ALLERGY RELIEF ANTIHISTAMINE- diphenhydramine hcl tablet
Cardinal Health (Leader) 49781
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
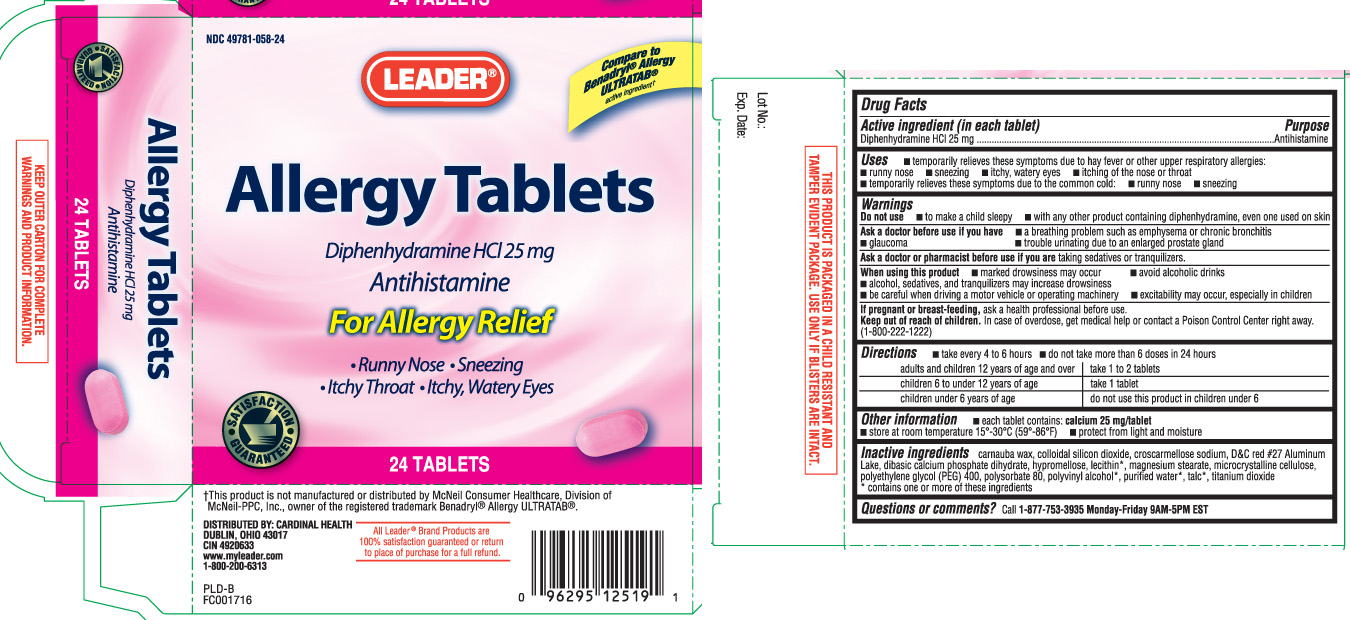
Drug Facts
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Directions
- take every 4 to 6 hours
- do not take more than 6 doses in 24 hours
| adults and children 12 years of age and over | take 1 to 2 tablets |
| children 6 to under 12 years of age | take 1 tablet |
| children under 6 years of age | do not use this product in children under 6 |
Other information
- each tablet contains: calcium 25 mg/tablet
- store at room temperature 15°-30°C (59°-86°F)
- protect from light and moisture
Inactive ingredients
carnauba wax, colloidal silicon dioxide, croscarmellose sodium, D&C red #27 Aluminum Lake, dibasic calcium phosphate dihydrate, hypromellose, lecithin*, magnesium stearate, microcrystalline cellulose, polyethylene glycol (PEG) 400, polysorbate 80, polyvinyl alcohol*, purified water*, talc*, titanium dioxide
*contains one or more of these ingredients
Principal Display Panel
Compare to Benadryl® Allergy ULTRATAB® active ingredient†
Allergy Tablets
Diphenhydramine HCl 25 mg
Antihistamine
For Allergy Relief
• Runny Nose • Sneezing
• Itchy Throat • Itchy, Watery Eyes
TABLETS
†This product is not manufactured or distributed by McNeil Consumer Healthcare, Division of McNeil-PPC, Inc., owner of the registered trademark Benadryl® Allergy ULTRATAB®.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
THIS PRODUCT IS PACKAGED IN A CHILD RESISTANT AND TAMPER EVIDENT PACKAGE. USE ONLY IF BLISTERS ARE INTACT.
DISTRIBUTED BY: CARDINAL HEALTH
DUBLIN, OH 43017
www.myleader.com
| ALLERGY RELIEF
ANTIHISTAMINE
diphenhydramine hcl tablet |
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||
| Labeler - Cardinal Health (Leader) 49781 (097537435) |