TOPIRAMATE- topiramate tablet, film coated
Lake Erie Medical & Surgical Supply DBA Quality Care Products LLC
----------
Topiramate Tablets
DESCRIPTION
Topiramate is a sulfamate-substituted monosaccharide. Topiramate tablets are available as 25 mg, 50 mg, 100 mg, and 200 mg circular tablets for oral administration.
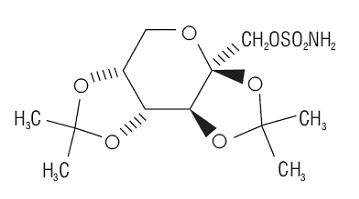
Topiramate is a white crystalline powder with a bitter taste. Topiramate USP is most soluble in alkaline solutions containing sodium hydroxide or sodium phosphate and having a pH of 9 to 10. It is freely soluble in acetone, chloroform, dimethylsulfoxide, and ethanol. The solubility in water is 9.8 mg/mL. Its saturated solution has a pH of 6.3. Topiramate has the molecular formula C12H21NO8S and a molecular weight of 339.37. Topiramate is designated chemically as 2,3:4,5-Di-O-isopropylidene-β-D-fructopyranose sulfamate and has the following structural formula:

Topiramate tablets contain the following inactive ingredients: anhydrous lactose, microcrystalline cellulose, pregelatinized starch, sodium starch glycolate, magnesium stearate, purified water, polyvinyl alcohol, titanium dioxide, polyethylene glycol and talc.
In addition, individual tablets contain:
50 mg tablets: iron oxide yellow
100 mg tablets: iron oxide yellow, and D&C Yellow # 10 Aluminum Lake
200 mg tablets: iron oxide red, lecithin (soya), and iron oxide black
CLINICAL STUDIES
The studies described in the following sections were conducted using topiramate tablets.
Adjunctive Therapy Controlled Trials in Patients With Partial Onset Seizures
The effectiveness of topiramate as an adjunctive treatment for adults with partial onset seizures was established in six multicenter, randomized, double-blind, placebo-controlled trials, two comparing several dosages of topiramate and placebo and four comparing a single dosage with placebo, in patients with a history of partial onset seizures, with or without secondarily generalized seizures.
Patients in these studies were permitted a maximum of two antiepileptic drugs (AEDs) in addition to topiramate tablets or placebo. In each study, patients were stabilized on optimum dosages of their concomitant AEDs during baseline phase lasting between 4 and 12 weeks. Patients who experienced a prespecified minimum number of partial onset seizures, with or without secondary generalization, during the baseline phase (12 seizures for 12-week baseline, 8 for 8-week baseline, or 3 for 4-week baseline) were randomly assigned to placebo or a specified dose of topiramate tablets in addition to their other AEDs.
Following randomization, patients began the double-blind phase of treatment. In five of the six studies, patients received active drug beginning at 100 mg per day; the dose was then increased by 100 mg or 200 mg/day increments weekly or every other week until the assigned dose was reached, unless intolerance prevented increases. In the sixth study (119), the 25 or 50 mg/day initial doses of topiramate were followed by respective weekly increments of 25 or 50 mg/day until the target dose of 200 mg/day was reached. After titration, patients entered a 4, 8, or 12-week stabilization period. The numbers of patients randomized to each dose, and the actual mean and median doses in the stabilization period are shown in Table 1.
Adjunctive Therapy Controlled Trial in Pediatric Patients Ages 2 to 16 Years With Partial Onset Seizures
The effectiveness of topiramate as an adjunctive treatment for pediatric patients ages 2 to 16 years with partial onset seizures was established in a multicenter, randomized, double-blind, placebo-controlled trial, comparing topiramate and placebo in patients with a history of partial onset seizures, with or without secondarily generalized seizures.
Patients in this study were permitted a maximum of two antiepileptic drugs (AEDs) in addition to topiramate tablets or placebo. In this study, patients were stabilized on optimum dosages of their concomitant AEDs during an 8-week baseline phase. Patients who experienced at least six partial onset seizures, with or without secondarily generalized seizures, during the baseline phase were randomly assigned to placebo or topiramate tablets in addition to their other AEDs.
Following randomization, patients began the double-blind phase of treatment. Patients received active drug beginning at 25 or 50 mg per day; the dose was then increased by 25 mg to 150 mg/day increments every other week until the assigned dosage of 125, 175, 225, or 400 mg/day based on patients' weight to approximate a dosage of 6 mg/kg per day was reached, unless intolerance prevented increases. After titration, patients entered an 8-week stabilization period.
Adjunctive Therapy Controlled Trial in Patients With Primary Generalized Tonic-Clonic Seizures
The effectiveness of topiramate as an adjunctive treatment for primary generalized tonic-clonic seizures in patients 2 years old and older was established in a multicenter, randomized, double-blind, placebo-controlled trial, comparing a single dosage of topiramate and placebo.
Patients in this study were permitted a maximum of two antiepileptic drugs (AEDs) in addition to topiramate or placebo. Patients were stabilized on optimum dosages of their concomitant AEDs during an 8-week baseline phase. Patients who experienced at least three primary generalized tonic-clonic seizures during the baseline phase were randomly assigned to placebo or topiramate in addition to their other AEDs.
Following randomization, patients began the double-blind phase of treatment. Patients received active drug beginning at 50 mg per day for four weeks; the dose was then increased by 50 mg to 150 mg/day increments every other week until the assigned dose of 175, 225, or 400 mg/day based on patients' body weight to approximate a dosage of 6 mg/kg per day was reached, unless intolerance prevented increases. After titration, patients entered a 12-week stabilization period.
Adjunctive Therapy Controlled Trial in Patients With Lennox-Gastaut Syndrome
The effectiveness of topiramate as an adjunctive treatment for seizures associated with Lennox-Gastaut syndrome was established in a multicenter, randomized, double-blind, placebo-controlled trial comparing a single dosage of topiramate with placebo in patients 2 years of age and older.
Patients in this study were permitted a maximum of two antiepileptic drugs (AEDs) in addition to topiramate or placebo. Patients who were experiencing at least 60 seizures per month before study entry were stabilized on optimum dosages of their concomitant AEDs during a 4-week baseline phase. Following baseline, patients were randomly assigned to placebo or topiramate in addition to their other AEDs. Active drug was titrated beginning at 1 mg/kg per day for a week; the dose was then increased to 3 mg/kg per day for one week then to 6 mg/kg per day. After titration, patients entered an 8-week stabilization period. The primary measures of effectiveness were the percent reduction in drop attacks and a parental global rating of seizure severity.
| Target Topiramate Dosage (mg/day) | ||||||||
| Protocol | Stabilization Dose | Placeboa | 200 | 400 | 600 | 800 | 1,000 | |
|
a Placebo dosages are given as the number of tablets. Placebo target dosages were as follows: Protocol Y1, 4 tablets/day; Protocols YD and Y2, 6 tablets/day; Protocol Y3 and 119, 8 tablets/day; Protocol YE, 10 tablets/day. |
||||||||
|
b Dose-response studies were not conducted for other indications or pediatric partial onset seizures. |
||||||||
| YD | N | 42 | 42 | 40 | 41 | -- | -- | |
| Mean Dose | 5.9 | 200 | 390 | 556 | -- | -- | ||
| Median Dose | 6.0 | 200 | 400 | 600 | -- | -- | ||
| YE | N | 44 | -- | -- | 40 | 45 | 40 | |
| Mean Dose | 9.7 | -- | -- | 544 | 739 | 796 | ||
| Median Dose | 10.0 | -- | -- | 600 | 800 | 1,000 | ||
| Y1 | N | 23 | -- | 19 | -- | -- | -- | |
| Mean Dose | 3.8 | -- | 395 | -- | -- | -- | ||
| Median Dose | 4.0 | -- | 400 | -- | -- | -- | ||
| Y2 | N | 30 | -- | -- | 28 | -- | -- | |
| Mean Dose | 5.7 | -- | -- | 522 | -- | -- | ||
| Median Dose | 6.0 | -- | -- | 600 | -- | -- | ||
| Y3 | N | 28 | -- | -- | -- | 25 | -- | |
| Mean Dose | 7.9 | -- | -- | -- | 568 | -- | ||
| Median Dose | 8.0 | -- | -- | -- | 600 | -- | ||
| 119 N | 90 | 157 | -- | -- | -- | -- | ||
| Mean Dose | 8 | 200 | -- | -- | -- | -- | ||
| Median Dose | 8 | 200 | -- | -- | -- | -- | ||
In all add-on trials, the reduction in seizure rate from baseline during the entire double-blind phase was measured. The median percent reductions in seizure rates and the responder rates (fraction of patients with at least a 50% reduction) by treatment group for each study are shown below in Table 2. As described above, a global improvement in seizure severity was also assessed in the Lennox-Gastaut trial.
| Target Topiramate Dosage (mg/day) | ||||||||
| Protocol | Efficacy Results | Placebo | 200 | 400 | 600 | 800 | 1,000 | ≈6 mg/kg/day* |
|
Comparisons with placebo: a p=0.080; b p<0.010; c p<0.001; d p<0.050; e p=0.065; f p<0.005;g p=0.071; |
||||||||
|
h Median % reduction and % responders are reported for PGTC Seizures; |
||||||||
|
i Median % reduction and % responders for drop attacks, i.e., tonic or atonic seizures; |
||||||||
|
j Percent of subjects who were minimally, much, or very much improved from baseline |
||||||||
|
* For Protocols YP and YTC, protocol-specified target dosages (<9.3 mg/kg/day) were assigned based on subject's weight to approximate a dosage of 6 mg/kg per day; these dosages corresponded to mg/day dosages of 125, 175, 225, and 400 mg/day. |
||||||||
| Partial Onset Seizures | ||||||||
| Studies in Adults | ||||||||
| YD | N | 45 | 45 | 45 | 46 | -- | -- | -- |
| Median % Reduction | 11.6 | 27.2a | 47.5b | 44.7c | -- | -- | -- | |
| % Responders | 18 | 24 | 44d | 46d | -- | -- | -- | |
| YE | N | 47 | -- | -- | 48 | 48 | 47 | -- |
| Median % Reduction | 1.7 | -- | -- | 40.8c | 41.0c | 36.0c | -- | |
| % Responders | 9 | -- | -- | 40c | 41c | 36d | -- | |
| Y1 | N | 24 | -- | 23 | -- | -- | -- | -- |
| Median % Reduction | 1.1 | -- | 40.7e | -- | -- | -- | -- | |
| % Responders | 8 | -- | 35d | -- | -- | -- | -- | |
| Y2 | N | 30 | -- | -- | 30 | -- | -- | -- |
| Median % Reduction | -12.2 | -- | -- | 46.4f | -- | -- | -- | |
| % Responders | 10 | -- | -- | 47c | -- | -- | -- | |
| Y3 | N | 28 | -- | -- | -- | 28 | -- | -- |
| Median % Reduction | -20.6 | -- | -- | -- | 24.3c | -- | -- | |
| % Responders | 0 | -- | -- | -- | 43c | -- | -- | |
| 119 N | 91 | 168 | -- | -- | -- | -- | -- | |
| Median % Reduction | 20.0 | 44.2c | -- | -- | -- | -- | -- | |
| % Responders | 24 | 45c | -- | -- | -- | -- | -- | |
| Studies in Pediatric Patients | ||||||||
| YP | N | 45 | -- | -- | -- | -- | -- | 41 |
| Median % Reduction | 10.5 | -- | -- | -- | -- | -- | 33.1d | |
| % Responders | 20 | -- | -- | -- | -- | -- | 39 | |
| Primary Generalized Tonic-Clonich | ||||||||
| YTC | N | 40 | -- | -- | -- | -- | -- | 39 |
| Median % Reduction | 9.0 | -- | -- | -- | -- | -- | 56.7d | |
| % Responders | 20 | -- | -- | -- | -- | -- | 56c | |
| Lennox-Gastaut Syndromei | ||||||||
| YL | N | 49 | -- | -- | -- | -- | -- | 46 |
| Median % Reduction | -5.1 | -- | -- | -- | -- | -- | 14.8d | |
| % Responders | 14 | -- | -- | -- | -- | -- | 28g | |
| Improvement in Seizure Severityj | 28 | -- | -- | -- | -- | -- | 52d | |
Subset analyses of the antiepileptic efficacy of topiramate tablets in these studies showed no differences as a function of gender, race, age, baseline seizure rate, or concomitant AED.
INDICATIONS AND USAGE
Monotherapy Epilepsy
Topiramate tablets are indicated as initial monotherapy in patients 10 years of age and older with partial onset or primary generalized tonic-clonic seizures.
Effectiveness was demonstrated in a controlled trial in patients with epilepsy who had no more than 2 seizures in the 3 months prior to enrollment. Safety and effectiveness in patients who were converted to monotherapy from a previous regimen of other anticonvulsant drugs have not been established in controlled trials.
Adjunctive Therapy Epilepsy
Topiramate tablets are indicated as adjunctive therapy for adults and pediatric patients ages 2 to 16 years with partial onset seizures, or primary generalized tonic-clonic seizures, and in patients 2 years of age and older with seizures associated with Lennox-Gastaut syndrome.
CONTRAINDICATIONS
Topiramate tablets are contraindicated in patients with a history of hypersensitivity to any component of this product.
ADVERSE REACTIONS
The data described in the following section were obtained using topiramate tablets.
Monotherapy Epilepsy
The adverse events in the controlled trial that occurred most commonly in adults in the 400 mg/day group and at a rate higher than the 50 mg/day group were: paresthesia, weight decrease, somnolence, anorexia, dizziness, and difficulty with memory NOS [see Table 5].
The adverse events in the controlled trial that occurred most commonly in children (10 years up to 16 years of age) in the 400 mg/day group and at a rate higher than the 50 mg/day group were: weight decrease, upper respiratory tract infection, paresthesia, anorexia, diarrhea, and mood problems [see Table 6].
Approximately 21% of the 159 adult patients in the 400 mg/day group who received topiramate as monotherapy in the controlled clinical trial discontinued therapy due to adverse events. Adverse events associated with discontinuing therapy (≥2%) included depression, insomnia, difficulty with memory (NOS), somnolence, paresthesia, psychomotor slowing, dizziness, and nausea.
Approximately 12% of the 57 pediatric patients in the 400 mg/day group who received topiramate as monotherapy in the controlled clinical trial discontinued therapy due to adverse events. Adverse events associated with discontinuing therapy (≥5%) included difficulty with concentration/attention.
The prescriber should be aware that these data cannot be used to predict the frequency of adverse events in the course of usual medical practice where patient characteristics and other factors may differ from those prevailing during the clinical study. Similarly, the cited frequencies cannot be directly compared with data obtained from other clinical investigations involving different treatments, uses, or investigators. Inspection of these frequencies, however, does provide the prescribing physician with a basis to estimate the relative contribution of drug and non-drug factors to the adverse event incidences in the population studied.
| Topiramate Dosage (mg/day) | ||
| Body System/ Adverse Event | 50 (N= 160) | 400 (N=159) |
|
a Values represent the percentage of patients reporting a given adverse event. Patients may have reported more than one adverse event during the study and can be included in more than one adverse event category. |
||
| Body as a Whole-General Disorders | ||
| Asthenia | 4 | 6 |
| Leg Pain | 2 | 3 |
| Chest Pain | 1 | 2 |
| Central & Peripheral Nervous System Disorders | ||
| Paresthesia | 21 | 40 |
| Dizziness | 13 | 14 |
| Hypoaesthesia | 4 | 5 |
| Ataxia | 3 | 4 |
| Hypertonia | 0 | 3 |
| Gastro-Intestinal System Disorders | ||
| Diarrhea | 5 | 6 |
| Constipation | 1 | 4 |
| Gastritis | 0 | 3 |
| Dry Mouth | 1 | 3 |
| Gastroesophageal Reflux | 1 | 2 |
| Liver and Biliary System Disorders | ||
| Gamma-GT Increased | 1 | 3 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 6 | 16 |
| Psychiatric Disorders | ||
| Somnolence | 9 | 15 |
| Anorexia | 4 | 14 |
| Difficulty with Memory NOS | 5 | 10 |
| Insomnia | 8 | 9 |
| Depression | 7 | 9 |
| Difficulty with Concentration/Attention | 7 | 8 |
| Anxiety | 4 | 6 |
| Psychomotor Slowing | 3 | 5 |
| Mood Problems | 2 | 5 |
| Confusion | 3 | 4 |
| Cognitive Problem NOS | 1 | 4 |
| Libido Decreased | 0 | 3 |
| Reproductive Disorders, Female | ||
| Vaginal Hemorrhage | 0 | 3 |
| Red Blood Cell Disorders | ||
| Anemia | 1 | 2 |
| Resistance Mechanism Disorders | ||
| Infection Viral | 6 | 8 |
| Infection | 2 | 3 |
| Respiratory System Disorders | ||
| Bronchitis | 3 | 4 |
| Rhinitis | 2 | 4 |
| Dyspnea | 1 | 2 |
| Skin and Appendages Disorders | ||
| Rash | 1 | 4 |
| Pruritus | 1 | 4 |
| Acne | 2 | 3 |
| Special Senses Other, Disorders | ||
| Taste Perversion | 3 | 5 |
| Urinary System Disorders | ||
| Cystitis | 1 | 3 |
| Renal Calculus | 0 | 3 |
| Urinary Tract Infection | 1 | 2 |
| Dysuria | 0 | 2 |
| Micturition Frequency | 0 | 2 |
| Topiramate Dosage (mg/day) | ||
| Body System/ Adverse Event | 50 (N=57) | 400 (N=57) |
|
a Values represent the percentage of patients reporting a given adverse event. Patients may have reported more than one adverse event during the study and can be included in more than one adverse event category. |
||
| Body as a Whole-General Disorders | ||
| Fever | 0 | 9 |
| Central & Peripheral Nervous System Disorders | ||
| Paresthesia | 2 | 16 |
| Gastro-Intestinal System Disorders | ||
| Diarrhea | 5 | 11 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 7 | 21 |
| Psychiatric Disorders | ||
| Anorexia | 11 | 14 |
| Mood Problems | 2 | 11 |
| Difficulty with Concentration/Attention | 4 | 9 |
| Cognitive Problems NOS | 0 | 7 |
| Nervousness | 4 | 5 |
| Resistance Mechanism Disorders | ||
| Infection Viral | 4 | 9 |
| Infection | 2 | 7 |
| Respiratory System Disorders | ||
| Upper Respiratory Tract Infection | 16 | 18 |
| Rhinitis | 2 | 7 |
| Bronchitis | 2 | 7 |
| Sinusitis | 2 | 5 |
| Skin and Appendages Disorders | ||
| Alopecia | 2 | 5 |
Adjunctive Therapy Epilepsy
The most commonly observed adverse events associated with the use of topiramate at dosages of 200 to 400 mg/day in controlled trials in adults with partial onset seizures, primary generalized tonic-clonic seizures, or Lennox-Gastaut syndrome, that were seen at greater frequency in topiramate-treated patients and did not appear to be dose-related were: somnolence, dizziness, ataxia, speech disorders and related speech problems, psychomotor slowing, abnormal vision, difficulty with memory, paresthesia and diplopia [see Table 7]. The most common dose-related adverse events at dosages of 200 to 1,000 mg/day were: fatigue, nervousness, difficulty with concentration or attention, confusion, depression, anorexia, language problems, anxiety, mood problems, and weight decrease [see Table 9].
Adverse events associated with the use of topiramate at dosages of 5 to 9 mg/kg/day in controlled trials in pediatric patients with partial onset seizures, primary generalized tonic-clonic seizures, or Lennox-Gastaut syndrome, that were seen at greater frequency in topiramate-treated patients were: fatigue, somnolence, anorexia, nervousness, difficulty with concentration/attention, difficulty with memory, aggressive reaction, and weight decrease [see Table 10].
In controlled clinical trials in adults, 11% of patients receiving topiramate 200 to 400 mg/day as adjunctive therapy discontinued due to adverse events. This rate appeared to increase at dosages above 400 mg/day. Adverse events associated with discontinuing therapy included somnolence, dizziness, anxiety, difficulty with concentration or attention, fatigue, and paresthesia and increased at dosages above 400 mg/day. None of the pediatric patients who received topiramate adjunctive therapy at 5 to 9 mg/kg/day in controlled clinical trials discontinued due to adverse events.
Approximately 28% of the 1,757 adults with epilepsy who received topiramate at dosages of 200 to 1,600 mg/day in clinical studies discontinued treatment because of adverse events; an individual patient could have reported more than one adverse event. These adverse events were: psychomotor slowing (4%), difficulty with memory (3.2%), fatigue (3.2%), confusion (3.1%), somnolence (3.2%), difficulty with concentration/attention (2.9%), anorexia (2.7%), depression (2.6%), dizziness (2.5%), weight decrease (2.5%), nervousness (2.3%), ataxia (2.1%), and paresthesia (2%). Approximately 11% of the 310 pediatric patients who received topiramate at dosages up to 30 mg/kg/day discontinued due to adverse events. Adverse events associated with discontinuing therapy included aggravated convulsions (2.3%), difficulty with concentration/attention (1.6%), language problems (1.3%), personality disorder (1.3%), and somnolence (1.3%).
Incidence in Epilepsy Controlled Clinical Trials - Adjunctive Therapy – Partial Onset Seizures, Primary Generalized Tonic-Clonic Seizures, and Lennox-Gastaut Syndrome
Table 7 lists treatment-emergent adverse events that occurred in at least 1% of adults treated with 200 to 400 mg/day topiramate in controlled trials that were numerically more common at this dose than in the patients treated with placebo. In general, most patients who experienced adverse events during the first eight weeks of these trials no longer experienced them by their last visit. Table 10 lists treatment-emergent adverse events that occurred in at least 1% of pediatric patients treated with 5 to 9 mg/kg topiramate in controlled trials that were numerically more common than in patients treated with placebo.
The prescriber should be aware that these data were obtained when topiramate was added to concurrent antiepileptic drug therapy and cannot be used to predict the frequency of adverse events in the course of usual medical practice where patient characteristics and other factors may differ from those prevailing during clinical studies. Similarly, the cited frequencies cannot be directly compared with data obtained from other clinical investigations involving different treatments, uses, or investigators. Inspection of these frequencies, however, does provide the prescribing physician with a basis to estimate the relative contribution of drug and non-drug factors to the adverse event incidences in the population studied.
Other Adverse Events Observed During Double-Blind Epilepsy Adjunctive Therapy Trials
Other events that occurred in more than 1% of adults treated with 200 to 400 mg of topiramate in placebo-controlled epilepsy trials but with equal or greater frequency in the placebo group were: headache, injury, anxiety, rash, pain, convulsions aggravated, coughing, fever, diarrhea, vomiting, muscle weakness, insomnia, personality disorder, dysmenorrhea, upper respiratory tract infection, and eye pain.
| Topiramate Dosage (mg/day) | ||||||||||
| Body System/ Adverse Eventc | Placebo (N=291) | 200 to 400 (N=183) | 600 to 1,000 (N=414) |
|||||||
|
a Patients in these add-on trials were receiving 1 to 2 concomitant antiepileptic drugs in addition to topiramate or placebo. |
||||||||||
|
b Values represent the percentage of patients reporting a given adverse event. Patients may have reported more than one adverse event during the study and can be included in more than one adverse event category. |
||||||||||
|
c Adverse events reported by at least 1% of patients in the topiramate 200 to 400 mg/day group and more common than in the placebo group are listed in this table. |
||||||||||
| Body as a Whole-General Disorders | ||||||||||
| Fatigue | 13 | 15 | 30 | |||||||
| Asthenia | 1 | 6 | 3 | |||||||
| Back Pain | 4 | 5 | 3 | |||||||
| Chest Pain | 3 | 4 | 2 | |||||||
| Influenza-Like Symptoms | 2 | 3 | 4 | |||||||
| Leg Pain | 2 | 2 | 4 | |||||||
| Hot Flushes | 1 | 2 | 1 | |||||||
| Allergy | 1 | 2 | 3 | |||||||
| Edema | 1 | 2 | 1 | |||||||
| Body Odor | 0 | 1 | 0 | |||||||
| Rigors | 0 | 1 | <1 | |||||||
| Central & Peripheral Nervous System Disorders | ||||||||||
| Dizziness | 15 | 25 | 32 | |||||||
| Ataxia | 7 | 16 | 14 | |||||||
| Speech Disorders/Related Speech Problems | 2 | 13 | 11 | |||||||
| Paresthesia | 4 | 11 | 19 | |||||||
| Nystagmus | 7 | 10 | 11 | |||||||
| Tremor | 6 | 9 | 9 | |||||||
| Language Problems | 1 | 6 | 10 | |||||||
| Coordination Abnormal | 2 | 4 | 4 | |||||||
| Hypoaesthesia | 1 | 2 | 1 | |||||||
| Gait Abnormal | 1 | 3 | 2 | |||||||
| Muscle Contractions Involuntary | 1 | 2 | 2 | |||||||
| Stupor | 0 | 2 | 1 | |||||||
| Vertigo | 1 | 1 | 2 | |||||||
| Gastro-Intestinal System Disorders | ||||||||||
| Nausea | 8 | 10 | 12 | |||||||
| Dyspepsia | 6 | 7 | 6 | |||||||
| Abdominal Pain | 4 | 6 | 7 | |||||||
| Constipation | 2 | 4 | 3 | |||||||
| Gastroenteritis | 1 | 2 | 1 | |||||||
| Dry Mouth | 1 | 2 | 4 | |||||||
| Gingivitis | <1 | 1 | 1 | |||||||
| GI Disorder | <1 | 1 | 0 | |||||||
| Hearing and Vestibular Disorders | ||||||||||
| Hearing Decreased | 1 | 2 | 1 | |||||||
| Metabolic and Nutritional Disorders | ||||||||||
| Weight Decrease | 3 | 9 | 13 | |||||||
| Muscle-Skeletal System Disorders | ||||||||||
| Myalgia | 1 | 2 | 2 | |||||||
| Skeletal Pain | 0 | 1 | 0 | |||||||
| Platelet, Bleeding, & Clotting Disorders | ||||||||||
| Epistaxis | 1 | 2 | 1 | |||||||
| Psychiatric Disorders | ||||||||||
| Somnolence | 12 | 29 | 28 | |||||||
| Nervousness | 6 | 16 | 19 | |||||||
| Psychomotor Slowing | 2 | 13 | 21 | |||||||
| Difficulty with Memory | 3 | 12 | 14 | |||||||
| Anorexia | 4 | 10 | 12 | |||||||
| Confusion | 5 | 11 | 14 | |||||||
| Depression | 5 | 5 | 13 | |||||||
| Difficulty with Concentration/Attention | 2 | 6 | 14 | |||||||
| Mood Problems | 2 | 4 | 9 | |||||||
| Agitation | 2 | 3 | 3 | |||||||
| Aggressive Reaction | 2 | 3 | 3 | |||||||
| Emotional Lability | 1 | 3 | 3 | |||||||
| Cognitive Problems | 1 | 3 | 3 | |||||||
| Libido Decreased | 1 | 2 | <1 | |||||||
| Apathy | 1 | 1 | 3 | |||||||
| Depersonalization | 1 | 1 | 2 | |||||||
| Reproductive Disorders, Female | ||||||||||
| Breast Pain | 2 | 4 | 0 | |||||||
| Amenorrhea | 1 | 2 | 2 | |||||||
| Menorrhagia | 0 | 2 | 1 | |||||||
| Menstrual Disorder | 1 | 2 | 1 | |||||||
| Reproductive Disorders, Male | ||||||||||
| Prostatic Disorder | <1 | 2 | 0 | |||||||
| Resistance Mechanism Disorders | ||||||||||
| Infection | 1 | 2 | 1 | |||||||
| Infection Viral | 1 | 2 | <1 | |||||||
| Moniliasis | <1 | 1 | 0 | |||||||
| Respiratory System Disorders | ||||||||||
| Pharyngitis | 2 | 6 | 3 | |||||||
| Rhinitis | 6 | 7 | 6 | |||||||
| Sinusitis | 4 | 5 | 6 | |||||||
| Dyspnea | 1 | 1 | 2 | |||||||
| Skin and Appendages Disorders | ||||||||||
| Skin Disorder | <1 | 2 | 1 | |||||||
| Sweating Increased | <1 | 1 | <1 | |||||||
| Rash Erythematous | <1 | 1 | <1 | |||||||
| Special Sense Other, Disorders | ||||||||||
| Taste Perversion | 0 | 2 | 4 | |||||||
| Urinary System Disorders | ||||||||||
| Hematuria | 1 | 2 | <1 | |||||||
| Urinary Tract Infection | 1 | 2 | 3 | |||||||
| Micturition Frequency | 1 | 1 | 2 | |||||||
| Urinary Incontinence | <1 | 2 | 1 | |||||||
| Urine Abnormal | 0 | 1 | <1 | |||||||
| Vision Disorders | ||||||||||
| Vision Abnormal | 2 | 13 | 10 | |||||||
| Diplopia | 5 | 10 | 10 | |||||||
| White Cell and RES Disorders | ||||||||||
| Leukopenia | 1 | 2 | 1 | |||||||
Incidence in Study 119 – Add-On Therapy– Adults with Partial Onset Seizures
Study 119 was a randomized, double-blind, placebo-controlled, parallel group study with 3 treatment arms: 1) placebo; 2) topiramate 200 mg/day with a 25 mg/day starting dose, increased by 25 mg/day each week for 8 weeks until the 200 mg/day maintenance dose was reached; and 3) topiramate 200 mg/day with a 50 mg/day starting dose, increased by 50 mg/day each week for 4 weeks until the 200 mg/day maintenance dose was reached. All patients were maintained on concomitant carbamazepine with or without another concomitant antiepileptic drug.
The incidence of adverse events (Table 8) did not differ significantly between the 2 topiramate regimens. Because the frequencies of adverse events reported in this study were markedly lower than those reported in the previous epilepsy studies, they cannot be directly compared with data obtained in other studies.
| Topiramate Dosage (mg/day) | ||
| Body System/
Adverse Eventc | Placebo (N=92) | 200 (N=171) |
|
a Patients in these add-on trials were receiving 1 to 2 concomitant antiepileptic drugs in addition to topiramate or placebo. |
||
|
b Values represent the percentage of patients reporting a given adverse event. Patients may have reported more than one adverse event during the study and can be included in more than one adverse event category. |
||
|
c Adverse events reported by at least 2% of patients in the topiramate 200 mg/day group and more common than in the placebo group are listed in this table. |
||
| Body as a Whole-General Disorders | ||
| Fatigue | 4 | 9 |
| Chest Pain | 1 | 2 |
| Cardiovascular Disorders, General | ||
| Hypertension | 0 | 2 |
| Central & Peripheral Nervous System Disorders | ||
| Paresthesia | 2 | 9 |
| Dizziness | 4 | 7 |
| Tremor | 2 | 3 |
| Hypoasthesia | 0 | 2 |
| Leg Cramps | 0 | 2 |
| Language Problems | 0 | 2 |
| Gastro-Intestinal System Disorders | ||
| Abdominal Pain | 3 | 5 |
| Constipation | 0 | 4 |
| Diarrhea | 1 | 2 |
| Dyspepsia | 0 | 2 |
| Dry Mouth | 0 | 2 |
| Hearing and Vestibular Disorders | ||
| Tinnitus | 0 | 2 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 4 | 8 |
| Psychiatric Disorders | ||
| Somnolence | 9 | 15 |
| Anorexia | 7 | 9 |
| Nervousness | 2 | 9 |
| Difficulty with Concentration/Attention | 0 | 5 |
| Insomnia | 3 | 4 |
| Difficulty with Memory | 1 | 2 |
| Aggressive Reaction | 0 | 2 |
| Respiratory System Disorders | ||
| Rhinitis | 0 | 4 |
| Urinary System Disorders | ||
| Cystitis | 0 | 2 |
| Vision Disorders | ||
| Diplopia | 0 | 2 |
| Vision Abnormal | 0 | 2 |
| Topiramate Dosage (mg/day) | ||||
|
Adverse Event | Placebo (N = 216) | 200 (N = 45) | 400 (N = 68) | 600 to 1,000 (N = 414) |
|
a Dose-response studies were not conducted for other adult indications or for pediatric indications. |
||||
| Fatigue | 13 | 11 | 12 | 30 |
| Nervousness | 7 | 13 | 18 | 19 |
| Difficulty with Concentration/Attention | 1 | 7 | 9 | 14 |
| Confusion | 4 | 9 | 10 | 14 |
| Depression | 6 | 9 | 7 | 13 |
| Anorexia | 4 | 4 | 6 | 12 |
| Language problems | <1 | 2 | 9 | 10 |
| Anxiety | 6 | 2 | 3 | 10 |
| Mood problems | 2 | 0 | 6 | 9 |
| Weight decrease | 3 | 4 | 9 | 13 |
| Body System/
Adverse Event | Placebo (N=101) | Topiramate (N=98) |
|
a Patients in these add-on trials were receiving 1 to 2 concomitant antiepileptic drugs in addition to topiramate or placebo. |
||
|
b Values represent the percentage of patients reporting a given adverse event. Patients may have reported more than one adverse event during the study and can be included in more than one adverse event category. |
||
| Body as a Whole - General Disorders | ||
| Fatigue | 5 | 16 |
| Injury | 13 | 14 |
| Allergic Reaction | 1 | 2 |
| Back Pain | 0 | 1 |
| Pallor | 0 | 1 |
| Cardiovascular Disorders, General | ||
| Hypertension | 0 | 1 |
| Central & Peripheral Nervous System Disorders | ||
| Gait Abnormal | 5 | 8 |
| Ataxia | 2 | 6 |
| Hyperkinesia | 4 | 5 |
| Dizziness | 2 | 4 |
| Speech Disorders/Related Speech Problems | 2 | 4 |
| Hyporeflexia | 0 | 2 |
| Convulsions Grand Mal | 0 | 1 |
| Fecal Incontinence | 0 | 1 |
| Paresthesia | 0 | 1 |
| Gastro-Intestinal System Disorders | ||
| Nausea | 5 | 6 |
| Saliva Increased | 4 | 6 |
| Constipation | 4 | 5 |
| Gastroenteritis | 2 | 3 |
| Dysphagia | 0 | 1 |
| Flatulence | 0 | 1 |
| Gastroesophageal Reflux | 0 | 1 |
| Glossitis | 0 | 1 |
| Gum Hyperplasia | 0 | 1 |
| Heart Rate and Rhythm Disorders | ||
| Bradycardia | 0 | 1 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 1 | 9 |
| Thirst | 1 | 2 |
| Hypoglycemia | 0 | 1 |
| Weight Increase | 0 | 1 |
| Platelet, Bleeding, & Clotting Disorders | ||
| Purpura | 4 | 8 |
| Epistaxis | 1 | 4 |
| Hematoma | 0 | 1 |
| Prothrombin Increased | 0 | 1 |
| Thrombocytopenia | 0 | 1 |
| Psychiatric Disorders | ||
| Somnolence | 16 | 26 |
| Anorexia | 15 | 24 |
| Nervousness | 7 | 14 |
| Personality Disorder (Behavior Problems) | 9 | 11 |
| Difficulty with Concentration/Attention | 2 | 10 |
| Aggressive Reaction | 4 | 9 |
| Insomnia | 7 | 8 |
| Difficulty with Memory NOS | 0 | 5 |
| Confusion | 3 | 4 |
| Psychomotor Slowing | 2 | 3 |
| Appetite Increased | 0 | 1 |
| Neurosis | 0 | 1 |
| Reproductive Disorders, Female | ||
| Leukorrhoea | 0 | 2 |
| Resistance Mechanism Disorders | ||
| Infection Viral | 3 | 7 |
| Respiratory System Disorders | ||
| Pneumonia | 1 | 5 |
| Respiratory Disorder | 0 | 1 |
| Skin and Appendages Disorders | ||
| Skin Disorder | 2 | 3 |
| Alopecia | 1 | 2 |
| Dermatitis | 0 | 2 |
| Hypertrichosis | 1 | 2 |
| Rash Erythematous | 0 | 2 |
| Eczema | 0 | 1 |
| Seborrhoea | 0 | 1 |
| Skin Discoloration | 0 | 1 |
| Urinary System Disorders | ||
| Urinary Incontinence | 2 | 4 |
| Nocturia | 0 | 1 |
| Vision Disorders | ||
| Eye Abnormality | 1 | 2 |
| Vision Abnormal | 1 | 2 |
| Diplopia | 0 | 1 |
| Lacrimation Abnormal | 0 | 1 |
| Myopia | 0 | 1 |
| White Cell and RES Disorders | ||
| Leukopenia | 0 | 2 |
Other Adverse Events Observed During All Epilepsy Clinical Trials
Topiramate has been administered to 2,246 adults and 427 pediatric patients with epilepsy during all clinical studies, only some of which were placebo controlled. During these studies, all adverse events were recorded by the clinical investigators using terminology of their own choosing. To provide a meaningful estimate of the proportion of individuals having adverse events, similar types of events were grouped into a smaller number of standardized categories using modified WHOART dictionary terminology. The frequencies presented represent the proportion of patients who experienced an event of the type cited on at least one occasion while receiving topiramate. Reported events are included except those already listed in the previous tables or text, those too general to be informative, and those not reasonably associated with the use of the drug.
Events are classified within body system categories and enumerated in order of decreasing frequency using the following definitions: frequent occurring in at least 1/100 patients; infrequent occurring in 1/100 to 1/1000 patients; rare occurring in fewer than 1/1000 patients.
Autonomic Nervous System Disorders: Infrequent: vasodilation.
Body as a Whole: Frequent: syncope. Infrequent: abdomen enlarged. Rare: alcohol intolerance.
Cardiovascular Disorders, General: Infrequent: hypotension, postural hypotension, angina pectoris.
Central & Peripheral Nervous System Disorders: Infrequent : neuropathy, apraxia, hyperaesthesia, dyskinesia, dysphonia, scotoma, ptosis, dystonia, visual field defect, encephalopathy, EEG abnormal. Rare: upper motor neuron lesion, cerebellar syndrome, tongue paralysis.
Gastrointestinal System Disorders: Infrequent: hemorrhoids, stomatitis, melena, gastritis, esophagitis. Rare: tongue edema.
Heart Rate and Rhythm Disorders: Infrequent: AV block.
Liver and Biliary System Disorders: Infrequent: SGPT increased, SGOT increased.
Metabolic and Nutritional Disorders: Infrequent: dehydration, hypokalemia, alkaline phosphatase increased, hypocalcemia, hyperlipemia, hyperglycemia, xerophthalmia, diabetes mellitus. Rare: hyperchloremia, hypernatremia, hyponatremia, hypocholesterolemia, hypophosphatemia, creatinine increased.
Musculoskeletal System Disorders: Frequent: arthralgia. Infrequent: arthrosis.
Neoplasms: Infrequent: thrombocythemia. Rare: polycythemia.
Platelet, Bleeding, and Clotting Disorders: Infrequent: gingival bleeding, pulmonary embolism.
Psychiatric Disorders: Frequent: impotence, hallucination, psychosis, suicide attempt. Infrequent: euphoria, paranoid reaction, delusion, paranoia, delirium, abnormal dreaming. Rare: libido increased, manic reaction.
Red Blood Cell Disorders: Frequent: anemia. Rare: marrow depression, pancytopenia.
Reproductive Disorders, Male: Infrequent: ejaculation disorder, breast discharge.
Skin and Appendages Disorders: Infrequent: urticaria, photosensitivity reaction, abnormal hair texture. Rare: chloasma.
Special Senses Other, Disorders: Infrequent: taste loss, parosmia.
Urinary System Disorders: Infrequent: urinary retention, face edema, renal pain, albuminuria, polyuria, oliguria.
Vascular (Extracardiac) Disorders: Infrequent: flushing, deep vein thrombosis, phlebitis. Rare: vasospasm.
Vision Disorders: Frequent: conjunctivitis. Infrequent: abnormal accommodation, photophobia, strabismus. Rare: mydriasis, iritis.
White Cell and Reticuloendothelial System Disorders: Infrequent: lymphadenopathy, eosinophilia, lymphopenia, granulocytopenia. Rare: lymphocytosis.
Postmarketing and Other Experience
In addition to the adverse experiences reported during clinical testing of topiramate, the following adverse experiences have been reported worldwide in patients receiving topiramate post-approval.
These adverse experiences have not been listed above and data are insufficient to support an estimate of their incidence or to establish causation. The listing is alphabetized: bullous skin reactions (including erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis), hepatic failure (including fatalities), hepatitis, maculopathy, pancreatitis, pemphigus, and renal tubular acidosis.
DRUG ABUSE AND DEPENDENCE
The abuse and dependence potential of topiramate has not been evaluated in human studies.
OVERDOSAGE
Overdoses of topiramate have been reported. Signs and symptoms included convulsions, drowsiness, speech disturbance, blurred vision, diplopia, mentation impaired, lethargy, abnormal coordination, stupor, hypotension, abdominal pain, agitation, dizziness and depression. The clinical consequences were not severe in most cases, but deaths have been reported after poly-drug overdoses involving topiramate.
Topiramate overdose has resulted in severe metabolic acidosis (see WARNINGS).
A patient who ingested a dose between 96 and 110 g topiramate was admitted to hospital with coma lasting 20 to 24 hours followed by full recovery after 3 to 4 days.
In acute topiramate overdose, if the ingestion is recent, the stomach should be emptied immediately by lavage or by induction of emesis. Activated charcoal has been shown to adsorb topiramate in vitro. Treatment should be appropriately supportive. Hemodialysis is an effective means of removing topiramate from the body.
HOW SUPPLIED
Topiramate tablets are available as debossed, film-coated, circular tablets in the following strengths and colors:
25
mg white (coded "S" on one side; "707" on the other)
50 mg yellow
(coded "S" on one side; "710" on the other)
100 mg yellow (coded
"S" on one side; "711" on the other)
200 mg brown (coded "S"
on one side; "712" on the other)
They are supplied as
follows:
25 mg tablets Bottles of 30's with Child Resistant Cap… …………NDC 62756-707-83
Bottles of 60's with Child Resistant Cap… …………NDC 62756-707-86
Bottles of 100's with Child Resistant Cap… … … …NDC 62756-707-88
Bottles of 100's with Non Child Resistant Cap………NDC 62756-707-08
Bottles of 500's with Non Child Resistant Cap………NDC 62756-707-13
Bottles of 1000's with Non Child Resistant Cap… …NDC 62756-707-18
50 mg tablets Bottles of 30's with Child Resistant Cap… …………NDC 62756-710-83
Bottles of 60's with Child Resistant Cap… …………NDC 62756-710-86
Bottles of 100's with Child Resistant Cap… … … …NDC 62756-710-88
Bottles of 100's with Non Child Resistant Cap………NDC 62756-710-08
Bottles of 500's with Non Child Resistant Cap………NDC 62756-710-13
Bottles of 1000's with Non Child Resistant Cap… …NDC 62756-710-18
100 mg tablets Bottles of 30's with Child Resistant Cap… …………NDC 62756-711-83
Bottles of 60's with Child Resistant Cap… …………NDC 62756-711-86
Bottles of 100's with Child Resistant Cap… … … …NDC 62756-711-88
Bottles of 100's with Non Child Resistant Cap………NDC 62756-711-08
Bottles of 500's with Non Child Resistant Cap………NDC 62756-711-13
Bottles of 1000's with Non Child Resistant Cap… …NDC 62756-711-18
200 mg tablets Bottles of 30's with Child Resistant Cap… …………NDC 62756-712-83
Bottles of 60's with Child Resistant Cap… …………NDC 62756-712-86
Bottles of 100's with Child Resistant Cap… … … …NDC 62756-712-88
Bottles of 100's with Non Child Resistant Cap………NDC 62756-712-08
Bottles of 500's with Non Child Resistant Cap………NDC 62756-712-13
Bottles of 1000's with Non Child Resistant Cap… …NDC 62756-712-18
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from moisture. Dispense in a tight container.
MEDICATION GUIDE
Topiramate Tablets
Read this Medication Guide before you start taking topiramate tablets and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. If you have any questions about topiramate tablets, talk to your healthcare provider or pharmacist.
What is the most important information I should know about topiramate tablets?
-
Topiramate tablets may cause eye problems. Serious eye problems include:
-
- any sudden decrease in vision with or without eye pain and redness
- a blockage of fluid in the eye causing increased pressure in the eye (secondary angle closure glaucoma).
- These eye problems can lead to permanent loss of vision if not treated. You should call your healthcare provider right away if you have any new eye symptoms.
- Topiramate tablets may cause decreased sweating and increased body temperature (fever). People, especially children, should be watched for signs of decreased sweating and fever, especially in hot temperatures. Some people may need to be hospitalized for this condition.
- Like other antiepileptic drugs, topiramate tablets may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
- thoughts about suicide or dying
- attempts to commit suicide
- new or worse depression
- new or worse anxiety
- feeling agitated or restless
- panic attacks
- trouble sleeping (insomnia)
- new or worse irritability
- acting aggressive, being angry, or violent
- acting on dangerous impulses
- an extreme increase in activity and talking (mania)
- other unusual changes in behavior or mood
Do not stop topiramate tablets without first talking to a healthcare provider.
- Stopping topiramate tablets suddenly can cause serious problems.
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
How can I watch for early symptoms of suicidal thoughts and actions?
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
What is topiramate tablet?
Topiramate tablet is a prescription medicine used:
- to treat certain types of seizures (partial onset seizures and primary generalized tonic-clonic seizures) in people 10 years and older
- with other medicines to treat certain types of seizures (partial onset seizures, primary generalized tonic-clonic seizures, and seizures associated with Lennox-Gastaut syndrome) in adults and children 2 years and older
What should I tell my healthcare provider before taking topiramate tablets?
Before taking topiramate tablets, tell your healthcare provider about all your medical conditions, including if you:
- have or have had depression, mood problems or suicidal thoughts or behavior
- have kidney problems, kidney stones, or are getting kidney dialysis
- have a history of metabolic acidosis (too much acid in the blood)
- have liver problems
- have osteoporosis, soft bones, or decreased bone density
- have lung or breathing problems
- have eye problems, especially glaucoma
- have diarrhea
- have a growth problem
- are on a diet high in fat and low in carbohydrates, which is called a ketogenic diet
- are having surgery
- are pregnant or plan to become pregnant. It is not known if topiramate tablets will harm your unborn baby. If you become pregnant while taking topiramate tablets, talk to your healthcare provider about registering with the North American Antiepileptic Drug Pregnancy Registry. You can enroll in this registry by calling 1-888-233-2334. The purpose of this registry is to collect information about the safety of antiepileptic medicine during pregnancy.
- are breastfeeding. It is not known if topiramate passes into breast milk and if it can harm your baby. Talk to your healthcare provider about the best way to feed your baby if you take topiramate tablets.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Topiramate tablets and other medicines may affect each other causing side effects.
Especially, tell your healthcare provider if you take:
- Valproic acid
- any medicines that impair or decrease your thinking, concentration, or muscle coordination.
- birth control pills. Topiramate tablets may make your birth control pills less effective. Tell your healthcare provider if your menstrual bleeding changes while you are taking birth control pills and topiramate tablets.
Ask you healthcare provider if you are not sure if your medicine is listed above.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine. Do not start a new medicine without talking with your healthcare provider.
How should I take topiramate tablets?
- Take topiramate tablets exactly as prescribed.
- Your healthcare provider may change your dose. Do not change your dose without talking to your healthcare provider.
- Topiramate tablets should be swallowed whole. Do not chew the tablets. They may leave a bitter taste.
- Do not store any medicine and food mixture for later use.
- Topiramate tablets can be taken before, during, or after a meal. Drink plenty of fluids during the day. This may help prevent kidney stones while taking topiramate tablets.
- If you take too many topiramate tablets, call your healthcare provider or poison control center right away or go to the nearest emergency room.
- If you miss a single dose of topiramate tablets, take it as soon as you can. However, if you are within 6 hours of taking your next scheduled dose, wait until then to take your usual dose of topiramate tablets, and skip the missed dose. Do not double your dose. If you have missed more than one dose, you should call your healthcare professional for advice.
- Do not stop taking topiramate tablets without talking to your healthcare provider. Stopping topiramate tablets suddenly may cause serious problems. If you have epilepsy and you stop taking topiramate tablets suddenly, you may have seizures that do not stop. Your healthcare provider will tell you how to stop taking topiramate tablets slowly.
- Your healthcare provider may do blood tests while you take topiramate tablets.
What should I avoid while taking topiramate tablets?
- Do not drink alcohol while taking topiramate tablets. Topiramate tablets and alcohol can affect each other causing side effects such as sleepiness and dizziness.
- Do not drive a car or operate heavy machinery until you know how topiramate tablets affect you. Topiramate tablets can slow your thinking and motor skills.
What are the possible side effects of topiramate tablets?
Topiramate tablets may cause serious side effects including:
See “What is the most important information I should know about topiramate tablets?”
- Metabolic Acidosis. Metabolic acidosis can cause:
-
- tiredness
- loss of appetite
- irregular heartbeat
- impaired consciousness
- High blood ammonia levels. High ammonia in the blood can affect your mental activities, slow your alertness, make you feel tired, or cause vomiting. This has happened when topiramate tablets are taken with a medicine called valproic acid.
- Kidney stones. Drink plenty of fluids when taking topiramate tablets to decrease your chances of getting kidney stones.
- Effects on Thinking and Alertness. Topiramate tablets may affect how you think, and cause confusion, problems with concentration, attention, memory, or speech. Topiramate tablets may cause depression or mood problems, tiredness, and sleepiness.
- Dizziness or Loss of Muscle Coordination.
Call your healthcare provider right away if you have any of the symptoms above.
The most common side effects of topiramate tablets include:
- tingling of the arms and legs (paresthesia)
- not feeling hungry
- nausea
- a change in the way foods taste
- diarrhea
- weight loss
- nervousness
- upper respiratory tract infection
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects of topiramate tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store topiramate tablets?
- Store topiramate tablets at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F).
- Keep topiramate tablets in a tightly closed container
- Keep topiramate tablets dry and away from moisture
- Keep topiramate tablets and all medicines out of the reach of children.
General information about topiramate tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use topiramate tablet for a condition for which it was not prescribed. Do not give topiramate tablets to other people, even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about topiramate tablets. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about topiramate tablets that is written for health professionals.
For more information, call 1-800-818-4555
What are the ingredients in topiramate tablets?
Active ingredient: topiramate
Inactive ingredients: anhydrous lactose, microcrystalline cellulose, pregelatinized starch, sodium starch glycolate, magnesium stearate, purified water, polyvinyl alcohol, titanium dioxide, polyethylene glycol and talc.
In addition, individual tablets contain:
50 mg tablets: iron oxide yellow
100 mg tablets: iron oxide yellow, and D&C Yellow # 10 Aluminum Lake
200 mg tablets: iron oxide red, lecithin (soya), and iron oxide black
This Medication Guide has been approved by the U.S. Food and Drug Administration.
| TOPIRAMATE
topiramate tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Lake Erie Medical & Surgical Supply DBA Quality Care Products LLC (831276758) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Lake Erie Medical & Surgical Supply DBA Quality Care Products LLC | 831276758 | repack(35356-469) | |
