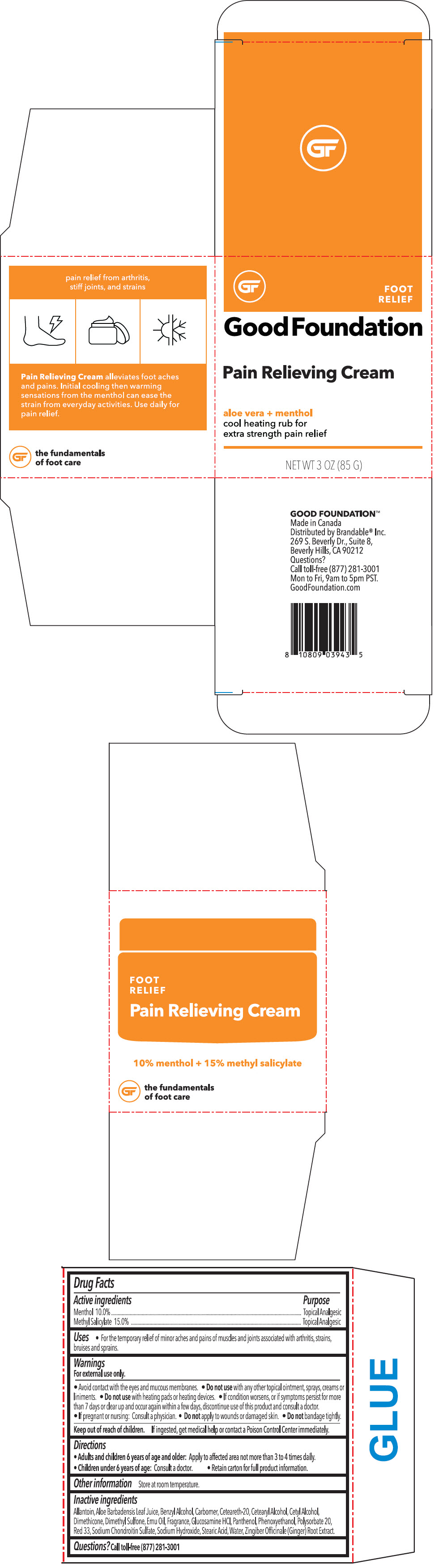
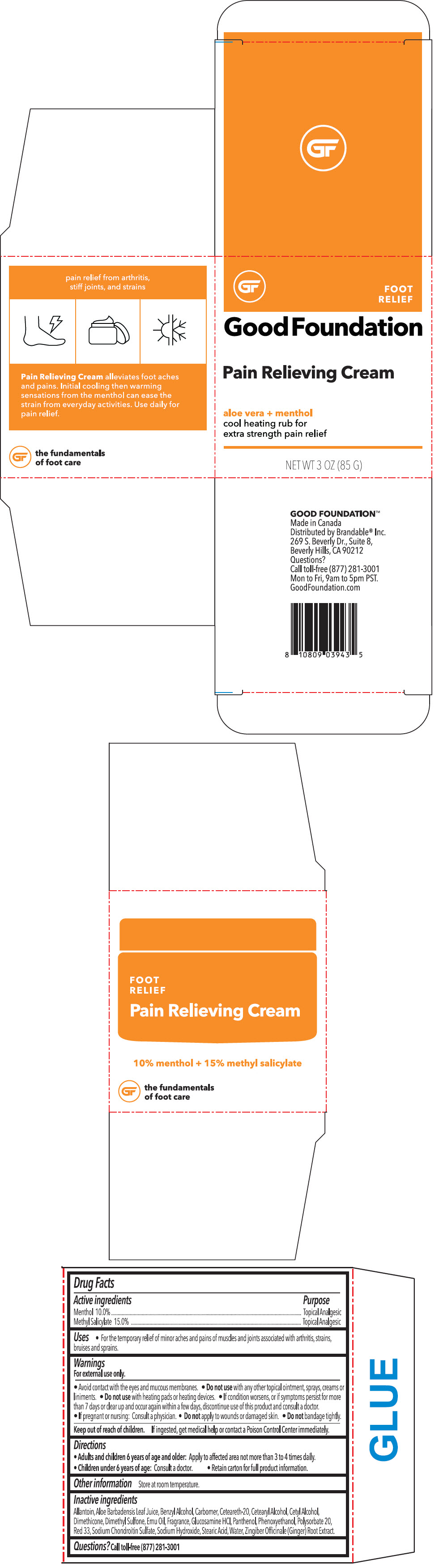
Label: PAIN RELIEVING FOOT- menthyl salicylate and menthol, unspecified form cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 72764-004-01 - Packager: Brandable
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 24, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
For external use only.
- Avoid contact with the eyes and mucous membranes.
- Do not use with any other topical ointment, sprays, creams or liniments.
- Do not use with heating pads or heating devices.
- Directions
- Other information
-
Inactive ingredients
Allantoin, Aloe Barbadensis Leaf Juice, Benzyl Alcohol, Carbomer, Ceteareth-20, Cetearyl Alcohol, Cetyl Alcohol, Dimethicone, Dimethyl Sulfone, Emu Oil, Fragrance, Glucosamine HCl, Panthenol, Phenoxyethanol, Polysorbate 20, Red 33, Sodium Chondroitin Sulfate, Sodium Hydroxide, Stearic Acid, Water, Zingiber Officinale (Ginger) Root Extract.
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 85 G Container Box
-
INGREDIENTS AND APPEARANCE
PAIN RELIEVING FOOT
menthyl salicylate and menthol, unspecified form creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72764-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHYL SALICYLATE, (+/-)- (UNII: 43XOA705ZD) (Salicylic Acid - UNII:O414PZ4LPZ) MENTHYL SALICYLATE, (+/-)- 750 mg in 5 g MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 500 mg in 5 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) Stearic Acid (UNII: 4ELV7Z65AP) Cetyl Alcohol (UNII: 936JST6JCN) Emu Oil (UNII: 344821WD61) Dimethyl Sulfone (UNII: 9H4PO4Z4FT) Dimethicone (UNII: 92RU3N3Y1O) ALOE VERA LEAF (UNII: ZY81Z83H0X) Allantoin (UNII: 344S277G0Z) SODIUM CHONDROITIN SULFATE (PORCINE; 5500 MW) (UNII: H5BJH23Z9A) GLUCOSAMINE HYDROCHLORIDE (UNII: 750W5330FY) GINGER (UNII: C5529G5JPQ) Panthenol (UNII: WV9CM0O67Z) Polysorbate 20 (UNII: 7T1F30V5YH) CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Phenoxyethanol (UNII: HIE492ZZ3T) Benzyl Alcohol (UNII: LKG8494WBH) Sodium Hydroxide (UNII: 55X04QC32I) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72764-004-01 1 in 1 BOX 04/01/2020 1 85 g in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part348 04/01/2020 Labeler - Brandable (080414109) Registrant - Garcoa Laboratories (036464697) Establishment Name Address ID/FEI Business Operations Sigan Industries 255106239 MANUFACTURE(72764-004)