4248 FIRST AID KIT- 4248 first aid kit
Honeywell Safety Products USA, INC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
0498-4248: First Aid Kit (Ammon, BZK , 1st aid Spray WS, alcohol wipe, Aypanal, Miralac, Burn Sp WS, Triple, EW, HC cr,, Pain stopper, sting relief, sinus decongest, tetrasine, cold spray- 68600PROAM )
Eyewash
Uses
- for flushing the eye to remove loose foreign material, air pollutants or chlorinated water
Eyewash
Warnings
For external use only Obtain immediate medical treatment for all open wounds in or near eyes. To avoid contamination, do not touch tip of container to any surface. Do not reuse. Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
Eyewash
Directions
- remove contacts before using
- twist top to remove
- flush the affected area as needed
- control rate of flow by pressure on the bottle
- if necessary, continue flushing with emergency eyewash or shower
Ammonia
Directions
- hold inhalant away from face and crush ampoule between thumb and forefinger at position indicated on sleeve.
- hold near nostrils for inhalation of volatile vapor
Ammonia
Inactive ingredient
alcohol USP, FD&C red #40, lavender oil, lemon oil fcc, nutmeg oil, purified water
Water Soluble 1st Aid Spray
Uses
for temporary relief of pain and itching and helps protect against infection in
- minor cuts and scrapes
- insect bites
- minor skin irritations
Water Soluble 1st Aid Spray
Warnings
For external use only
Flammable
- keep away from fire or flame
- contents under pressure
- do not puncture or incinerate container
- do not expose to temperature above 120 0 F
Do not use
- in the eyes or other mucous membranes
- in cases of serious burns
- in case of deep orpuncture wounds
- for a prolonged period of time
- on large portion of the body
Water Soluble 1st Aid Spray
Directions
- clean the affected area
- shake can well before using
- hold 4 - 6 inches from surface and spray area until wet
- may be covered with a sterile bandage. If bandaged, let dry first
- for adult institutional use only
- not intended for use on children
Water Soluble 1st Aid Spray
Other information
- avoid inhaling
- use only as directed
- intentional misuse by deliberately concentrating and inhaling the contents may be harmful or fatal
Burn Relief Water Soluble
Active ingredients
Benzethonium chloride 0.2% w/w
Benzocaine 10% w/w
Menthol 0.33% w/w
Burn Relief Water Soluble
Uses
for the temporary relief of pain and itching and helps protect against infection in:
- minor cuts and scrapes
- burns
- sunburn
- insect bites
- minor skin irritations
Burn Relief Water Soluble
Warnings
For external use only
Flammable keep away from fire or flame
- contents under pressure
- do not puncture or incinerate container
- do not expose to temperatures above 120 0 F
Do not use
- in or near the eyes or other mucous membranes
- in case of serious burns
- in case of deep or puncture wounds
- for prolonged period of time
- on large portion of the body
Burn Relief Water Soluble
Directions
- clean the affected area
- shake can well before using
- hold 4 - 6 inches from surface and spray area until wet
- may be covered with a sterile bandage, if bandaged let dry first
- for adult institutional use only
- not intended for use on children
Burn Relief Water Soluble
Other information
- avoid inhaling
- use only as directed
- intentional misuse by deliberately concentrating or inhaling the contents may be harmful or fatal
Triple
Active ingredients
Bacitracin zinc 400 units
Neomycin sulfate (5 mg equivalent to 3.5 mg Neomycin base)
Polymyxin B sulfate 5000 units
Triple
Warnings
For external use only
Allergy alert do not use if you are allergic to any of the ingredients
Triple
Directions
- clean the affected area
- apply a small amount of the product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Triple
Other information
- store at 15 0 to 25 0 C (59 0 to 77 0 F)
- tamper evident sealed packets
- do not use if packet is torn or opened
Alcohol
Directions
- clean the affected area
- apply wipe to affected area 1 to 3 times daily
- may be covered with a sterile bandage
- discard wipe after single use
Miralac
Uses
for the relief of
- acid indigestion
- heartburn
- sour stomach
- upset stomach associated with these symptoms
Warnings
Ask a doctor before use if you have
- kidney stones
- calcium-restricted diet
Ask a doctor before use if you are
- presently taking a prescription drug. Antacids may interfere with certain prescription drugs
Miralac
Other information
- each tablet contains: calcium 170 mg
- sucrose free
- lactose free
- store at room temperature
- TAMPER EVIDENT PACKETS- DO NOT USE IF OPEN OR TORN
BZK
Warnings
For external use only
Do not use
- in the eyes or over large areas of the body
- on mucous membranes
- on irritated skin
- in case of deep puncture wounds, animal bites or serious burns, consult a doctor
- longer than 1 week unless directed by a doctor
BZK
Other information
- store at room temperature 15 0 to 30 0 C (5 0 - 86 0 F)
- do not reuse towelette
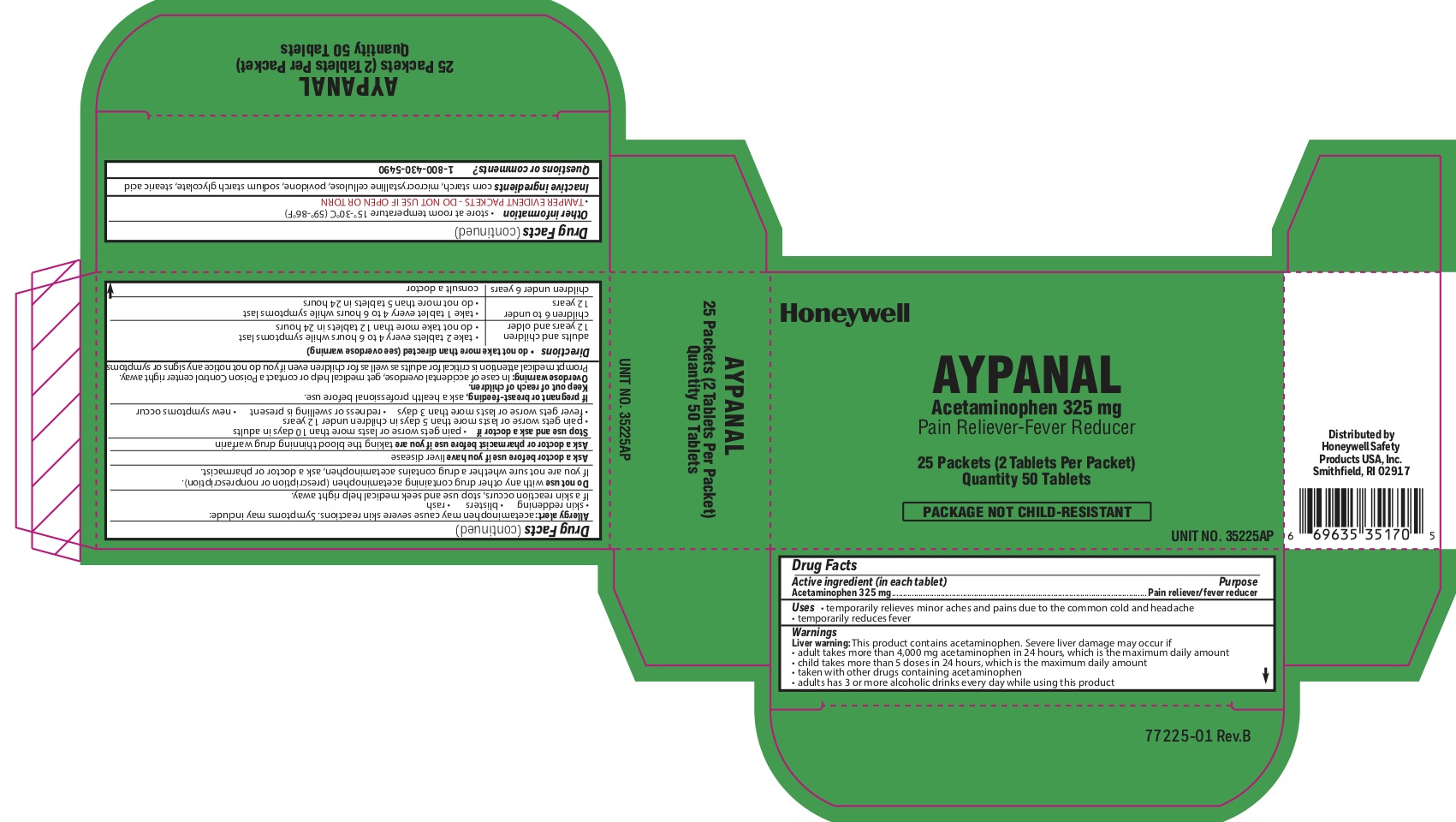
Aypanal
Uses
- temporarily relieves minor aches and pains due to the common cold and headache
- temporarily reduces fever
Aypanal
Warnings
Liver Warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4,000 mg in 24 hours, which is the maximum daily amount.
- with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If skin reaction occurs, stop use and seek medical help right away
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription).
- If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
Stop use and ask a doctor if
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
Keep out of reach of children.
Keep out of reach of children.
Overdose warning: In case ofl overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Prompt medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Aypanal
Directions
- do not take more than directed (see overdose warning)
- adults and children 12 years of age and over: Take 2 tablets with water every 6 hours while symptoms last.
- do not take any more than 8 tablets in 24 hours.
- children under 12: consult a doctor
Aypanal
Other information
- store at room temperature 15 0 -30 0 C (59 0 -86 0 F)
- TAMPER EVIDENT- DO NOT USE IF OPEN OR TORN
Aypanal
Inactive igredients
microcrystalline cellulose, povidone, sodium starch glycolate, starch, stearic acid
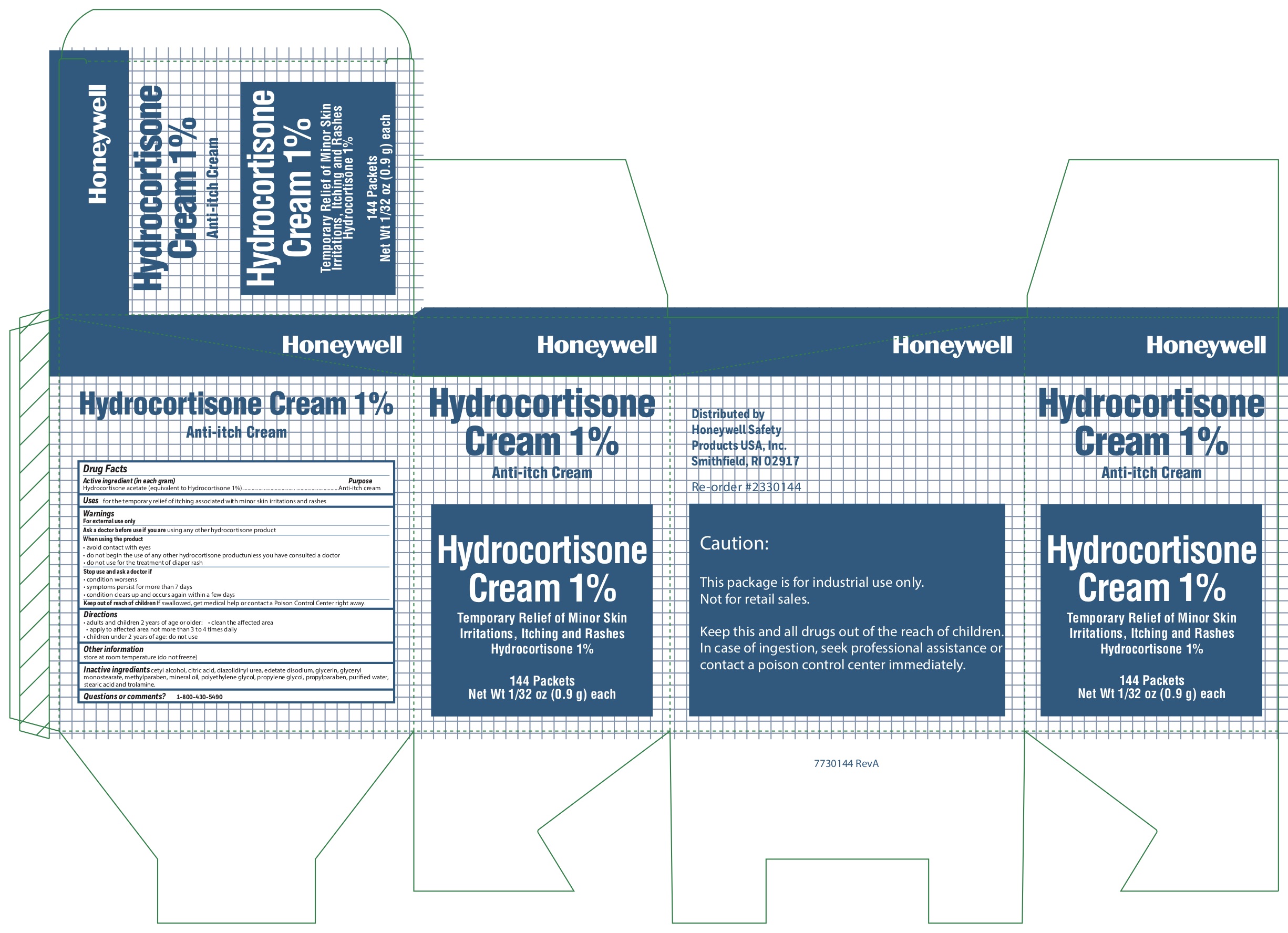
Hydrocortisone
Active ingredient (in each gram)
Hydrocortisone acetate (equivalent to Hydrocortisone 1%)
Hydrocortisone
Uses
- for the temporary relief of itching associated with minor skin irritations and rashes
Hydrocortisone
Warnings
For external use only
When using the product
- avoid contact with eyes
- do not begin use of any other hydrocortisone product unless you have consulted a doctor
- do not use for the treatment of diaper rash
Hydrocortisone
Directions
- adults and children 2 years and older:
- clean the affected area
- apply to the area not more than 3 to 4 times daily
- children under 2 years of age: consult a doctor
Hydrocortisone
Inactive ingredients
cetyl alcohol, citric acid, diazolidinyl urea, edetate disodium, glycerin, glyceryl monostearate, methylparaben, mineral oil, polyethylene glycol, propylene glycol, propylparaben, purified water, stearic acid, trolamine
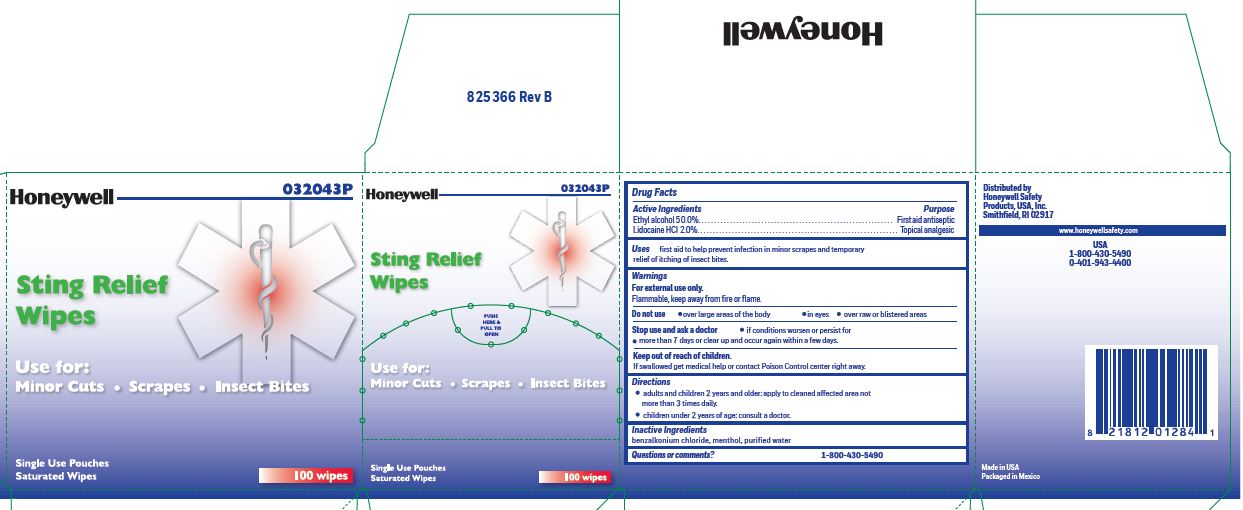
Sting Relief
Uses
- prevent infection in minor scrapes, and temporary relief of itching of insect bites
Sting Relief
Directions
- adults and children 2 years and older: Apply to cleaned affected area not more than 3 times daily.
- children under 2 years of age: consult a doctor.
Pain Stopper
Active ingredient (in each tablet)
Acetaminophen 110mg
Aspirin 162mg (NSAID)*
Caffeine 32.4mg
Salicylamide 152mg (NSAID)*
*nonsteroidal anti-inflammatory drug
Pain Stopper
Purpose
Pain reliever/fever reducer
Pain reliever/fever reducer
Diuretic
Pain reliever/fever reducer
Pain Stopper
Uses
for the temporary relief of minor aches and pains due to:
• common cold
• headache
• muscular aches
• premenstrual and menstrual cramps
Pain Stopper
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If skin reaction occurs, stop use and seek medical help right away.
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 12 tablets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist
- for more than 10 days for pain unless directed by a doctor
- for more than 3 days for fever unless directed by a doctor
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you are taking a diuretic
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
Stop using and ask a doctor if
- symptoms do not improve
- new symptoms occur
- pain or fever persists or gets worse
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pin that does not get better
- if ringing in the ears or a loss of hearing occurs, consult a doctor before taking any more of this product.
Pain Stopper
Directions
- adults and children 12 years of age and over, take 2 tablets every 4 hours while symptoms persist
- do not take more than 12 tablets in 24 hours
- children under 12 years: consult a doctor
Pain Stopper
Other information
- store at a controlled room temperature 15 0 -30 0 C (59 0 -86 0 F)
- TAMPER EVIDENT-DO NOT USE IF OPEN OR TORN
Pain Stopper
Inactive ingredients
FD&C Yellow #6, magnesium stearate, microcrystalline cellulose, povidone, starch, stearic acid,
Sinus Decongestant
Uses
- temporarily relieves these symptoms associated with hayfever and other upper respiratory allergies
- headache
- sinus congestion and pressure
- nasal congestion
- minor aches and pains
- helps decongest sinus openings and passages
Sinus Decongestant
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur
- if you take more than 4,00 mg acetaminophen in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If skin reaction occurs, stop use and seek medical attention right away.
Do not use
- with any other product containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibiter (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before use.
Ask a doctor before use if you have
- heart disease
- high blood pressure
- diabetes
- thyroid disease
- difficulty in urination due to enlargement of the prostate gland
- liver disease
Sinus Decongestant
Directions
do not take more than directed (see overdose warning)
Adults and children 12 years of age and older: take 2 tablets every 4-6 hours while symptoms persist
- do not exceed 8 tablets in 24 hours unless directed by a doctor
Children under 12 years: consult a doctor
Sinus Decongestant
Other information
- store at room temperature 15-30C (59-86F)
- avoid excessive heat and humidity
- TAMPER EVIDENT PACKETS - DO NOT USE IF OPEN OR TORN
Sinus Deacongestant
Inactive ingredients
FD&C Blue #1, microcrystalline cellulose, povidone, sodium starch glycolate, starch, stearic acid
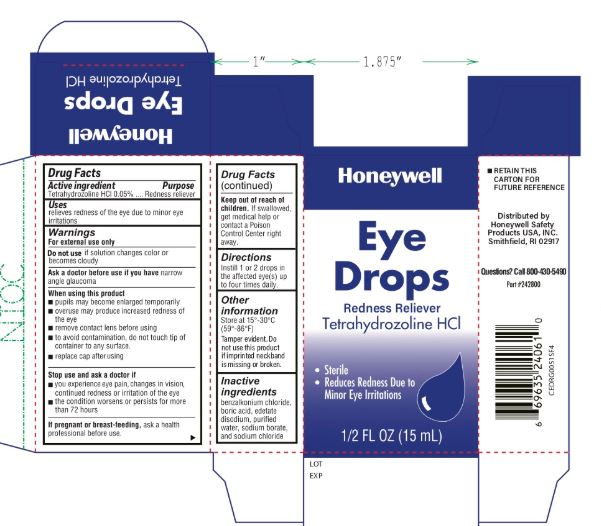
Tetrahydrozoline HCL
Warnings
For external use only
When using this product
- pupils may become enlarged temporarily
- overuse may produce increased redness of the eye
- remove contact lenses before using
- to avoid contamination, do not touch tip of container to any surface
- replace cap after using
Tetrahydrozoline HCL
Other information
Store at 15° to 30°C (59° to 86°F)
- Tamper evident. Do not use this product if imprinted neckband is missing or broken.
- RETAIN THIS CARTON FOR FUTURE REFERENCE
Distributed by
North by Honeywell
900 Douglas Pike
Smithfield, RI 02917
Tetrahydrozoline HCL
Inactive ingredients
benzalkonium chloride, boric acid, edetate disodium, purified water, sodium borate, sodium chloride
4248
68600PROAM KIT CONTENTS
1 1X3 PLASTIC 100/BOX
1 WOVEN 2" X 3" 25/BOX
1 FINGERTIP 8 WOVEN 25/BOX
1 FINGERTIP "T" WOVEN 40/BOX
1 BUTTERFLY CLOS MED 100/BX DS
1 SWIFT KNUCKLE 40/BX
1 1 X 3 WOVEN 100/BOX
1 AMMONIA INHALANTS 10 PER
1 EYE DRESS PKT W/4 ADH STRIPS
2 GAUZE BANDAGE, 4" X 6 YD
1 INSTANT COLD PACK 4" X 6"
1 HYDROCORTISON,1.O%,1/32 OZ,10P
1 ELASTIC TAPE 1" X 5YD
1 O/H TAPE ADHESIVE TRI-CUT
2 GAUZE BANDAGE 1" x 2 YDS
2 GAUZE BANDAGE 2"X2 YDS STRETCH GZ
2 GAUZE BANDAGE 4"X2 YDS STRETCH GZ
1 FIRST AID GUIDE ASHI
2 BLOODSTOPPER
1 NON ADHERENT PADS 2"X3" 50'S
1 ABD COMBINE PAD 5" X 9"
1 GZE PADS STERILE 4"X 4" 25'S
1 CO-FLEX BANDAGE 2"X 5YDS TAN
1 CPR FILTERSHIELD 77-100
1 COTTON TIPS 100 PER VIAL
1 ANTISEPTIC WIPES BZK CHL 20'S
1 FIRST AID SPRAY AEROSOL 3 OZ
1 ALCOHOL WIPES 50'S
1 AYPANAL NON-ASP IND 2/ENV 250
1 PAIN STOPPERS IND PK 2ENV 250
1 MIRALAC TABS IND PK 2/ENV 250
1 BURN SPRAY 3 OZ
1 SINUS DECONGESTANT 2/ENV 100
1 TRIPLE BIOTIC .5 GRAM PKT 20
1 HYDROCORTISONE 1% .9 GRM 20'S
1 TETRASINE EYE DROPS 1/2 OZ
2 HAND & SKIN LOTION 1GM 20/BX
1 SWIFT-STAT BLOOD CLOTTER 3 OZ
1 COLD SPRAY 4 OZ
1 POISON OAK/IVY CLEANSER 4 OZ
1 COLD PACK 5"X9" BOXED
2 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SPLINTER FORCEP 4 1/2"
1 SCISSOR LISTER BDG S/S 5 1/2"
1 POCKET INSERT RED #600 KIT
1 600 EMPTY KIT BLANK
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
1 LBL STOCK 3"x1-7/8"
2 2 PR LRG NITRILE GLVES ZIP BAG
6 WATER-JEL BURN DRESSING 2 X 6
6 WATER-JEL BURN DRESSING 2 X 2
2 TRI BNDG NON WOVEN 40"X40"X56"
1 STING Relief SWAB 10
1 RED BIO BAGS 2/BX
| 4248 FIRST AID KIT
4248 first aid kit kit |
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Honeywell Safety Products USA, INC (118768815) |