Label: 4379 FIRST AID KIT kit
4334 FIRST AID KIT kit
-
NDC Code(s):
0498-0121-00,
0498-0143-04,
0498-0203-00,
0498-0501-00, view more0498-0750-35, 0498-2110-01, 0498-4334-01, 0498-4379-01
- Packager: Honeywell Safety Products USA, INC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 22, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Triple Active ingredients
- Triple Purpose
- Triple Uses
- Triple Warnings
- Triple Directions
- Triple Other information
- Triple Inactive ingredient
- Alcohol Active ingredient
- Alcohol Purpose
- Alcohol Uses
- Alcohol Warnings
- Alcohol Directions
- Alcohol Other information
- Alcohol Inactive ingredient
- Foille Active ingredient
- Foille Purpose
- Foille Uses
- Foille Warnings
- Foille Directions
- Foille Other information
- Foille Inactive ingredients
- Burn Jel Active ingredient
- Burn Jel Purpose
- Burn Jel Uses
- Burn Jel Warnings
- Burn Jel Directions
- Burn Jel Other information
- Burn Jel Inactive ingredients
- Burn Jel Questions
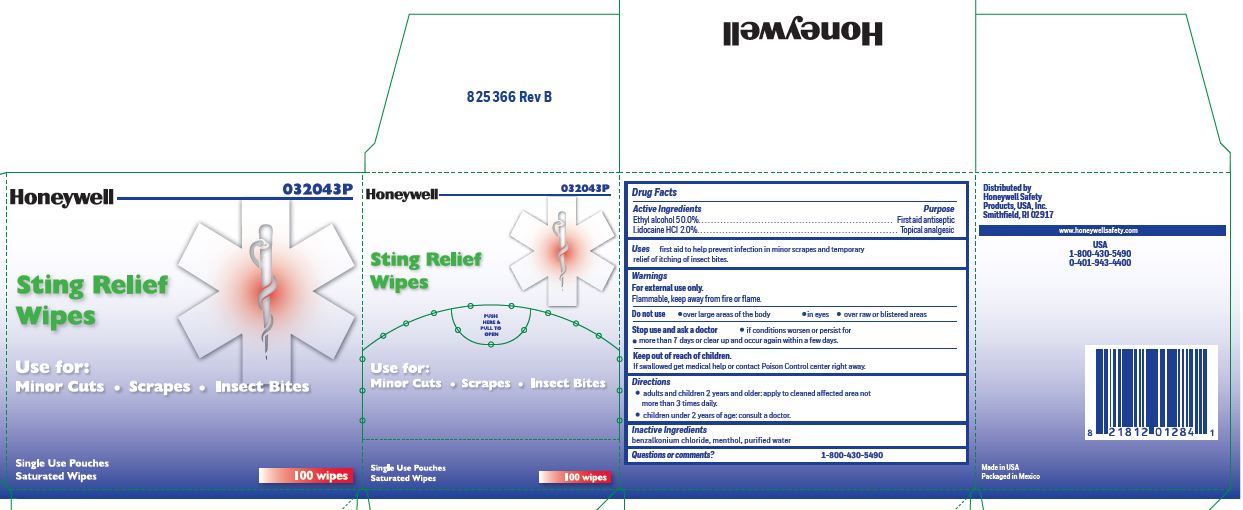
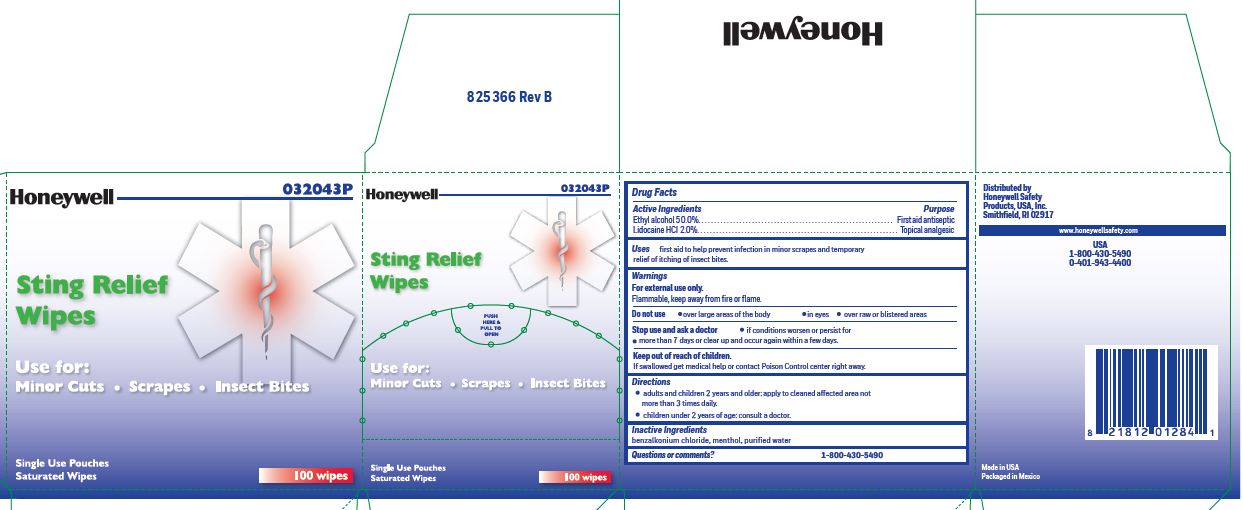
- Sting Relief Active ingredients (in each wipe)
- Sting Relief Purpose
- Sting Relief Uses
- Sting Relief Warnings
- Sting Relief Directions
- Sting Relief Inactive ingredients
- Sting Relief Questions or Comments?
- PVP Wipe Active ingredient
- PVP Purpose
- PVP USes
- PVP Warnings
- PVP Directions
- PVP Other information
- PVP Inactive ingredients
- PVP Questions
- Aypanal EX Active ingredient
- Aypanal EX Purpose
- Aypanal EX Uses
-
Aypanal EX
Warnings
Liver Warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4,000 mg in 24 hours, which is the maximum daily amount.
- with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If skin reaction occurs, stop use and seek medical help right away
Do Not Use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
Stop use and ask a doctor if
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
- Aypanal EX Directions
- Aypanal EX Other information
- Aypanal EX Inactive ingredients
- Aypanal EX Questions or Comments?
- BZK Active ingredient
- BZK Purpose
- BZK Uses
-
BZK
Warnings
For external use only
Do not use
- in the eyes or over large areas of the body
- on mucous membranes
- on irritated skin
- in case of deep puncture wounds, animal bites or serious burns, consult a doctor
- longer than 1 week unless directed by a doctor
- BZK Directions
- BZK Other information
- BZK Inactive ingredients
- BZK Questions
-
4334
Z019819 KIT CONTENTS
1 TRIPLE ANTIBIOTIC 10 PER
1 INSTANT COLD PACK 4" X 6"
2 ADHESIVE BDG,PLSTIC,1"X3"16PER
1 BURN JEL 1/8 OZ, 6 PER
1 ALCOHOL PREP PADS 10P
1 PVP IODINE WIPES 10 PER
1 NITRILE GLOVES 2PR BBP
1 ANTIMCRBL ANTSPTC TWLETTS
1 ADHESIVE TAPE W/P 1/2"X 5 YD
1 TWEEZER PLASTICS 4"
1 FLEXICON 2"X 4.1 YD
1 FIRST AID GUIDE ASHI
1 ABD COMBINE PAD 5" X 9"
1 SCISSOR BDGE 4" RED PLS HDL
LBL STOCK 6-3/8"X4"
LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 LBL CONTS 6 3/4"X3 1/2" ID B
1 LABEL COVER, GRAINGER Z019819
1 KIT PP 24 UNIT FA
3 SAFETEC STING RELIEF WIPES BULK
1 FOILLE BURN/F A OINT 1/2 OZ
4 GAUZE PADS 2"X2" 12PLY
1 GAUZE PADS 4"X4" 12PLY
4 WOVEN FINGERTIP BANDAGE 2"
4 WOVEN KNUCKLE BANDAGE
4 HEAVY FLEX LARGE PATCH 2" X 3"
1 GAUZE PADS 3"X3" 4/BX
1 TRIANG 37X37X52 UNIT
8 AYPANAL EXTRA BULK 2/PK
-
4379
Z63158002 Kit Contents
1 TRIPLE ANTIBIOTIC 10 PER
1 INSTANT COLD PACK 4" X 6"
2 ADHESIVE BDG,PLSTIC,1"X3"16PER
1 BURN JEL 1/8 OZ, 6 PER
1 ALCOHOL PREP PADS 10P
1 PVP IODINE WIPES 10 PER
1 NITRILE GLOVES 2PR BBP
1 ANTIMCRBL ANTSPTC TWLETTS
1 ADHESIVE TAPE W/P 1/2"X 5 YD
1 TWEEZER PLASTICS 4"
1 FLEXICON 2"X 4.1 YD
1 FIRST AID GUIDE ASHI
1 ABD COMBINE PAD 5" X 9"
1 SCISSOR BDGE 4" RED PLS HDL
LBL STOCK 6-3/8"X4"
LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 LBL CONTS 6 3/4"X3 1/2" ID B
1 LABEL COVER, GRAINGER Z019819
1 KIT PP 24 UNIT FA
3 SAFETEC STING RELIEF WIPES BULK
1 FOILLE BURN/F A OINT 1/2 OZ
4 GAUZE PADS 2"X2" 12PLY
1 GAUZE PADS 4"X4" 12PLY
4 WOVEN FINGERTIP BANDAGE 2"
4 WOVEN KNUCKLE BANDAGE
4 HEAVY FLEX LARGE PATCH 2" X 3"
1 GAUZE PADS 3"X3" 4/BX
1 TRIANG 37X37X52 UNIT
8 AYPANAL EXTRA BULK 2/PK
- Triple Principal Display Panel
- Alcohol Principal Display Panel
- Foille Principal Display Panel
- Burn Jel Principal Display Panel
- Sting Relief Principal Display Panel
- PVP Principal Display Panel
- Aypanal EX Principal Display Panel
- BZK Principal Display Panel
- 4334 Kit Label Z019819
- 4373 Kit Label Z63158002
-
INGREDIENTS AND APPEARANCE
4379 FIRST AID KIT
4379 first aid kit kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-4379 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-4379-01 1 in 1 KIT; Type 0: Not a Combination Product 10/18/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 10 POUCH 4 mL Part 2 1 TUBE 14 g Part 3 6 PACKET 21 g Part 4 1 PACKET 1.4 mL Part 5 3 POUCH 1.2 mL Part 6 10 POUCH 3 mL Part 7 8 PACKET 16 Part 8 10 PACKET 9 g Part 1 of 8 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC:0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 2 of 8 BLISTEX FOILLE MEDICATED FIRST AID
benzocaine and chloroxylenol ointmentProduct Information Item Code (Source) NDC:10157-9302 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLOROXYLENOL (UNII: 0F32U78V2Q) (CHLOROXYLENOL - UNII:0F32U78V2Q) CHLOROXYLENOL 0.1 g in 100 g BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 5 g in 100 g Inactive Ingredients Ingredient Name Strength CERESIN (UNII: Q1LS2UJO3A) EUGENOL (UNII: 3T8H1794QW) MALEIC ANHYDRIDE (UNII: V5877ZJZ25) POLYETHYLENE GLYCOL 1500 (UNII: 1212Z7S33A) CORN OIL (UNII: 8470G57WFM) SODIUM BORATE (UNII: 91MBZ8H3QO) YELLOW WAX (UNII: 2ZA36H0S2V) CALCIUM HYDROXIDE (UNII: PF5DZW74VN) WATER (UNII: 059QF0KO0R) SODIUM LAURYL SULFATE (UNII: 368GB5141J) BENZYL ALCOHOL (UNII: LKG8494WBH) EDETATE CALCIUM DISODIUM ANHYDROUS (UNII: 8U5D034955) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 14 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/05/2013 Part 3 of 8 BURN JEL
gel for burns gelProduct Information Item Code (Source) NDC:0498-0203 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 2 g in 100 g Inactive Ingredients Ingredient Name Strength TROLAMINE (UNII: 9O3K93S3TK) CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) PROPYLPARABEN (UNII: Z8IX2SC1OH) OCTOXYNOL-9 (UNII: 7JPC6Y25QS) TEA TREE OIL (UNII: VIF565UC2G) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) EDETATE DISODIUM (UNII: 7FLD91C86K) DIPROPYLENE GLYCOL (UNII: E107L85C40) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0203-00 3.5 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/19/2018 Part 4 of 8 ANTISEPTIC TOWELETTE
benzalkonium chloride liquidProduct Information Item Code (Source) NDC:0498-0501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0501-00 1.4 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/21/2017 Part 5 of 8 STING RELIEF PAD
ethyl alcohol, lidocaine swabProduct Information Item Code (Source) NDC:0498-0733 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 20 mg in 1 mL ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.5 mL in 1 mL Inactive Ingredients Ingredient Name Strength MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/23/2017 Part 6 of 8 PVP IODINE WIPE
povidone-iodine 10% swabProduct Information Item Code (Source) NDC:0498-0121 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength NONOXYNOL-9 (UNII: 48Q180SH9T) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0121-00 0.3 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 7 of 8 AYPANAL EX
acetaminophen tabletProduct Information Item Code (Source) NDC:0498-2110 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE (UNII: FZ989GH94E) Product Characteristics Color white Score no score Shape ROUND Size 12mm Flavor Imprint Code FR1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-2110-01 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/02/2017 Part 8 of 8 TRIPLE ANTIBIOTIC
bacitracin zinc, polymyxin b sulfate, neomycin sulfate ointmentProduct Information Item Code (Source) NDC:0498-0750 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0750-35 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/19/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/18/2018 4334 FIRST AID KIT
4334 first aid kit kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-4334 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-4334-01 1 in 1 KIT; Type 0: Not a Combination Product 10/18/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 PACKET 1.4 mL Part 2 3 POUCH 1.2 mL Part 3 10 POUCH 3 mL Part 4 8 PACKET 16 Part 5 10 PACKET 9 g Part 6 10 POUCH 4 mL Part 7 1 TUBE 14 g Part 8 6 PACKET 21 g Part 1 of 8 ANTISEPTIC TOWELETTE
benzalkonium chloride liquidProduct Information Item Code (Source) NDC:0498-0501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0501-00 1.4 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/21/2017 Part 2 of 8 STING RELIEF PAD
ethyl alcohol, lidocaine swabProduct Information Item Code (Source) NDC:0498-0733 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 20 mg in 1 mL ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.5 mL in 1 mL Inactive Ingredients Ingredient Name Strength MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/23/2017 Part 3 of 8 PVP IODINE WIPE
povidone-iodine 10% swabProduct Information Item Code (Source) NDC:0498-0121 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength NONOXYNOL-9 (UNII: 48Q180SH9T) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0121-00 0.3 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 4 of 8 AYPANAL EX
acetaminophen tabletProduct Information Item Code (Source) NDC:0498-2110 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE (UNII: FZ989GH94E) Product Characteristics Color white Score no score Shape ROUND Size 12mm Flavor Imprint Code FR1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-2110-01 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/02/2017 Part 5 of 8 TRIPLE ANTIBIOTIC
bacitracin zinc, polymyxin b sulfate, neomycin sulfate ointmentProduct Information Item Code (Source) NDC:0498-0750 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0750-35 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/19/2018 Part 6 of 8 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC:0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 7 of 8 BLISTEX FOILLE MEDICATED FIRST AID
benzocaine and chloroxylenol ointmentProduct Information Item Code (Source) NDC:10157-9302 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLOROXYLENOL (UNII: 0F32U78V2Q) (CHLOROXYLENOL - UNII:0F32U78V2Q) CHLOROXYLENOL 0.1 g in 100 g BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 5 g in 100 g Inactive Ingredients Ingredient Name Strength SODIUM LAURYL SULFATE (UNII: 368GB5141J) BENZYL ALCOHOL (UNII: LKG8494WBH) EDETATE CALCIUM DISODIUM ANHYDROUS (UNII: 8U5D034955) CERESIN (UNII: Q1LS2UJO3A) EUGENOL (UNII: 3T8H1794QW) MALEIC ANHYDRIDE (UNII: V5877ZJZ25) POLYETHYLENE GLYCOL 1500 (UNII: 1212Z7S33A) CORN OIL (UNII: 8470G57WFM) SODIUM BORATE (UNII: 91MBZ8H3QO) YELLOW WAX (UNII: 2ZA36H0S2V) CALCIUM HYDROXIDE (UNII: PF5DZW74VN) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 14 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/05/2013 Part 8 of 8 BURN JEL
gel for burns gelProduct Information Item Code (Source) NDC:0498-0203 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 2 g in 100 g Inactive Ingredients Ingredient Name Strength TROLAMINE (UNII: 9O3K93S3TK) CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) PROPYLPARABEN (UNII: Z8IX2SC1OH) OCTOXYNOL-9 (UNII: 7JPC6Y25QS) TEA TREE OIL (UNII: VIF565UC2G) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) EDETATE DISODIUM (UNII: 7FLD91C86K) DIPROPYLENE GLYCOL (UNII: E107L85C40) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0203-00 3.5 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/19/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/18/2018 Labeler - Honeywell Safety Products USA, INC (118768815)