LACTATED RINGERS- lactated ringers injection, solution
WG Critical Care, LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Lactated Ringer’s Injection, USP
DESCRIPTION
Lactated Ringer’s Injection is a sterile, nonpyrogenic solution for fluid and electrolyte replenishment in single dose containers for parenteral administration. It contains no antimicrobial agents.
COMPOSITION:
Each 100 mL of intravenous infusion contains:
- Sodium chloride 600 mg
Electrolytes: The calcium, potassium and sodium contents are approximately 2.7, 4 and 130 milliequivalents per liter, respectively. Osmolarity: 273 mOsm/l
CLINICAL PHARMACOLOGY
A multiple electrolyte intravenous solution is intended to restore the electrolyte balance and water for hydration. A combination of multiple electrolytes and sodium lactate, an alkalinizing agent, will provide electrolyte balance and normalize the pH of the acid-base of the physiological system.
INDICATIONS
Lactated Ringer’s Injection is indicated for the correction of electrolyte depletion, metabolic acidosis and dehydration of cattle, calves, horses, sheep and swine.
CONTRAINDICATIONS
Lactated Ringer’s Injection is contraindicated in patients with a known hypersensitivity to sodium lactate; congestive heart failure or severe impairment of renal function; clinical states in which the administration of sodium and chloride is detrimental.
WARNINGS
The introduction of additives to any solution, regardless of type of container, requires special attention to ensure that no incompatibilities result. While some incompatibilities are readily absorbed, one must be aware that subtle physical, chemical and pharmacological incompatibilities can occur. The medical literature, the package insert and other available sources of information should be reviewed for thorough understanding of possible incompatibilities.
Lactated Ringer’s Injection should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency, and in clinical states in which there exists edema and sodium retention.
Lactated Ringer’s Injection should be used with great care, if at all, in patients with hyperkalemia, severe renal failure, and in conditions in which potassium retention is present.
Lactated Ringer’s Injection should be used with great care in patients with metabolic or respiratory alkalosis. The administration of lactate ions should be done with great care in those conditions in which there is an increased level or an impaired utilization of these ions, such as severe hepatic insufficiency.
Lactated Ringer’s Injection should not be administered simultaneously with blood through the same administration set because of likelihood of coagulation.
The intravenous administration of Lactated Ringer’s Injection can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, over-hydration, congested states, or pulmonary edema. The risk of dilutional states is inversely proportional to the electrolyte concentrations of the injections. The risk of solute overloading causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of the injections.
In patients with diminished renal function, administration of Lactated Ringer’s Injection may result in sodium or potassium retention.
Do not administer to horses by intraperitoneal injection. Do not administer to animals with inadequate renal function. Not for use in lactic acidosis.
ADVERSE REACTIONS
Adverse reactions may occur due to the solution or the technique of administration including fever response, infection at the site of injection or allergic reactions. Prolonged intravenous infusion of this type of product may cause venous thrombosis or phlebitis extending from the site of injection, extravasation, and hypervolemia.
If an adverse reaction does occur, discontinue the infusion and evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary.
PRECAUTIONS
This is a single dose unit. It contains no preservatives. Use entire contents when first opened.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid base balance during prolonged therapy or whenever the condition of the patient warrants such evaluation.
Lactated Ringer’s Injection should be used with caution. Excess administration may result in metabolic alkalosis.
Do not administer unless solution is clear and both seal and container are intact.
Solution must be warmed to body temperature prior to administration and administered at a slow rate. Use solution promptly following initial entry.
Reactions which may occur because of the solution or the technique of administration, include febrile response, infection at the site of injection, extravasation, and hypervolemia.
If an adverse reaction does occur, discontinue the infusion and evaluate the patient, institute appropriate therapeutic countermeasures, and save the remainder of the fluid for examination if deemed necessary.
DOSAGE AND ADMINISTRATION
To be used as directed by a licensed veterinarian. Dosage is dependent upon the age, weight and clinical conditions of the patient as well as laboratory determinations.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
For use in one patient on one occasion only. Discard any unused portion. Care should be taken with administration technique to avoid administration site reactions and infection.
Additives may be incompatible. Complete information is not available. Those additives known to be incompatible should not be used. Consult with pharmacist, if available. If, in the informed judgement of the doctor, it is deemed advisable to introduce additives, use aseptic technique. Mix thoroughly when additives have been introduced. Do not store solutions containing additives.
OVERDOSAGE:
In an event of over-hydration or solute overload, re-evaluate the patient and institute appropriate corrective measures. See Warnings, Precautions and Adverse Events.
PACKAGING
1000 mL bag with clear overwrap: NDC 44567-960-10
3000 mL bag with clear overwrap: NDC 44567-960-30
5000 mL bag with clear overwrap: NDC 44567-960-50
STORAGE:
Store below 25°C (77°F).
Keep out of reach of children.
Directions for Use of Plastic Container:
To Open
Tear overwrap at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing solution container firmly. If leaks are found, discard solution as sterility may be impaired. If supplemental medication is desired, follow directions below:
Preparation for Administration
(Use Aseptic Technique)
1. Close flow control clamp of administration set.
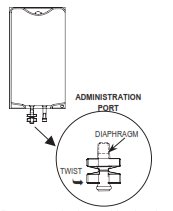
2. Twist off the protector cap from the administration port of the bags.
3. Insert the spike, a slight resistance should be felt as the port membrane is broken.
Use a twisting motion during insertion. Insert the spike until the shoulder of the spike is level with the port.
NOTE: See full directions on administration set packaging.
4. Suspend container from hanger.
5. Squeeze and release drip chamber to establish proper fluid level in chamber.
6. Open flow control clamp and clear air from set. Close clamp.
7. Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
8. Regulate rate of administration with flow control clamp.
WARNING: Do not use flexible container in series connections.
To Add Medication
WARNING: Additives may be incompatible.
To add medication before solution administration
1. Prepare medication site.
2. Using syringe with 0.63 mm to 0.80 mm needle, puncture medication port and inject.
3. Mix solution and medication thoroughly. For high density medication such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
To add medication during solution administration
1. Close the clamp on the administration set.
2. Prepare medication site.
3. Using syringe with 0.63 mm to 0.80 mm needle, puncture medication port and inject.
4. Remove container from IV pole and/or turn to an upright position.
5. Evacuate both ports by squeezing them while container is in the upright position.
6. Mix solution and medication thoroughly.
7. Return container to in use position and continue administration.
CAUTION:
Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Manufactured for:
WG Critical Care,LLC
Paramus, NJ 07652
Manfactured by:
INFOMED FLUIDS S.R.L., Bucharest, Romania
Revised March 2016
| LACTATED RINGERS
lactated ringers injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - WG Critical Care, LLC (829274633) |