4272 FIRST AID KIT- 4272 first aid kit
Honeywell Safety Products USA, INC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
0498-4272: First Aid Kit (Pyrocaine Sp, EW, Burn Jel, PVP wipe, 1st aid Pack: triple, amm. Inh, Sting relief- SF00001416)
Eyewash
Uses
- for flushing the eye to remove loose foreign material, air pollutants or chlorinated water
Eyewash
Warnings
For external use only Obtain immediate medical treatment for all open wounds in or near eyes. To avoid contamination, do not touch tip of container to any surface. Do not reuse. Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
Eyewash
Directions
- remove contacts before using
- twist top to remove
- flush the affected area as needed
- control rate of flow by pressure on the bottle
- if necessary, continue flushing with emergency eyewash or shower
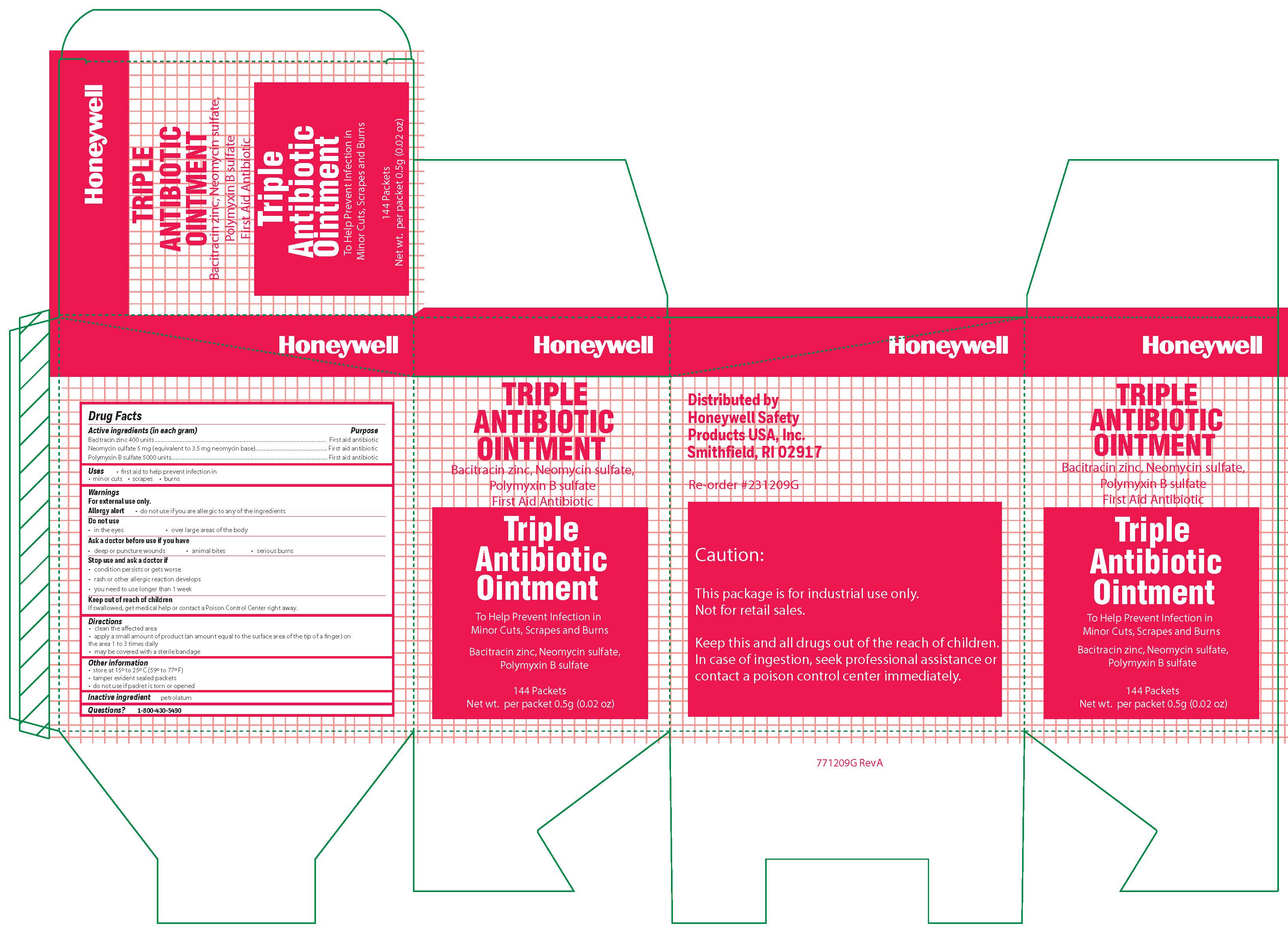
Triple
Active ingredient (each gram contains)
Bacitracin zinc 400 units - Neomycin sulfate (5 mg equivalent to 3.5 mg Neomycin base) Polymyxin B sulfate 5000 units
Triple
Warnings
For external use only
Allergy alert do not use if you are allergic to any of the ingredients
Triple
Directions
- clean the affected area
- apply a small amount of the product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Triple
Other information
store at 15 0 to 25 0 C (59 0 to 77 0 F) tamper evident sealed packets - do not use if packet is torn or opened
Ammonia
Directions
- hold inhalant away from face and crush ampoule between thumb and forefinger at position indicated on sleeve.
- hold near nostrils for inhalation of volatile vapor
Ammonia
Inactive ingredient
alcohol USP, FD&C red #40, lavender oil, lemon oil fcc, nutmeg oil, purified water
Pyrocaine
Uses
For the temporary relief of pain and itching, and to help protect against skin infection in:
- minor burns
- minor skin irritations
- minor cuts and scrapes
- insect bites
- sunburns
Pyrocaine
Warnings
For external use only
Flammable
- keep away from fire or flame
- contents under pressure
- do not puncture, incinerate or expose container to temperatures above 120 o F
Pyrocaine
Directions
- clean the affected area
- shake can well before using
- hold can 6 to 12 inches away from the affected area and spray liberally
- apply to affected area not more than 3 times daily
- for adult institutional use only
- not intended for use on children
Pyrocaine
Other information
- avoid inhaling
- use only as directed
- intentional misuse by deliberately concentrating and inhaling the contents may be harmful or fatal
Burn Jel
Warnings
For external use only
Burn Jel
Directions
- adults and children 2 years of age and older; apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
- you may report a serious reaction to this product to 800-430-5490
Burn Jel
Inactive ingredients
carbopol 940, carbopol 1342, diazolidinyl urea, glycerin, melaleuca alternifolia (tea tree) leaf oil, methylparaben, octoxynol-9, propylene glycol, propylparaben, trolamine, water
PVP
Warnings
For external use only.
PVP
Directions
- clean the affected area
- apply1 to 3 times daily
- may be covered with a sterile bandage
- if bandaged, let dry first
- discard wipe after single use
PVP
Other information
- do not use on individuals who are allergic or sensitive to iodine
- store at controlled temperature 59-86ºF (15-30ºC)
- do not use if pouch is open or torn
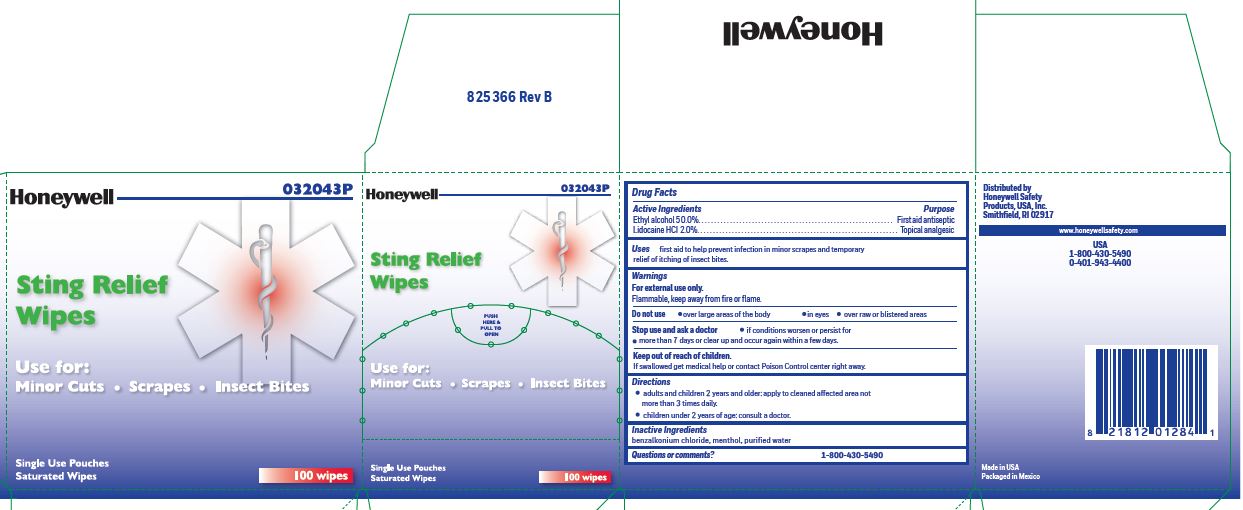
Sting Relef
Uses
- prevent infection in minor scrapes, and temporary relief of itching of insect bites
Sting Relief
Warnings
For external use only
Flammable, keep away from open fire or flame
Sting Relief
Directions
- adults and children 2 years and older: Apply to cleaned affected area not more than 3 times daily.
- children under 2 years of age: consult a doctor.
4272
SF00001416 Kit Contents
1 TRIPLE ANTIBIOTIC 10 PER
1 AMMONIA INHALANTS 10 PER
1 GAUZE BANDAGE, 4" X 6 YD
1 TOURNIQUET, 1 PER
2 TRIANGULAR BDG, NON-STERILE
1 ADH TAPE, .5" X 2.5 YD, 2 PER
1 GAUZE COMPRESS, 1728 SQ IN 1
2 INSTANT COLD PACK 4" X 6"
3 ADHESIVE BDG,PLSTIC,1"X3"16PER
1 1 OZ EYE WASH W/PADS & STRIPS
1 BURN JEL 1/8 OZ, 6 PER
1 WATER JEL DRESSING 4" X 4"
1 FIRST AID GUIDE ASHI
1 BLOODSTOPPER
1 SCISSOR BDGE 4" RED PLS HDL
1 KIT TWEEZER 3 1/2" SLANTED
LBL STOCK 6-3/8"X4"
LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
1 PR LRG NITRILE GLVES ZIP BAG
1 KIT PP 24 UNIT FA
2 PVP IODINE SWABS 10
1 STING Relief SWAB 10
1 PYRO-CAINE AERO 2/BX
| 4272 FIRST AID KIT
4272 first aid kit kit |
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
| Labeler - Honeywell Safety Products USA, INC (118768815) |