GENTEAL SEVERE- hypromellose gel
Novartis Pharmaceuticals Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Uses
- temporarily relieves discomfort due to minor irritations of the eye or to exposure to wind or sun
- as a protectant against further irritation or to relieve dryness of the eye
Do not use
- if gel changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Inactive ingredients
carbopol 980, phosphonic acid, purified water, sodium hydroxide, sodium perborate, and sorbitol
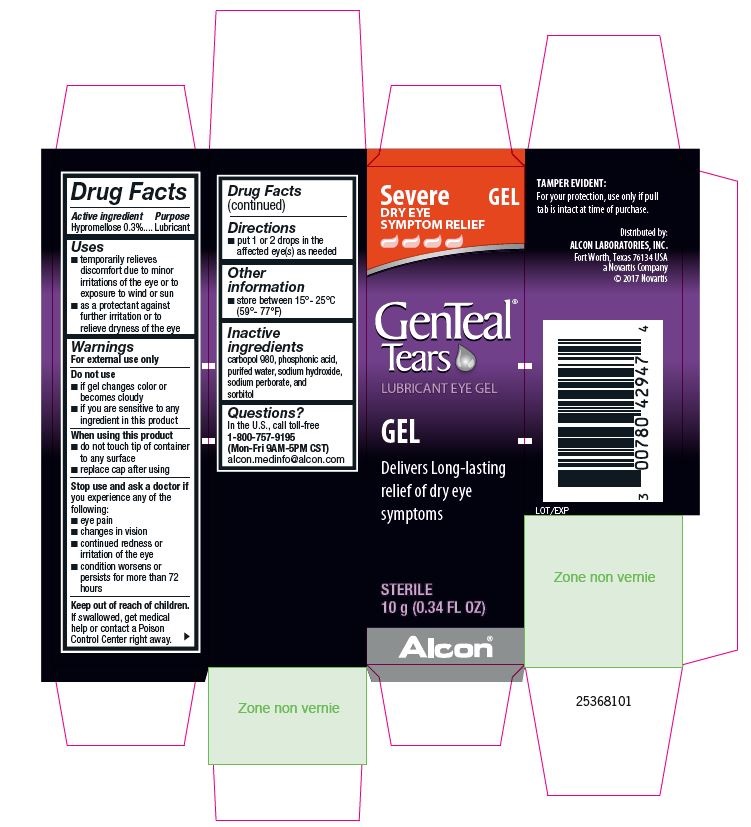
PRINCIPAL DISPLAY PANEL
Severe DRY EYE SYMPTOM RELIEF
GEL
GenTeal® Tears
LUBRICANT EYE GEL
GEL
Delivers Long-lasting relief of dry eye symptoms
STERILE
10 g (0.34 FL OZ)
Alcon®
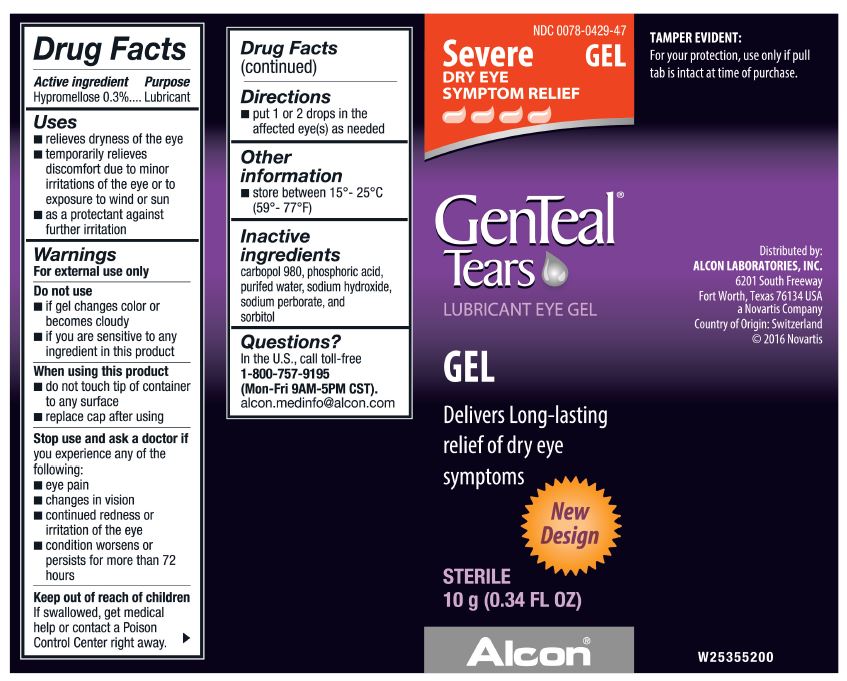
NDC 0078-0429-47
Severe
DRY EYE SYMPTOM RELIEF
GEL
GenTeal® Tears
LUBRICANT EYE GEL
GEL
Delivers Long-lasting relief of dry eye symptoms
New Design
STERILE
10 g (0.34 FL OZ)
Alcon®
Distributed by:
ALCON LABORATORIES, INC.
6201 South Freeway
Fort Worth, Texas 76134 USA
a Novartis Company
Country of Origin: Switzerland
© 2016 Novartis
| GENTEAL
SEVERE
hypromellose gel |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Novartis Pharmaceuticals Corporation (002147023) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Akorn AG | 482198285 | manufacture(0078-0429) , label(0078-0429) , pack(0078-0429) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Excelvision | 274234566 | manufacture(0078-0429) , label(0078-0429) , pack(0078-0429) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SERVIPACK | 571772875 | label(0078-0429) , pack(0078-0429) | |