Label: ACID REDUCER- ranitidine tablet, coated
- NDC Code(s): 69842-869-62
- Packager: CVS HEALTH CORP
- This is a repackaged label.
- Source NDC Code(s): 55111-404
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated September 30, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Use(s)
-
Warnings
Allergy alert: Do not use if you are allergic to ranitidine or other acid reducers
Do not use
- if you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools.These may be signs of a serious condition. See your doctor.
- with other acid reducers
- if you have kidney disease, except under the advice and supervision of a doctor
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
-
Directions
- adults and children 12 years and over:
- to relieve symptoms, swallow 1 tablet with a glass of water
- to prevent symptoms, swallow 1 tablet with a glass of water 30 to 60 minutes before eating food or drinking beverages that cause heartburn
- can be used up to twice daily (do not take more than 2 tablets in 24 hours)
- children under 12 years: ask a doctor
- adults and children 12 years and over:
- Other information
- Inactive ingredients
- Questions
-
Consumer Information
What you should know about
MAXIMUM STRENGTH
Ranitidine Tablets USP, 150 mg
(Please read all of this information before taking MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg. Save this leaflet for future reference.)
What are MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg?
- MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg contains 150 mg of ranitidine (as ranitidine hydrochloride USP, 168 mg), a medicine that doctors have prescribed more than 200 million times worldwide.
Excellent Safety Record
- The ingredient in MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg, ranitidine, has been prescribed by doctors for years to treat millions of patients safely and effectively. The active ingredient in MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg has been taken safely with many frequently prescribed medications.
- MAXIMUM STRENGTH Ranitidne Tablets USP, 150 mg are sodium and sugar free.
What symptoms does MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg relieve and prevent?
- MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg relieve and prevent heartburn associated with acid Indigestion and sour stomach. Eating or drinking certain foods or beverages, and even lying down to sleep, can cause heartburn associated with acid indigestion and sour stomach. It is normal to the stomach to produce acid, especially after consuming food or beverages. However, acid in the wrong place, such as the esophagus, or too much acid, can cause burning pain and discomfort.
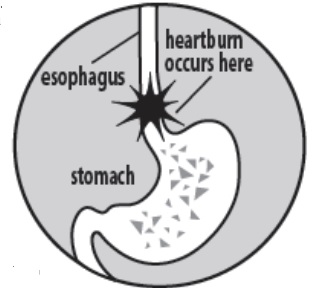
How should I take MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg?
-
To relieve symptoms, swallow 1 tablet with a glass of water.
-
To prevent symptoms, swallow 1 tablet with a glass of water 30 to 60 minutes before eating food or drinking beverages that cause heartburn. This medicine can be used up to twice daily (up to 2 tablets in 24 hours).
-
Do not chew tablet.
-
MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg should not be given to children under 12 years old unless directed by a doctor.
-
Allergy alert: Do not use if you are allergic to ranitidine or other acid reducers.
How does MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg work?
- MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg reduce the production of stomach acid. This is what make MAXIMUM STRENGTH Ranitidine Tablets USP, 150 mg different from antacids, which neutralize the acid already in your stomach. Antacids do not reduce the production of acid.
Tips for managing heartburn
- Do not lie flat or bend over soon after eating
- Do not eat late at night, or just before bedtime
- Certain foods or drinks are more likely to cause heartburn, such as rich, spicy, fatty, and fried foods chocolate, caffeine, alcohol, even some fruits and vegetables
- Eat slowly and do not eat big meals
- If you are overweight, lose weight
- If you smoke, quit smoking
- Raise the head of your bed
- Wear loose fitting clothing around your stomach
When should I see a doctor?
Do not Use
- If you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools. These may be signs of a serious condition. See your doctor. with other acid reducers
- if you have kidney disease, except under the advice and supervision of a doctor
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- heartbrun with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Stop use and ask a doctor if
- your heartburn continues or worsens
- you need to take this product for more than 14 days
- If pregnant or breast-feeding, ask a health professional before use.
- Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Questions? Call 1-888-375-3784
BOTTLES: Bottle is sealed with printed foil under cap. Do not use if printed foil is open or torn.
BLISTERS: Do not use if the individual blister unit is open or torn.
- Consumer Information
- SPL UNCLASSIFIED SECTION
- PACKAGE LABEL PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ACID REDUCER
ranitidine tablet, coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-869(NDC:55111-404) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Ranitidine Hydrochloride (UNII: BK76465IHM) (RANITIDINE - UNII:884KT10YB7) RANITIDINE 150 mg Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) FERROSOFERRIC OXIDE (UNII: XM0M87F357) magnesium stearate (UNII: 70097M6I30) cellulose, microcrystalline (UNII: OP1R32D61U) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) titanium dioxide (UNII: 15FIX9V2JP) Product Characteristics Color PINK Score no score Shape ROUND Size 9mm Flavor Imprint Code R150 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-869-62 1 in 1 CARTON 05/01/2010 09/30/2019 1 95 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078192 05/01/2010 Labeler - CVS HEALTH CORP (062312574)




